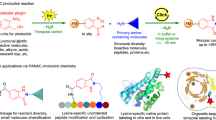
Abstract
A simple method for naked-eye detection of free amine groups during solid-phase peptide synthesis is reported. The technique involves base-catalyzed cyclization of 2-(2-oxo-2H-acenaphthylene-1-ylidene)-malononitrile (1) and subsequent oxidative substitution of aryl moiety by free amine group present at the N-terminus of the peptide chain that leads to the formation of a chromophore, which distinguishes the deprotected peptide chain from the protected one. The reaction is fast, does not require heating, and allows N-terminal modification of peptides suitable for imaging specific cells.
Graphical Abstract
Base catalyzed cyclization of 2-(2-oxo-2H-acenaphthylene-1-ylidene)-malononitrile and subsequent oxidative substitution by free amine group leads to the formation of a chromophore, which permits naked-eye detection of the deprotected peptide on resin support.










Similar content being viewed by others
References
Toy P H and Lam Y 2012 Solid-Phase Organic Synthesis: Concepts, Strategies, and Applications (Hoboken, NJ: Wiley)
Atherton E and Sheppard R C 1989 Solid Phase Peptide Synthesis: A Practical Approach (London and New York: Oxford University Press)
Chan W C and White P D 2000 Fmoc Solid Phase Peptide Synthesis: A Practical Approach (New York: Oxford University Press)
Kaiser E, Colescot R L, Bossinge C D and Cook P I 1970 Color test for detection of free terminal amino groups in solid-phase synthesis of peptides Anal. Biochem. 34 595
Sarin V K, Kent S B, Tam J P and Merrifield R B 1981 Quantitative monitoring of solid-phase peptide synthesis by the ninhydrin reaction Anal. Biochem. 117 147
Hodges R S and Merrifield R B 1975 Monitoring of solid phase peptide synthesis by an automated spectrophotometric picrate method Anal. Biochem. 65 241
Salisbury S A, Tremeer E J, John W, Davies A, David E I and Owen A 1990 Acylation monitoring in solid phase peptide synthesis by the equilibrium distribution of colored ions J. Chem. Soc. Chem. Commun. 538
Hancock W S and Battersby J E 1976 A new micro-test for the detection of incomplete coupling reactions in solid-phase peptide synthesis using 2, 4, 6-trinitrobenzene-sulphonic acid Anal. Biochem. 71 260
Gisin B F 1972 The monitoring of reactions in solid-phase peptide synthesis with picric acid Anal. Chim. Acta 58 248
Brunfeldt K and Christensen T 1972 Process monitoring in solid phase peptide synthesis, amino group blocking effect of impure methylene chloride FEES Lett. 19 345
Brunfeldt K and Halstrem J 1970 Tritylation of a partially protected pentapeptide synthesized by the Merrifield solid phase method Acta Chem. Scand. 24 3013
Brunfeldt K, Roepstorff P and Thomson J 1969 Process control in automated peptide synthesis In Peptides 1969 E Scoffone (Ed.) p. 148 (North Holland, Amsterdam: Elsevier)
Brunfeldt K, Christensen T and Villemoes P 1972 Automatic monitoring of solid phase synthesis of a decapeptide FEBS Lett. 22 238
Lee Y P and Takahashi T 1966 An improved colorimetric determination of amino acids with the use of ninhydrin Anal. Biochem. 14 71
Merrifield R B 1963 Solid phase peptide synthesis I: the synthesis of a tetrapeptide J. Am. Chem. Soc. 85 2149
Merrifield R B 1964 Solid-phase peptide synthesis III: an improved synthesis of bradykinin Biochemistry 3 1385
Peptide Synthesis Protocols 1995 In: Methods in Molecular Biology (Book Series) Vol. 35 Michael W Pennington and Ben M Dunn (Eds.) (Totowa, NJ: Humana Press Inc.)
Jullian M, Hernandez A, Maurras A, Puget K, Amblard M, Martinez J and Subra G 2009 N-terminus FITC labeling of peptides on solid support: the truth behind the spacer Tetrahedron Lett. 50 260
Walker J M 1984 The dansyl method for identifying N-terminal amino acids Methods Mol. Biol. 1 203
Birtalan E, Rudat B, Kölmel D K, Fritz D, Vollrath S B L, Schepers U and Braise S 2011 Investigating rhodamine B-labeled peptoids: scopes and limitations of its applications Biopolymers 96 694
Lenk R, Tessier A, Lefranc P, Silvestre V, Planchat A, Blot V, Dubreuil D and Lebreton J 2014 1-Oxo-1H-phenalene-2,3-dicarbonitrile heteroaromatic scaffold: revised structure and mechanistic studies J. Org. Chem. 79 9754
Acknowledgements
We thank Ms. I Janani for help in octapeptide binding to A431 cells. This work was carried out as part of the PhD program registered at the University of Madras, using a grant BSC0201 from Council of Scientific and Industrial Research, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rajavenkatesh, K., Santoshkumar, S., Purnasai, K. et al. A facile method for monitoring solid-phase peptide synthesis and for N-terminal modification of peptides: synthesis of short peptides for imaging specific cells. J Chem Sci 131, 108 (2019). https://doi.org/10.1007/s12039-019-1683-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-019-1683-9