Abstract
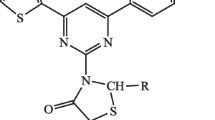
Different substituted diesters of thiazolopyrimidine were prepared by the treatment of 3,4 dihydropyrimidine2-thione with α-haloesters using ethanol under reflux condition affording 71–85% yield. IR, 1HNMR, 13CNMR and elemental analyses were used for the characterization of these compounds. The crystal and molecular structure of one of the product, 5-phenyl-3,7-dimethyl-5H-thiazolo[3,2-a]pyrimidine-2,6-dicarboxylic acid diethyl ester (3e) was verified by single crystal X-ray diffraction method. The antimicrobial activity was evaluated against four bacterial strains and one fungal species. Few of the derivatives exhibited antibacterial and antifungal activities.

We have reported the synthesis of some thiazolopyrimidine derivatives by cyclo-condensation of dihydropyrimidine with halo esters. Crystal structure analysis of one of the derivatives has been carried out. Some of the derivatives exhibited antibacterial and antifungal activities.
Similar content being viewed by others
References
Ozair A, Suroor A K, Nadeem S and Waquar A 2010 Med. Chem. Res. 19 1245
Kappe C O, Fabian W M F and Semones M A 1997 Tetrahedron 53 2803
Atwal K S, Swanson B N, Unger S E, Floyd D M, Moreland S, Hedberg A and O’Reilly B C 1991 J. Med. Chem. 34 806
Cho H, Ueda M, Shima K, Mizuno A, Hayashimatsu M, Ohnaka Y, Takeuchi Y, Hamaguchi M, Aisaka K, Hidaka T, Kawai M, Takeda M, Ishihara T, Funahashi K, Satah F, Morita M and Noguchi T 1989 J. Med. Chem. 32 2399
Atwal K, Rovnyak G C, Schwartz J, Moreland S, Hedberg A, Gougoutas J Z, Malley M F and Floyd D M 1990 J. Med. Chem. 33 1510
Atwal K, Rovnyak G C, Kimball S D, Floyd D M, Moreland S, Swanson S, Gougoutas J Z, Schwartz J, Smillie K M and Malley M F 1990 J. Med. Chem. 33 2629
Atwal K S, Swanson B N, Unger S E, Floyd D M, Moreland S, Hedberg A and O’Reilly B C 1991 J. Med. Chem. 34 806
Grover G J, Dzwonczyk S, McMullen D M, Normadinam C S and Sleph P G and Moreland S 1995 J. Cardiovasc. Pharmacol. 26 289
Rovnyak G C, Atwal K S, Hedberg A, Kimball S D, Moreland S, Gougoutas J Z and O’Reilly B C 1992 J. Med. Chem. 35 3254
Rovnyak G C, Kimball S D, Beyer B, Cucinotta G, DiMarco J D, Gougoutas J Z, Hedberg A, Malley M, McCarthy J P, Zhang R and Moreland S 1995 J. Med. Chem. 38 119
Patil A D, Kumar N V, Kokke W C, Bean M F, Freyer A J, Debrossin C, Mai S, Truneh A, Faulkner P, Johnson R K, Westley J W and Potts B C M 1995 J. Org. Chem. 60 1182
Rajeshwar Rao V and Ravinder Reddy V 2006 Phosphorous, Sulphur Silicon 181 147
Singh S, Schober A, Michael G and Alexander G G 2011 Tetrahedron 52 3814
Kulakov I V, Nurkenov O A, Turdybekov D M, Issabaeva G M, Mahmutova A S and Turdybekov K M 2009 Chem. Heterocycl. Comp. 45 856
Ozair A, Suroor A K, Nadeem S, Waquar A, Suraj P V and Sadaf J G 2010 Eur. J. Med. Chem. 45 5113
Sayed H H, Morsy E M H and Kotb E R 2010 Syn. Com. 40 2712
Salwa F M, Eman M F, Abd El-Galil E A and Abd El-Shafy D N 2010 Eur. J. Med. Chem. 45 1494
Hui Z, Lan-mei C, Lin-lin Z, Si-jie L, David C C W, Huang-quan L and Chun H 2008 ARKIVOC. 8 266
Bruker 1998 SMART, SAINT-Plus and SADABS. Bruker AXS Inc, Madison
Sheldrick G M 2008 Acta Cryst. A64 112 Wisconsin USA
Farrugia L J and ORTEP-3 1999 J. Appl. Cryst. 32 837
Nardelli M 1983 Acta Cryst. C39 1141
Rawlinson L B, Ryan S M, Mantovani G, Syrett J A, Haddleton D M and Brayden D J 2010 Biomacromolecules 11 443
Delignette-Muller M L and Flandrois J P 1994 J. Antimicrob. Chemother. 34 73
Kappe C O 1998 Molecules 3 1
Nagarajaiah H and Begum N S 2011 Acta Cryst. E67 o3444
Mukesh M J, Bharat B B and Jerry P 2010 Acta Cryst. E66 o599
Cremer D and Pople J A 1975 J. Am. Chem. Soc. 97 1354
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
NAGARAJAIAH, H., KHAZI, I.M. & BEGUM, N.S. Synthesis, characterization and biological evaluation of thiazolopyrimidine derivatives. J Chem Sci 124, 847–855 (2012). https://doi.org/10.1007/s12039-012-0271-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-012-0271-z