Abstract
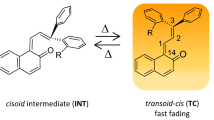
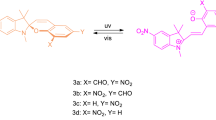
Diarylbenzopyrans and their annulated derivatives are a class of photochromic compounds, which have been extensively investigated for photochromism. In fact, some naphthopyrans are employed industrially in ophthalmic lens applications. Photoirradiation of 2,2-diphenylbenzopyran leads to the formation of colored o-quinonoid intermediates. The latter revert too rapidly to allow their observation only at low temperatures (173–263 K). Annulation and other strategies are exploited to observe the colored o-quinonoid intermediates that persist for a few minutes at room temperature. We have examined photochromism of a set of silyloxy-substituted 2,2-diphenylbenzo- and naphthopyrans to explore how the mesomeric effects transmitted by the strongly e-donating silyloxy group substituted in the ring and at phenyl groups manifest in the spectrokinetic properties of their photogenerated colored o-quinonoid intermediates. We show that silyloxy substitution in the benzopyran ring leads to remarkable stabilization of the colored intermediates to enable their persistence at 283 K for a few minutes. In contrast, similar substitution in the C2-phenyl rings destabilizes the colored intermediates.
Graphical Abstract:






Similar content being viewed by others
References
Li Q 2013 Intelligent Stimuli-Responsive Materials: From Well-Defined Nanostructures to Applications (Hoboken: Wiley)
Theato P, Sumerlin B S, O’Reilly R K and Epps III T H 2013 Stimuli-responsive materials Chem. Soc. Rev. 42 7055
Dürr H and Bouas-Laurent H 2003 Photochromism: Molecules and Systems (Amsterdam: Elsevier)
Crano J C and Guglielmetti R J 2002 Organic photochromic and thermochromic compounds (New York: Kluwer) Vol. 1 p.111
Irie M 2000 Photochromism: Memories and Switches Chem. Rev. 100 1683
Bouas-Laurent H and Dürr H 2001 Organic photochromism (IUPAC technical report) Pure Appl. Chem. 73 639
Minkin V I 2004 Photo-, thermo-, solvato-, and electrochromic spiroheterocyclic compounds Chem. Rev. 104 2751
Delbaere S, Vermeersch G and Micheau J C 2011 Quantitative analysis of the dynamic behaviour of photochromic systems J. Photochem. Photobiol. C 12 74
Klajn R 2014 Spiropyran-based dynamic materials Chem. Soc. Rev. 43 148
Becker R S and Michl J 1966 Photochromism of synthetic and naturally occurring 2H-chromenes and 2H-pyrans J. Am. Chem. Soc. 88 5931
Lenoble C and Becker R S 1986 Photophysics, photochemistry and kinetics of photochromic \(2H\)-pyrans and chromenes J. Photochem. 33 187
Becker R S and Favaro G 2011 New concepts in photochemistry and photophysics: Photochromic and other type molecules J. Photochem. Photobiol. C 12 167
Crano J C, Flood T, Knowles D, Kumar A and Gemert B V 1996 Photochromic compounds: Chemistry and application in ophthalmic lenses Pure Appl. Chem. 68 1395
Corns S N, Partington S M and Towns A D 2009 Industrial organic photochromic dyes Color. Technol. 125 249
Pozzo J-L, Lokshin V, Samat A, Guglielmetti R, Dubest R and Aubard J 1998 Effect of heteroaromatic annulation with five-membered rings on the photochromism of 2H-[1]-benzopyrans J. Photochem. Photobiol. A 114 185
Moustrou C, Rebière N, Samat A, Guglielmetti R, Yassar A E, Dubest R and Aubard J 1998 Synthesis of thiophene-substituted 3\(H\)-naphtho[2,1-\(b\)]pyrans, precursors of photomodulated materials Helv. Chim. Acta 81 1293
Salvador M A, Coelho P J, Burrows H D, Oliveira M M and Carvalho L M 2004 Studies under continuous irradiation of photochromic spiro[fluorenopyran-thioxanthenes] Helv. Chim. Acta 87 1400
Sousa C M, Pina J, de Melo J S, Berthet J, Delbaere S and Coelho P J 2011 Preventing the formation of the long-lived colored transoid-trans photoisomer in photochromic benzopyrans Org. Lett. 13 4040
Frigoli M, Maurel F, Berthet J, Delbaere S, Marrot J and Oliveira M M 2012 The control of photochromism of [\(3H\)]-naphthopyran derivatives with intramolecular \(\text{ CH- }\pi \) bonds Org. Lett. 14 4150
Sousa C M, Berthet J, Delbaere S, Polónia A and Coelho P J 2015 Fast color change with photochromic fused naphthopyrans J. Org. Chem. 80 12177
Moorthy J N, Venkatakrishnan P, Sengupta S and Baidya M 2006 Facile synthesis, fluorescence, and photochromism of novel helical pyrones and chromenes Org. Lett. 8 4891
Moorthy J N, Venkatakrishnan P, Samanta S and Kumar D K 2007 Photochromism of arylchromenes: remarkable modification of absorption properties and lifetimes of o-quinonoid intermediates Org. Lett. 9 919
Moorthy J N, Koner A L, Samanta S, Roy A and Nau W M 2009 Modulation of spectrokinetic properties of \(o\)-quinonoid reactive intermediates by electronic factors: Time-resolved laser flash and steady-state photolysis investigations of photochromic 6- and 7-arylchromenes Chem. Eur. J. 15 4289
Mukhopadhyay A, Maka V K and Moorthy J N 2016 Remarkable influence of phenyl/arylethynylation on the photo-chromism of 2,2-diphenylbenzo-pyrans (chromenes) Eur. J. Org. Chem. 2016 274
Mandal S, Parida K N, Samanta S and Moorthy J N 2011 Influence of (2,3,4,5,6-pentamethyl/phenyl)phenyl scaffold: Stereoelectronic control of the persistence of \(o\)-quinonoid reactive intermediates of photochromic chromenes J. Org. Chem. 76 7406
Moorthy J N, Mandal S and Parida K N 2012 Through-space control of the persistence of photogenerated \(o\)-quinonoid intermediates in naphthalenes containing cofacially oriented chromenes and arenes Org. Lett. 14 2438
Moorthy J N, Mandal S, Mukhopadhyay A and Samanta S 2013 Helicity as a steric force: Stabilization and helicity-dependent reversion of colored \(o\)-quinonoid intermediates of helical chromenes J. Am. Chem. Soc. 135 6872
Mukhopadhyay A and Moorthy J N 2016 Phenomenon to functions: Photochromism of diarylpyrans, spectrokinetic properties and functional materials J. Photochem. Photobiol. C 29 73
Mukhopadhyay A, Maka V K, Savitha G and Moorthy J N 2018 Photochromic 2D metal-organic framework nanosheets (MONs): Design, synthesis, and functional MON-ormosil composite Chem 4 1059
Mukhopadhyay A, Maka V K and Moorthy J N 2016 Fluoride-triggered ring-opening of photochromic diarylpyrans into merocyanine dyes: Naked-eye sensing in subppm levels J. Org. Chem. 81 7741
Li L, Shen X, Xu Q-H and Yao S Q 2013 A switchable two-photon membrane tracer capable of imaging membrane-associated protein tyrosine phosphatase activities Angew. Chem. Int. Ed. 52 424
Ottavi G, Favaro G and Matatesta V 1998 Spectrokinetic study of 2,2-diphenyl-5,6-benzo(2\(H\)) chromene: A thermoreversible and photoreversible photochromic system J. Photochem. Photobiol. A 115 123
Gorner H and Chibisov A K 2002 Photoprocesses in 2,2-diphenyl-5,6-benzo(2\(H\))chromene J. Photochem. Photobiol. A 149 83
Hobley J, Malatesta V, Hatanaka K, Kajimoto S, Williams S L and Fukumura H 2002 Picosecond and nanosecond photo-dynamics of a naphthopyran merocyanine Phys. Chem. Chem. Phys. 4 180
Evans R A, Hanley T L, Skidmore M A, Davis T P, Such G K, Yee L H, Ball G E and Lewis D A 2005 The generic enhancement of photochromic dye switching speeds in a rigid polymer matrix Nat. Mater. 4 249
Malic N, Campbell J A and Evans R A 2008 Superior photochromic performance of naphthopyrans in a rigid host matrix using polymer conjugation: Fast, dark, and tunable Fast, dark, and tunable Macromolecules 41 1206
Acknowledgements
JNM is thankful to the Science and Engineering Research Board (SERB, Grant Number: SB/S2/JCB-52/2014), New Delhi, for financial support through the J. C. Bose fellowship. AM and VKM sincerely acknowledge the Council of Scientific and Industrial Research (CSIR), New Delhi for senior research fellowships. Mr. Debanjan Bhattacharjee is thanked for his contribution during the initial stages of the project.
Author information
Authors and Affiliations
Corresponding author
Additional information
\(^{\S }\)In honor of Prof. M. V. George on the occasion of his 90\(^\mathrm{th}\) Birthday.
Special Issue on Photochemistry, Photophysics and Photobiology
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mukhopadhyay, A., Maka, V.K. & Moorthy, J.N. Influence of silyloxy substitution on the photochromic properties of diarylbenzo- and naphthopyrans\(^{\S }\). J Chem Sci 130, 140 (2018). https://doi.org/10.1007/s12039-018-1542-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-018-1542-0