Abstract
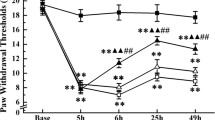
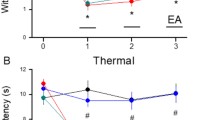
Inflammatory pain is difficult to treat clinically, but electroacupuncture (EA) has been demonstrated to be effective in alleviating inflammatory pain. Programmed cell death ligand-1 (PD-L1) and its downstream signal, Src homology region two domain-containing phosphatase-1 (SHP-1) have a critical role in relieving inflammatory pain. However, whether the PD-L1/PD-1-SHP-1 pathway mediates the analgesic and anti-inflammatory effects of EA in inflammatory pain remains unclear. Here, we observed that EA reversed the complete Freund’s adjuvant (CFA)-induced hyperalgesia. EA reduced the expression of IL-6, iNOS, and NF-κB pathway in dorsal root ganglia (DRG) on day 7 after CFA injection but had no effect on the expression of IL-6, iNOS, and NF-κB PP65 on day 21 after CFA injection. Moreover, EA upregulated the protein levels of the PD-L1/PD-1-SHP-1 pathway on day 7 and day 21 after CFA injection. Furthermore, EA upregulated PD-L1 expression in calcitonin gene-related peptide (CGRP)+ but not in isohaemagglutinin B4 (IB4)+ and NF200+ neurons on day 7 and day 21 after CFA injection. Intrathecal injection of the PD-L1/PD-1 inhibitor BMS-1 (50 or 100 µg) blocked the EA-induced analgesic effect, significantly increased IL-6 and iNOS levels, and reduced the levels of PD-L1/PD-1-SHP-1. BMS-1 (50 or 100 µg) significantly reduced the expression of PD-L1 in IB4+, CGRP+, and NF200+ neurons. Our results show that EA’s anti-inflammatory and analgesic effects are associated with activating the PD-L1/PD-1-SHP-1 pathway and suppressing its regulated neuroinflammation. This study provides a new potential therapeutic target for treating inflammatory pain.









Similar content being viewed by others
Data Availability
All the data supporting the findings of this study are available within the article and from the corresponding author upon reasonable request.
Abbreviations
- EA:
-
Electroacupuncture
- PD-L1:
-
Programmed death ligand-1
- PD-1:
-
Programmed cell death receptor
- SHP-1:
-
Src homology region two domain-containing phosphatase-1
- DRG:
-
Dorsal root ganglia
- Sting:
-
Stimulator of interferon
- NGF:
-
Nerve growth factor
- IL-2:
-
Interleukin-2
- IFN-γ:
-
Interferon-γ
- PWT:
-
Paw withdraw threshold
- PWL:
-
Paw withdraw latency
- CGRP:
-
Calcitonin gene-related peptide
- IB4:
-
IsohemagglutininB4
- NF-κB:
-
Nuclear transcription factor kappa B
- TNF-α:
-
Tumour necrosis factor-α
- PBS:
-
Phosphate buffer saline
- IL-1β:
-
Interleukin-1β
- IL-6:
-
Interleukin-6
- 5-HT (serotonin):
-
5-Hydroxytryptamine
- iNOS:
-
Inducible nitric oxide synthase
- NSAIDs:
-
Nonsteroidal anti-inflammatory drugs
References:
Woolf CJ (2010) What is this thing called pain? J Clin Invest 120:3742–3744
Basbaum AI, Bautista DM, Scherrer G, Julius D (2009) Cellular and molecular mechanisms of pain. Cell 139:267–284
Hucho T, Levine JD (2007) Signaling pathways in sensitization: toward a nociceptor cell biology. Neuron 55:365–376
Pinho-Ribeiro FA, Verri WJ, Chiu IM (2017) Nociceptor sensory neuron-immune interactions in pain and inflammation. Trends Immunol 38:5–19
Held M, Karl F, Vlckova E, Rajdova A, Escolano-Lozano F, Stetter C, Bharti R, Forstner KU, Leinders M, Dusek L, Birklein F, Bednarik J, Sommer C, Uceyler N (2019) Sensory profiles and immune-related expression patterns of patients with and without neuropathic pain after peripheral nerve lesion. Pain 160:2316–2327
Chen G, Kim YH, Li H, Luo H, Liu DL, Zhang ZJ, Lay M, Chang W, Zhang YQ, Ji RR (2017) PD-L1 inhibits acute and chronic pain by suppressing nociceptive neuron activity via PD-1. Nat Neurosci 20:917–926
Ji RR, Samad TA, Jin SX, Schmoll R, Woolf CJ (2002) p38 MAPK activation by NGF in primary sensory neurons after inflammation increases TRPV1 levels and maintains heat hyperalgesia. Neuron 36:57–68
Yue JX, Wang RR, Yu J, Tang YY, Hou WW, Lou GD, Zhang SH, Chen Z (2014) Histamine upregulates Nav1.8 expression in primary afferent neurons via H2 receptors: involvement in neuropathic pain. CNS Neurosci Ther 20:883–92
Sharma P, Allison JP (2015) The future of immune checkpoint therapy. Science 348:56–61
Keir ME, Butte MJ, Freeman GJ, Sharpe AH (2008) PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol 26:677–704
Hebeisen M, Baitsch L, Presotto D, Baumgaertner P, Romero P, Michielin O, Speiser DE, Rufer N (2013) SHP-1 phosphatase activity counteracts increased T cell receptor affinity. J Clin Invest 123:1044–1056
Uceyler N, Gobel K, Meuth SG, Ortler S, Stoll G, Sommer C, Wiendl H, Kleinschnitz C (2010) Deficiency of the negative immune regulator B7–H1 enhances inflammation and neuropathic pain after chronic constriction injury of mouse sciatic nerve. Exp Neurol 222:153–160
Wang K, Gu Y, Liao Y, Bang S, Donnelly CR, Chen O, Tao X, Mirando AJ, Hilton MJ, Ji RR (2020) PD-1 blockade inhibits osteoclast formation and murine bone cancer pain. J Clin Invest 130:3603–3620
Shi S, Han Y, Wang D, Guo P, Wang J, Ren T, Wang W (2020) PD-L1 and PD-1 expressed in trigeminal ganglia may inhibit pain in an acute migraine model. Cephalalgia 40:288–298
Liu BL, Cao QL, Zhao X, Liu HZ, Zhang YQ (2020) Inhibition of trpv1 by shp-1 in nociceptive primary sensory neurons is critical in pd-l1 analgesia. JCI Insight 5(20)
Xiao X, Zhao XT, Xu LC, Yue LP, Liu FY, Cai J, Liao FF, Kong JG, Xing GG, Yi M, Wan Y (2015) Shp-1 dephosphorylates TRPV1 in dorsal root ganglion neurons and alleviates CFA-induced inflammatory pain in rats. Pain 156:597–608
Fang JQ, Du JY, Liang Y, Fang JF (2013) Intervention of electroacupuncture on spinal p38 MAPK/ATF-2/VR-1 pathway in treating inflammatory pain induced by CFA in rats. Mol Pain 9:13
Gu Y, Chen S, Mo Y, Tu Y, Chen N, Zhao X, Li S, Yu Q, Dai Q, Wang J (2020) Electroacupuncture attenuates CFA-induced inflammatory pain by regulating CaMKII. Neural Plast 2020:8861994
Xiang X, Wang S, Shao F, Fang J, Xu Y, Wang W et al (2019) Electroacupuncture stimulation alleviates CFA-induced inflammatory pain via suppressing P2X3 expression. Int J Mol Sci 20(13)
Chen L, Xu A, Yin N, Zhao M, Wang Z, Chen T, Gao Y, Chen Z (2017) Enhancement of immune cytokines and splenic CD4+ T cells by electroacupuncture at ST36 acupoint of SD rats. PLoS ONE 12:e175568
Su TF, Zhao YQ, Zhang LH, Peng M, Wu CH, Pei L, Tian B, Zhang J, Shi J, Pan HL, Li M (2012) Electroacupuncture reduces the expression of proinflammatory cytokines in inflamed skin tissues through activation of cannabinoid CB2 receptors. Eur J Pain 16:624–635
Yu ML, Wei RD, Zhang T, Wang JM, Cheng Y, Qin FF, Fu SP, Lu ZG, Lu SF (2020) Electroacupuncture relieves pain and attenuates inflammation progression through inducing IL-10 production in CFA-induced mice. Inflammation 43:1233–1245
Zimmermann M (1983) Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16:109–110
Ghasemlou N, Chiu IM, Julien JP, Woolf CJ (2015) CD11b+Ly6G- myeloid cells mediate mechanical inflammatory pain hypersensitivity. Proc Natl Acad Sci U S A 112:E6808–E6817
Li QH, Xie WX, Li XP, Huang KT, Du ZH, Cong WJ, Zhou LH, Ye TS, Chen JF (2015) Adenosine A2A receptors mediate anti-inflammatory effects of electroacupuncture on synovitis in mice with collagen-induced arthritis. Evid Based Complement Alternat Med 2015:809560
Ye TS, Du ZH, Li ZH, Xie WX, Huang KT, Chen Y, Chen ZY, Hu H, Wang JL, Fang JQ (2016) Repeated electroacupuncture persistently elevates adenosine and ameliorates collagen-induced arthritis in rats. Evid Based Complement Alternat Med 2016:3632168
Lao L, Zhang RX, Zhang G, Wang X, Berman BM, Ren K (2004) A parametric study of electroacupuncture on persistent hyperalgesia and Fos protein expression in rats. Brain Res 1020:18–29
Zhang RX, Lao L, Wang X, Fan A, Wang L, Ren K, Berman BM (2005) Electroacupuncture attenuates inflammation in a rat model. J Altern Complement Med 11:135–142
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53:55–63
Xiang HC, Lin LX, Hu XF, Zhu H, Li HP, Zhang RY, Hu L, Liu WT, Zhao YL, Shu Y, Pan HL, Li M (2019) AMPK activation attenuates inflammatory pain through inhibiting NF-kappaB activation and IL-1beta expression. J Neuroinflammation 16:34
Chen Y, Liu Y, Wang Y, Chen X, Wang C, Chen X, Yuan X, Liu L, Yang J, Zhou X (2022) Prevotellaceae produces butyrate to alleviate PD-1/PD-L1 inhibitor-related cardiotoxicity via PPARalpha-CYP4X1 axis in colonic macrophages. J Exp Clin Cancer Res 41:1
Baral P, Udit S, Chiu IM (2019) Pain and immunity: implications for host defence. Nat Rev Immunol 19:433–447
Marret E, Kurdi O, Zufferey P, Bonnet F (2005) Effects of nonsteroidal antiinflammatory drugs on patient-controlled analgesia morphine side effects: meta-analysis of randomized controlled trials. Anesthesiology 102:1249–1260
Kim JH, Min BI, Schmidt D, Lee HJ, Park DS (2000) The difference between electroacupuncture only and electroacupuncture with manipulation on analgesia in rats. Neurosci Lett 279:149–152
Li A, Wang Y, Xin J, Lao L, Ren K, Berman BM, Zhang RX (2007) Electroacupuncture suppresses hyperalgesia and spinal Fos expression by activating the descending inhibitory system. Brain Res 1186:171–179
Liu XJ, Gingrich JR, Vargas-Caballero M, Dong YN, Sengar A, Beggs S, Wang SH, Ding HK, Frankland PW, Salter MW (2008) Treatment of inflammatory and neuropathic pain by uncoupling Src from the NMDA receptor complex. Nat Med 14:1325–1332
Pomeranz B, Stux G, Han C (1989) Scientific bases of acupuncture. Springer-Verlag, Berlin
Wan C, Xu Y, Cen B, Xia Y, Yao L, Zheng Y, Zhao J, He S, Chen Y (2021) Neuregulin1-ErbB4 signaling in spinal cord participates in electroacupuncture analgesia in inflammatory pain. Front Neurosci 15:636348
Kythreotou A, Siddique A, Mauri FA, Bower M, Pinato DJ (2018) PD-L1. J Clin Pathol 71:189–194
Sim YB, Park SH, Kang YJ, Jung JS, Ryu OH, Choi MG, Choi SS, Suh HW (2013) Interleukin-1beta (IL-1beta) increases pain behavior and the blood glucose level: possible involvement of glucocorticoid system. Cytokine 64:351–356
Luchting B, Heyn J, Woehrle T, Rachinger-Adam B, Kreth S, Hinske LC, Azad SC (2016) Differential expression of P2X7 receptor and IL-1beta in nociceptive and neuropathic pain. J Neuroinflammation 13:100
He H, Zhou Y, Zhou Y, Zhuang J, He X, Wang S, Lin W (2018) Dexmedetomidine mitigates microglia-mediated neuroinflammation through upregulation of programmed cell death protein 1 in a rat spinal cord injury model. J Neurotrauma 35:2591–2603
Wu J, Sun L, Li H, Shen H, Zhai W, Yu Z, Chen G (2017) Roles of programmed death protein 1/programmed death-ligand 1 in secondary brain injury after intracerebral hemorrhage in rats: selective modulation of microglia polarization to anti-inflammatory phenotype. J Neuroinflammation 14:36
Yao A, Liu F, Chen K, Tang L, Liu L, Zhang K, Yu C, Bian G, Guo H, Zheng J, Cheng P, Ju G, Wang J (2014) Programmed death 1 deficiency induces the polarization of macrophages/microglia to the M1 phenotype after spinal cord injury in mice. Neurotherapeutics 11:636–650
An H, Hou J, Zhou J, Zhao W, Xu H, Zheng Y, Yu Y, Liu S, Cao X (2008) Phosphatase SHP-1 promotes TLR- and RIG-I-activated production of type I interferon by inhibiting the kinase IRAK1. Nat Immunol 9:542–550
Adhikari A, Martel C, Marette A, Olivier M (2017) Hepatocyte SHP-1 is a critical modulator of inflammation during endotoxemia. Sci Rep 7:2218
Funding
This work was supported by the National Key Research and Development Program of China (grant 2018YFC2001802 to X. Chen); the National Natural Science Foundation (grant 82071251 to X. Chen); the Hubei Province Key Research and Development Program (grant 2021BCA145 to X. Chen).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by Daling Deng and Feng Xu. The first draft of the manuscript was written by Daling Deng, and all authors commented on previous versions of the manuscript. Daling Deng and Feng Xu contribute equally to this work. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All experimental procedures were carried out in accordance with the ethical guidelines of the International Association for the Study of Pain (Zimmermann 1983) and approved by the Animal Care Committee at Huazhong University of Science and Technology.
Consent to Participate
Not applicable.
Consent for Publication
All authors have consented to publish in the journal Molecular Neurobiology.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deng, D., Xu, F., Ma, L. et al. Electroacupuncture Alleviates CFA-Induced Inflammatory Pain via PD-L1/PD-1-SHP-1 Pathway. Mol Neurobiol 60, 2922–2936 (2023). https://doi.org/10.1007/s12035-023-03233-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03233-x