Abstract
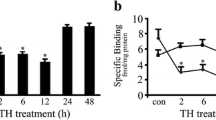
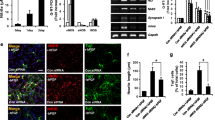
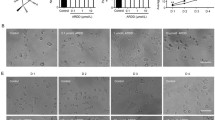
Docosahexaenoic acid (DHA), an important ω-3 fatty acid, is abundantly present in the central nervous system and is important in every step of brain development. Much of this knowledge has been based on studies of the role of DHA in the function of the neurons, and reports on its effect on the glial cells are few and far between. We have previously reported that DHA facilitates astrocyte differentiation in primary culture. We have further explored the signaling mechanism associated with this event. It was observed that a sustained activation of the extracellular signal-regulated kinase (ERK) appeared to be critical for DHA-induced differentiation of the cultured astrocytes. Prior exposure to different endocytic inhibitors blocked both ERK activation and differentiation of the astrocytes during DHA treatment suggesting that the observed induction of ERK-2 was purely endosomal. Unlike the β1-adrenergic receptor (β1-AR) antagonist, atenolol, pre-treatment of the cells with the β2-adrenergic receptor (β2-AR) antagonist, ICI-118,551 inhibited the DHA-induced differentiation process, indicating a downstream involvement of β2-AR in the differentiation process. qRT-PCR and western blot analysis demonstrated a significant induction in the mRNA and protein expression of β2-AR at 18–24 h of DHA treatment, suggesting that the induction of β2-AR may be due to transcriptional upregulation. Moreover, DHA caused activation of PKA at 6 h, followed by activation of downstream cAMP response element-binding protein, a known transcription factor for β2-AR. Altogether, the observations suggest that DHA upregulates β2-AR in astrocytes, which undergo endocytosis and signals for sustained endosomal ERK activation to drive the differentiation process.









Similar content being viewed by others
References
Horrocks LA, Farooqui AA (2004) Docosahexaenoic acid in the diet: its importance in maintenance and restoration of neural membrane function. Prostaglandins Leukotrienes Essent. Fatty Acids 70:361–372
Innis SM (2007) Dietary (n-3) fatty acids and brain development. J Nutr 137:855–859
Tassoni D, Kaur G, Weisinger RS, Sinclair AJ (2008) The role of eicosanoids in the brain. Asia Pac J Clin Nutr 17:220–228
Luchtman DW, Song C (2013) Cognitive enhancement by omega-3 fatty acids from child-hood to old age: findings from animal and clinical studies. Neuropharmacology 64:550–565
Parletta N, Milte CM, Meyer BJ (2013) Nutritional modulation of cognitive function and mental health. J Nutr Biochem 24:725–743
de Urquiza AM, Liu S, Sjoberg M, Zetterstrom RH, Griffiths W, Sjovall J, Perlmann T (2000) Docosahexaenoic acid, a ligand for the retinoid X receptor in mouse brain. Science 290:2140–2144
Miyauchi S, Hirasawa A, Iga T, Liu N, Itsubo C, Sadakane K, Hara T, Tsujimoto G (2009) Distribution and regulation of protein expression of the free fatty acid receptor GPR120. Naunyn Schmiedeberg’s Arch Pharmacol 379:427–434
Oh DY, Talukdar S, Bae EJ, Imamura T, Morinaga H, Fan W, Li P, Lu WJ et al (2010) GPR120 is anomega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 142:687–698
Ma D, Zhang M, Larsen CP, Xu F, Hua W, Yamashima T, Mao Y, Zhou L (2010) DHA promotes the neuronal differentiation of rat neural stem cells transfected with GPR40. Gene 1330:1–8
Yamashima T (2015) ‘PUFA-GPR40-CREB signaling’ hypothesis for the adult primate neurogenesis. Prog Lipid Res 51:221–231
Wellhauser L, Belsham DD (2014) Activation of the omega-3 fatty acid receptor GPR120 mediates anti-inflammatory actions in immortalized hypothalamic neurons. J Neuroinflammation 11:60–73
Gharami K, Das M, Das S (2015) Essential role of docosahexaenoic acid towards development of a smarter brain. Neurochem Int 89:51–62
Moore SA, Yoder E, Murphy S, Dutton GR, Spector AA (1991) Astrocytes, not neurons, produce docosahexaenoic acid (22:6-3) and arachidonic acid (20:4-6). J Neurochem 56:518–524
Moore SA (1994) Local synthesis and targeting of essential fatty acids at the cellular interface between blood and brain: a role for cerebral endothelium and astrocytes in the accretion of CNS docosahexaenoic acid. World Rev Nutr Diet 75:128–133
Bernoud N, Fenart L, Benistant C et al (1998) Astrocytes are mainly responsible for the polyunsaturated fatty acid enrichment in blood-brain barrier endothelial cells in vitro. J Lipid Res 39:1816–1824
Moore SA, Yoder E, Spector AA (1990) Role of the blood-brain barrier in the formation of long-chain omega-3 and omega-6 fatty acids from essential fatty acid precursors. J Neurochem 55:391–402
Moore SA (2001) Polyunsaturated fatty acid synthesis and release by brain-derived cells in vitro. J Mol Neurosci 16:195–200
Garcia MC, Kim HY (1997) Mobilization of arachidonate and docosahexaenoate by stimulation of the 5-HT2A receptor in rat C6 glioma cells. Brain Res 768:43–48
Kim HY, Edsall L, Garcia M, Zhang H (1999) The release of polyunsaturated fatty acids and their lipoxygenation in the brain. Adv Exp Med Biol 447:75–85
Strokin M, Sergeeva M, Reiser G (2003) Docosahexaenoic acid and arachidonic acid release in rat brain astrocytes is mediated by two separate isoforms of phospholipase A2 and is differently regulated by cyclic AMP and Ca2+. Br J Pharmacol 139:1014–1022
Calderon F, Kim HY (2004) Docosahexaenoic acid promotes neurite growth in hippocampal neurons. J Neurochem 90:979–988
Cao D, Xue R, Xu J, Liu Z (2005) Effects of docosahexaenoic acid on the survival and neurite outgrowth of rat cortical neurons in primary cultures. J Nutr Biochem 16:538–546
Cao D, Kevala K, Kim J, Moon HS, Jun SB, Lovinger D, Kim HY (2009) Docosahexaenoic acid promotes hippocampal neuronal development and synaptic function. J Neurochem 111:510–521
Champeil-Potokar G, Chaumontet C, Guesnet P, Lavialle M, Denis I (2006) Docosahexaenoic acid (22:6n-3) enrichment of membrane phospholipids increases gap junction coupling capacity in cultured astrocytes. Eur J Neurosci 24:3084–3090
Grintal B, Champeil-Potokar G, Lavialle M, Vancassel S, Breton S, Denis I (2009) Inhibition of astroglial glutamate transport by polyunsaturated fatty acids: evidence for a signaling role of docosahexaenoic acid. Neurochem Int 54:535–543
Ximenes da Silva A, Lavialle F, Gendrot G, Guesnet P, Alessandri JM, Lavialle M (2002) Glucose transport and utilization are altered in the brain of rats deficient in n-3 polyunsaturated fatty acids. J Neurochem 81:1328–1337
Pifferi F, Roux F, Langelier B et al (2005) (n-3) polyunsaturated fatty acid deficiency reduces the expression of both isoforms of the brain glucose transporter GLUT1 in rats. J Nutr 135:2241–2246
Champeil-Potokar G, Hennebelle M, Latour A, Vancassel S, Denis I (2016) Docosahexaenoic acid (DHA) prevents corticosterone-induced changes in astrocytes morphology and function. J Neurochem 136:1155–1167
Joardar A, Sen AK, Das S (2006) Docosahexaenoic acid facilitates cell maturation and beta-adrenergic transmission in astrocytes. J Lipid Res 47:571–581
Federoff S (1986) Prenatal ontogenesis of astrocytes. In: Federoff S, Vernadakis A (eds) Astrocytes: development, morphology, and regional specialization of astrocytes. Academic Press, Orlando, pp. 35–74
Lim R, Mitsunobu K, Li WKP (1973) Maturation-stimulating effect of brain extract and dibutyryl cyclic AMP on dissociated embryonic brain cells in culture. Exp Cell Res 79:243–246
Moonen G, Cam Y, Sensenbrenner M, Mandel P (1975) Variability of the effects of serum-free medium, dibutyryl cyclic AMP or theophylline on the morphology of cultured newborn astrocytcs. Cell Tissue Rex 163:365–372
Gavaret JM, Delbauffe DT, Chalaye DB, Pomerance M, Pierce M (1991) Thyroid hormone action: induction of morphological changes and protein secretion in astroglial cultures. Dev Brain Res 58:43–49
Paul S, Das S, Poddar R, Sarkar PK (1996) Effect of thyroid hormone in the morphological differentiation and maturation of astrocytes: temporal correlation with synthesis and organisation of actin. Eur J Neurosci 8:2361–2370
Rakic P (1972) Mode of cell migration to the superficial layers of fetal monkey neocortex. J Comp Neurol 145:61–81
Silver J, Lorenz SE, Wahlsten D, Coughlin J (1982) Axonal guidance during development of the great cerebral commissures: descriptive experimental studies, in vivo, on the role of preformed glial pathways. J Comp Neurol 210:10–29
Guillery RW, Walsh C (1987) Changing glial organization relates to changing fiber order in the developing optic nerve of ferrets. J Comp Neurol 265:203–217
Abe K, Saito H (2000) The p44/42 mitogen-activated protein kinase cascade is involved in the induction and maintenance of astrocyte stellation mediated by protein kinase C. Neurosci Res 36:251–257
Ueki T, Fujita M, Sato K, Asai K, Yamada K, Kato T (2001) Epidermal growth factor down-regulates connexin-43 expression in cultured rat cortical astrocytes. Neurosci Lett 313:53–56
Gharami K, Das S (2000) Thyroid hormone-induced morphological differentiation and maturation of astrocytes are mediated through the beta-adrenergic receptor. J Neurochem 75:1962–1969
Gharami K, Das S (2004) Delayed but sustained induction of mitogen-activated protein kinase activity is associated with β-adrenergic receptor-mediated morphological differentiation of astrocytes. J Neurochem 88:12–22
Grynberg A, Fournier A, Sergiel JP, Athias P (1995) Effect of docosahexaenoic acid and eicosapentaenoic acid in the phospholipids of rat heart muscle cells on adrenoceptor responsiveness and mechanism. J Mol Cell Cardiol 27:2507–2520
Ponsard B, Durot I, Delerive P, Oudot F, Cordelet C, Grynberg A, Athias P (1999) Cross-influence of membrane polyunsaturated fatty acids and hypoxia-reoxygenation on alpha- and beta-adrenergic function of rat cardiomyocytes. Lipids 34:457–466
Delerive P, Oudot F, Ponsard B, Talpin S, Sergiel JP, Cordelet C, Athias P, Grynberg A (1999) Hypoxia-reoxygenation and polyunsaturated fatty acids modulate adrenergic functions in cultured cardiomyocytes. J Mol Cell Cardiol 31:377–386
Skúladóttir GV, Schiöth HB, Gudbjarnason S (1993) Polyunsaturated fatty acids in heart muscle and alpha 1-adrenoceptor binding properties. Biochim Biophys Acta 1178:49–54
Begg DP, Puskás LG, Kitajka K, Ménesi D, Allen AM, Li D, Mathai ML, Shi JR et al (2012) Hypothalamic gene expression in ω-3 PUFA-deficient male rats before, and following, development of hypertension. Hypertens Res 35:381–387
Ghosh M, Gharami K, Paul S, Das S (2005) Thyroid hormone-induced morphological differentiation and maturation of astrocytes involves activation of protein kinase A and ERK signaling pathway. Eur J Neurosci 22:1609–1617
Rashid MA, Katakura M, Kharebava G, Kevala K, Kim HY (2013) N-docosahexaenoylethanolamine is a potent neurogenic factor for neural stem cell differentiation. J Neurochem 125:869–884
Park T, Chen H, Kevala K, Lee JW, Kim HY (2016) N-docosahexaenoylethanolamine ameliorates LPS-induced neuroinflammation via cAMP/PKA-dependent signaling. J Neuroinflammation 13:284
Angulo-Rojo C, Manning-Cela R, Aguirre A, Ortega A, López-Bayghen E (2013) Involvement of the Notch pathway in terminal astrocytic differentiation: role of PKA. ASN Neuro 5:e00130
Takahashi M, Li Y, Dillon TJ, Stork PJS (2017) Phosphorylation of Rap1 by cAMP-dependent protein kinase (PKA) creates a binding site for KSR to sustain ERK activation by cAMP. J Biol Chem 292:1449–1461
Dugan LL, Kim JS, Zhang Y, Bart RD, Sun Y, Holtzman DM, Gutmann DH (1999) Differential effects of cAMP in neurons and astrocytes. Role of B-raf. J Biol Chem 274:25842–25848
Wu J, Dent P, Jelinek T, Wolfman A, Weber MJ, Sturgill TW (1993) Inhibition of the EGF-activated MAP kinase signaling pathway by adenosine 3’,5’-monophosphate. Science 262:1065–1106
Cook SJ, McCormick F (1993) Inhibition by cAMP of Ras-dependent activation of Raf. Science 262:1069–1072
Marshall CJ (1995) Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell 80:179–185
Kao S, Jaiswal RK, Kolch W, Landreth GE (2001) Identification of the mechanisms regulating the differential activation of the mapk cascade by epidermal growth factor and nerve growth factor in PC12 cells. J Biol Chem 276:18169–18177
Sun P, Watanabe H, Takano K, Yokoyama T, Fujisawa J, Endo T (2006) Sustained activation of M-Ras induced by nerve growth factor is essential for neuronal differentiation of PC12 cells. Genes Cells 11:1097–1113
Tang G, Dong X, Huang X, Huang XJ, Liu H, Wang Y, Ye WC, Shi L (2015) A natural diarylheptanoid promotes neuronal differentiation via activating ERK and PI3K-Akt dependent pathways. Neuroscience 303:389–401
Zogovic N, Tovilovic-Kovacevic G, Misirkic-Marjanovic M, Vucicevic L, Janjetovic K, Harhaji-Trajkovic L, Trajkovic V (2015) Coordinated activation of AMP-activated protein kinase, extracellular signal-regulated kinase, and autophagy regulates phorbol myristate acetate-induced differentiation of SH-SY5Y neuroblastoma cells. J Neurochem 133:223–232
Das M, Ghosh M, Das S (2016) Thyroid hormone-induced differentiation of astrocytes is associated with transcriptional upregulation of β-arrestin-1 and β-adrenergic receptor-mediated endosomal signaling. Mol Neurobiol 53:5178–5190
DeFea KA, Zalevsky J, Thoma MS, Déry O, Mullins RD, Bunnett NW (2000) β-Arrestin–dependent endocytosis of proteinase-activated receptor 2 is required for intracellular targeting of activated ERK1/2. J. Cell Biol 148:1267–1281
Murphy JE, Padilla BE, Hasdemir B, Cottrell GS, Bunnett NW (2009) Endosomes: a legitimate platform for the signaling train. Proc Natl Acad Sci U S A 106:17615–17622
Luttrell LM, Gesty-Palmer D (2010) Beyond desensitization: physiological relevance of arrestin-dependent signaling. Pharmacol Rev 62:305–330
Ghosh M, Das S (2007) Increased β2-adrenergic receptor activity by thyroid hormone possibly leads to differentiation and maturation of astrocytes in culture. Cell Mol Neurobiol 27:1007–1021
Deb I, Das S (2011) Thyroid hormones protect astrocytes from morphine-induced apoptosis by regulating nitric oxide and pERK 1/2 pathways. Neurochem Int 58:861–871
Samuels NH, Stanley F, Casanova Z (1979) Depletion of L-3,5,3′- triiodothyronine and L-thyroxine in euthyroid calf serum for use in cell culture studies of the action of thyroid hormone. Endocrinol 105:80–85
Das M, Das S (2016) Identification of cytotoxic mediators and their putative role in the signaling pathways during docosahexaenoic acid (DHA)-induced apoptosis of cancer cells. Apoptosis 21:1408–1421
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin-phenol reagent. J Biol Chem 193:265–275
Impey S, Obrietan K, Wong ST, Poser S, Yano S, Wayman G, Deloulme JC, Chan G et al (1998) Cross talk between ERK and PKA is required for Ca2+ stimulation of CREB-dependent transcription and ERK nuclear translocation. Neuron 21:869–883
Wu C, Lai CF, Mobley WC (2001) Nerve growth factor activates persistent Rap1 signaling in endosomes. J Neurosci 21:5406–5416
Er EE, Mendoza MC, Mackey AM, Rameh LE, Blenis J (2013) AKT facilitates EGFR trafficking and degradation by phosphorylating and activating PIKfyve. Sci Signal 6:ra45
Zastrow MV (2002) Regulation of G protein coupled receptors by phosphorylation and endocytosis in Neuropsychopharmacology: the fifth generation of progress. Edited by Davis K. L., Charney D., Coyle J. T. And Nemeroff C. 59-70. ACNP publications.
Drake MT, Shenoy SK, Lefkowitz RJ (2006) Trafficking of G protein-coupled receptors. Circ Res 99:570–582
Hanyaloglu AC, von Zastrow M (2008) Regulation of GPCRs by endocytic membrane trafficking and its potential implications. Annu Rev Pharmacol Toxicol 48:537–568
Ferguson SSG, Barak LS, Zhang J, Caron MG (1996) G-protein-coupled receptor regulation: role of G-protein-coupled receptor kinases and arrestins. Can J Physiol Pharmacol 74:1095–1110
Collins S, Altschmied J, Herbsman O, Caron MG, Mellon PL, Lefkowitz RJ (1990) A cAMP response element in the beta 2-adrenergic receptor gene confers transcriptional autoregulation by cAMP. J Biol Chem 265:19330–19335
Wassall SR, Stillwell W (2009) Polyunsaturated fatty acid-cholesterol interactions: domain formation in membranes. Biochim Biophys Acta 1788:24–32
Shaikh SR, Teague H (2012) N-3 fatty acids and membrane microdomains: from model membranes to lymphocyte function. Prostaglandins Leukot Essent Fatty Acids 87:205–208
Litman BJ, Niu S, Polozova A, Mitchell DC (2001) The role of docosahexaenoic acid containing phospholipids in modulating G protein-coupled signaling pathways visual transduction. J Mol Neurosci 16:237–242
Guixà-González R, Javanainen M, Gómez-Soler M et al (2016) Membrane omega-3 fatty acids modulate the oligomerisation kinetics of adenosine A2A and dopamine D2 receptors. Sci Rep 22:19839
Tuller ER, Beavers CT, Lou JR, Ihnat MA, Benbrook DM, Ding WQ (2009) Docosahexaenoic acid inhibits superoxide dismutase 1 gene transcription in human cancer cells: the involvement of peroxisome proliferator-activated receptor alpha and hypoxia-inducible factor-2alpha signaling. Mol Pharmacol 76:588–595
Machová E, Nováková J, Lisá V, Dolezal V (2006) Docosahexaenoic acid supports cell growth and expression of choline acetyltransferase and muscarinic receptors in NG108-15 cell line. J Mol Neurosci 30:25–26
Fang IM, Yang CH, Yang CM (2014) Docosahexaenoic acid reduces linoleic acid induced monocyte chemoattractant protein-1 expression via PPARγ and nuclear factor-κB pathway in retinal pigment epithelial cells. Mol Nutr Food Res 58:2053–2065
Puskas LG, Kitajka K, Nyakas C, Barcelo-Coblijn G, Farkas T (2003) Short-term administration of omega 3 fatty acids from fish oil results in increased transthyretin transcription in old rat hippocampus. Proc Natl Acad Sci U S A 100:1580–1585
Rojas CV, Martinez JI, Flores I, Hoffman DR, Uauy R (2003) Gene expression analysis in human fetal retinal explants treated with docosahexaenoic acid. Invest Ophthalmol Vis Sci 44:3170–3177
Liu BH, Wang YC, Kuo CF, Cheng WM, Shen TF, Ding ST (2005) The effects of docosahexaenoic acid oil and soybean oil on the expression of lipid metabolism related mRNA in pigs. Asian-Australas J Anim Sci 18:1451–1456
Zhao G, Etherton TD, Martin KR, Vanden Heuvel JP, Gillies PJ et al (2005) Anti-inflammatory effects of polyunsaturated fatty acids in THP-1 cells. Biochem Biophys Res Commun 336:909–917
Calder PC (2006) Polyunsaturated fatty acids and inflammation. Prostaglandins Leukot Essent Fatty Acids 75:197–202
Casañas-Sánchez V, Pérez JA, Fabelo N, Quinto-Alemany D, Díaz ML (2015) Docosahexaenoic (DHA) modulates phospholipid-hydroperoxide glutathione peroxidase (Gpx4) gene expression to ensure self-protection from oxidative damage in hippocampal cells. Front Physiol 6:203
Huang CW, Chen YJ, Yang JT, Chen CY, Ajuwon KM, Chen SE, Su NW, Chen YS et al (2017) Docosahexaenoic acid increases accumulation of adipocyte triacylglycerol through up-regulation of lipogenic gene expression in pigs. Lipids Health Dis 16:33
Kitajka K, Puskás LG, Zvara A et al (2002) The role of n-3 polyunsaturated fatty acids in brain: modulation of rat brain gene expression by dietary n-3 fatty acids. Proc Natl Acad Sci U S A 99:2619–2624
Kitajka K, Sinclair AJ, Weisinger RS, Weisinger HS, Mathai M, Jayasooriya AP, Halver JE, Puskas LG (2004) Effects of dietary omega-3 polyunsaturated fatty acids on brain gene expression. Proc Natl Acad Sci U S A 101:10931–10936
Revelli JP, Pescini R, Muzzin P, Seydoux J, Fitzgerald MG, Fraser CM, Giacobino JP (1991) Changes in beta 1- and beta 2-adrenergic receptor mRNA levels in brown adipose tissue and heart of hypothyroid rats. Biochem J 277:625–629
Rubio A, Raasmaja A, Maia AL, Kim KR, Silva JE (1995) Effects of thyroid hormone on norepinephrine signaling in brown adipose tissue. I. Beta 1- and beta 2-adrenergic receptors and cyclic adenosine 3′,5′-monophosphate generation. Endocrinology 136:3267–3276
Manzano J, Bernal J, Morte B (2007) Influence of thyroid hormones on maturation of rat cerebellar astrocytes. Int J Dev Neurosci 25:171–179
Kirigiti P, Bai Y, Yang YF, Li X, Li B, Brewer G, Machida CA (2001) Agonist-mediated down-regulation of rat beta1-adrenergic receptor transcripts: role of potential post-transcriptional degradation factors. Mol Pharmacol 60:1308–1324
Blaxall BC, Pellett AC, Wu SC, Pende A, Port JD (2000) Purification and characterization of beta-adrenergic receptor mRNA-binding proteins. J Biol Chem 274:4290–4297
Hosoda K, Fitzgerald LR, Vaidya VA, Feussner GK, Fishman PH, Duman RS (1995) Regulation of beta 2-adrenergic receptor mRNA and gene transcription in rat C6 glioma cells: effects of agonist, forskolin, and protein synthesis inhibition. Mol Pharmacol 48:206–211
Serini S, Fasano E, Piccioni E, Monego G, Cittadini ARM, Celleno L, Ranelletti FO, Calviello G (2012) DHA induces apoptosis and differentiation in human melanoma cells in vitro: involvement of HuR-mediated COX-2 mRNA stabilization and β-catenin nuclear translocation. Carcinogenesis 33:164–173
Hadcock JR, Malbon CC (1988) Down-regulation of beta-adrenergic receptors: agonist-induced reduction in receptor mRNA levels. Proc Natl Acad Sci U S A 85:5021–5025
Zhu Z, Tan Z, Li Y, Luo H, Hu X, Tang M, Hescheler J, Mu Y et al (2015) Docosahexaenoic acid alters Gsα localization in lipid raft and potentiates adenylate cyclase. Nutrition 31:1025–1030
Macia E, Ehrlich M, Massol R, Boucrot E, Brunner C, Kirchhausen T (2006) Dynasore, a cell-permeable inhibitor of dynamin. Dev Cell 10:839–850
Hurtado-Lorenzo A, Skinner M, El AJ et al (2006) V-ATPase interacts with ARNO and Arf6 in early endosomes and regulates the protein degradative pathway. Nat Cell Biol 8:124–136
van Dam EM, Stoorvogel W (2002) Dynamin-dependent transferrin receptor recycling by endosome-derived clathrin-coated vesicles. Mol Biol Cell 13:169–182
van Dam EM, Ten Broeke T, Jansen K, Spijkers P, Stoorvogel W (2002) Endocytosed transferrin receptors recycle via distinct dynamin and phosphatidylinositol 3-kinase-dependent pathways. J Biol Chem 277:48876–48883
Robertson SE, Setty SR, Sitaram A, Marks MS, Lewis RE, Chou MM (2006) Extracellular signal-regulated kinase regulates clathrin-independent endosomal trafficking. Mol Biol Cell 17:645–657
Shenoy SK, Drake MT, Nelson CD, Houtz DA, Xiao K, Madabushi S, Reiter E, Premont RT et al (2006) β-Arrestin-dependent, G protein-independent ERK1/2 activation by the β2 adrenergic receptor. J Biol Chem 281:1261–1273
Tohgo A, Choy EW, Gesty-Palmer D, Pierce KL, Laporte S, Oakley RH, Caron MG, Lefkowitz RJ et al (2003) The stability of the G protein-coupled receptor-beta-arrestin interaction determines the mechanism and functional consequence of ERK activation. J Biol Chem 278:6258–6267
Luttrell LM, Ferguson SSG, Daaka Y, Miller WE, Maudsley S, Della Rocca GJ, Lin FT, Kawakatsu H et al (1999) β-Arrestin-dependent formation of β2 adrenergic receptor-Src protein kinase complexes. Science 283:655–661
Candelario J, Tavakoli H, Chachisvilis M (2012) PTH1 receptor is involved in mediating cellular response to long-chain polyunsaturated fatty acids. PLoS One 7:e52583
Daaka Y, Luttrell LM, Ahn S, Della Rocca GJ, Ferguson SSG, Caron MG, Lefkowitz RJ (1998) Essential role for G protein-coupled receptor endocytosis in the activation of mitogen activated protein kinase. J Biol Chem 273:685–688
Vardjan N, Kreft M, Zorec R (2014) Dynamics of β-adrenergic/cAMP signaling and morphological changes in cultured astrocytes. Glia 62:566–579
Funding
The work was carried out from the financial assistance of the Council of Scientific & Industrial Research, New Delhi. MD was a recipient of fellowship also from the Council of Scientific & Industrial Research, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Animal experimentation was approved by the institutional animal ethics committee appointed by CPCSEA (Committee for the Purpose of Control and Supervision of Experiments on Animals) of the animal welfare division under the Ministry of Environment and Forest, Government of India (Registration no. 147/CPCSEA).
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Das, M., Das, S. Docosahexaenoic Acid (DHA) Induced Morphological Differentiation of Astrocytes Is Associated with Transcriptional Upregulation and Endocytosis of β2-AR. Mol Neurobiol 56, 2685–2702 (2019). https://doi.org/10.1007/s12035-018-1260-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1260-0