Abstract
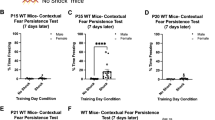
Infantile amnesia (IA) refers to the inability of adults to recall episodic memories from infancy or early childhood. While several hypotheses have been proposed to explain the occurrence of IA, the neurobiological and molecular bases for this accelerated forgetting phenomenon remain elusive. Using hippocampus-dependent object-location memory and contextual fear conditioning tasks, we confirmed that infant mice trained at postnatal day 20 (P20) displayed deficits in long-term memory retention compared to adult (P60) mice. The percentage of CA1 pyramidal neurons expressing phosphorylated cAMP-responsive element-binding protein after fear conditioning was significantly lower in P20 than P60 mice. P20 mice exhibited attenuated basal excitatory synaptic transmission and early-phase long-term potentiation (E-LTP) at Schaffer collateral-CA1 synapses compared to P60 mice, but conversely, P20 mice have a greater susceptibility to induce time-dependent reversal of LTP by low-frequency afferent stimulation than P60 mice. The protein levels of GluN2B subunit of N-methyl-d-aspartate receptors (NMDARs), protein kinase Mζ (PKMζ), and protein phosphatase 2B (PP2B) in hippocampal CA1 region were significantly higher in P20 than P60 mice. We also found that the levels of calcium/calmodulin-dependent protein kinase II α autophosphorylation at Thr286, GluA1 phosphorylation at Ser831, and PKMζ protein biosynthesis occurred during the ensuing maintenance of E-LTP were significantly lower in P20 than P60 mice. Pharmacological blockade of GluN2B-containing NMDARs or PP2B effectively restored deficits of E-LTP and long-term memory retention observed in P20 mice. Altogether, these findings suggest that developmental immaturity of the maintenance mechanisms for E-LTP is linked to the occurrence of IA.






Similar content being viewed by others
References
Madsen HB, Kim JH (2016) Ontogeny of memory: an update on 40 years of work on infantile amnesia. Behav Brain Res 298:4–14
Campbell BA, Spear NE (1972) Ontogeny of memory. Psychol Rev 79:215–236
Callaghan BL, Li S, Richardson R (2013) The elusive engram: what can infantile amnesia tell us about memory? Trends Neurosci 37:47–53
Josselyn SA, Frankland PW (2012) Infantile amnesia: a neurogenic hypothesis. Learn Mem 19:423–433
Rovee-Collier CK, Gekoski MJ (1979) The economics of infancy: a review of conjugate reinforcement. Adv Child Dev Behav 13:195–255
Miller JS, Jagielo JA, Spear NE (1991) Differential effectiveness of various prior-cuing treatments in the reactivation and maintenance of memory. J Exp Psychol Anim Behav Process 17:249–258
Kim JH, Richardson R (2007) Immediate post-reminder injection of GABA agonist midazolam attenuates reactivation of forgotten fear in the infant rat. Behav Neurosci 121:1328–1332
Travaglia A, Bisaz R, Sweet ES, Blitzer RD, Alberini CM (2016) Infantile amnesia reflects a developmental critical period for hippocampal learning. Nat Neurosci 19:1225–1233
Frankland PW, Köhler S, Josselyn SA (2013) Hippocampal neurogenesis and forgetting. Trends Neurosci 36:497–503
Akers KG, Martinez-Canabal A, Restivo L, Yiu AP, De Cristofaro A, Hsiang HL, Wheeler AL, Guskjolen A et al (2014) Hippocampal neurogenesis regulates forgetting during adulthood and infancy. Science 344:598–602
Kandel ER, Dudai Y, Mayford MR (2014) The molecular and systems biology of memory. Cell 157:163–186
Poo MM, Pignatelli M, Ryan TJ, Tonegawa S, Bonhoeffer T, Martin KC, Rudenko A, Tsai LH et al (2016) What is memory? The present state of the engram. BMC Biol 14:40
Nicoll RA, Roche KW (2013) Long-term potentiation: peeling the onion. Neuropharmacology 74:18–22
Cooke SF, Bliss TV (2006) Plasticity in the human central nervous system. Brain 129:1659–1673
Mayford M, Siegelbaum SA, Kandel ER (2012) Synapses and memory storage. Cold Spring Harb Perspect Biol 4:a005751
Winder DG, Sweatt JD (2001) Roles of serine/threonine phosphatases in hippocampal synaptic plasticity. Nat Rev Neurosci 2:461–474
Malenka RC, Bear MF (2004) LTP and LTD: an embarrassment of riches. Neuron 44:5–21
Yasuda H, Barth AL, Stellwagen D, Malenka RC (2003) A developmental switch in the signaling cascades for LTP induction. Nat Neurosci 6:15–16
Huang CC, Chou PH, Yang CH, Hsu KS (2005) Neonatal isolation accelerates the developmental switch in the signalling cascades for long-term potentiation induction. J Physiol 569:789–799
Yang CH, Huang CC, Hsu KS (2012) A critical role for protein tyrosine phosphatase nonreceptor type 5 in determining individual susceptibility to develop stress-related cognitive and morphological changes. J Neurosci 32:7550–7562
Huang YF, Yang CH, Huang CC, Tai MH, Hsu KS (2010) Pharmacological and genetic accumulation of hypoxia-inducible factor-1alpha enhances excitatory synaptic transmission in hippocampal neurons through the production of vascular endothelial growth factor. J Neurosci 30:6080–6093
Huang CC, Liang YC, Hsu KS (2001) Characterization of the mechanism underlying the reversal of long term potentiation by low frequency stimulation at hippocampal CA1 synapses. J Biol Chem 276:48108–48117
Valverde F (1998) Golgi atlas of the postnatal mouse brain, 15th edn. Springer-Verlag, Chicago
Franklin K, Paxinos G (2008) The mouse brain in stereotaxic coordinates, 3rd edn. Elsevier Academic Press, San Diego
Kojima N, Sakamoto T, Endo S, Niki H (2005) Impairment of conditioned freezing to tone, but not to context, in Fyn-transgenic mice: relationship to NMDA receptor subunit 2B function. Eur J Neurosci 21:1359–1369
Mouri A, Noda Y, Shimizu S, Tsujimoto Y, Nabeshima T (2010) The role of cyclophilin D in learning and memory. Hippocampus 20:293–304
Izquierdo LA, Barros DM, Vianna MR, Coitinho A, deDavid e Silva T, Choi H, Moletta B, Medina JH et al (2002) Molecular pharmacological dissection of short- and long-term memory. Cell Mol Neurobiol 22:269–287
Han JH, Kushner SA, Yiu AP, Cole CJ, Matynia A, Brown RA, Neve RL, Guzowski JF et al (2007) Neuronal competition and selection during memory formation. Science 316:457–460
Hussain RJ, Carpenter DO (2001) Development of synaptic responses and plasticity at the SC-CA1 and MF-CA3 synapses in rat hippocampus. Cell Mol Neurobiol 21:357–368
Shenolikar S, Nairn AC (1991) Protein phosphatases: recent progress. Adv Second Messenger Phosphoprotein Res 23:1–121
Blitzer RD, Connor JH, Brown GP, Wong T, Shenolikar S, Iyengar R, Landau EM (1998) Gating of CaMKII by cAMP-regulated protein phosphatase activity during LTP. Science 280:1940–1942
Akers KG, Arruda-Carvalho M, Josselyn SA, Frankland PW (2012) Ontogeny of contextual fear memory formation, specificity, and persistence in mice. Learn Mem 19:598–604
Lisman J, Yasuda R, Raghavachari S (2012) Mechanisms of CaMKII action in long-term potentiation. Nat Rev Neurosci 13:169–182
Herring BE, Nicoll RA (2016) Long-term potentiation: from CaMKII to AMPA receptor trafficking. Annu Rev Physiol 78:351–365
Anggono V, Huganir RL (2012) Regulation of AMPA receptor trafficking and synaptic plasticity. Curr Opin Neurobiol 22:461–469
Lee HK, Barbarosie M, Kameyama K, Bear MF, Huganir RL (2000) Regulation of distinct AMPA receptor phosphorylation sites during bidirectional synaptic plasticity. Nature 405:955–959
Ling DS, Benardo LS, Serrano PA, Blace N, Kelly MT, Crary JF, Sacktor TC (2002) Protein kinase Mζ is necessary and sufficient for LTP maintenance. Nat Neurosci 5:295–296
Sacktor TC (2011) How does PKMζ maintain long-term memory? Nat Rev Neurosci 12:9–15
Volk LJ, Bachman JL, Johnson R, Yu Y, Huganir RL (2013) PKMζ is not required for hippocampal synaptic plasticity, learning and memory. Nature 493:420–423
Westmark PR, Westmark CJ, Wang S, Levenson J, O'Riordan KJ, Burger C, Malter JS (2010) Pin1 and PKMζ sequentially control dendritic protein synthesis. Sci Signal 3:ra18
Schuette SR, Fernández-Fernández D, Lamla T, Rosenbrock H, Hobson S (2016) Overexpression of protein kinase Mζ in the hippocampus enhances long-term potentiation and long-term contextual but not cued fear memory in rats. J Neurosci 36:4313–4324
Yashiro K, Philpot BD (2008) Regulation of NMDA receptor subunit expression and its implications for LTD, LTP, and metaplasticity. Neuropharmacology 55:1081–1094
Paoletti P, Bellone C, Zhou Q (2013) NMDA receptor subunit diversity: impact on receptor properties, synaptic plasticity and disease. Nat Rev Neurosci 14:383–400
Sachser RM, Santana F, Crestani AP, Lunardi P, Pedraza LK, Quillfeldt JA, Hardt O, Alvares Lde O (2016) Forgetting of long-term memory requires activation of NMDA receptors, L-type voltage-dependent Ca2+ channels, and calcineurin. Sci Rep 6:22771
Wang YT, Huang CC, Lin YS, Huang WF, Yang CY, Lee CC, Yeh CM, Hsu KS (2017) Conditional deletion of Eps8 reduces hippocampal synaptic plasticity and impairs cognitive function. Neuropharmacology 112:113–123
Dong Z, Han H, Li H, Bai Y, Wang W, Tu M, Peng Y, Zhou L et al (2015) Long-term potentiation decay and memory loss are mediated by AMPAR endocytosis. J Clin Invest 125:234–247
Frey U, Huang YY, Kandel ER (1993) Effects of cAMP simulate a late stage of LTP in hippocampal CA1 neurons. Science 260:1661–1664
Kelleher RJ 3rd, Govindarajan A, Tonegawa S (2004) Translational regulatory mechanisms in persistent forms of synaptic plasticity. Neuron 44:59–73
Cowan CS, Callaghan BL, Richardson R (2016) The effects of a probiotic formulation (Lactobacillus rhamnosus and L. helveticus) on developmental trajectories of emotional learning in stressed infant rats. Transl Psychiatry 6:e823
Shinohara K, Hata T (2014) Post-acquisition hippocampal NMDA receptor blockade sustains retention of spatial reference memory in Morris water maze. Behav Brain Res 259:261–267
Liu X, Ramirez S, Pang PT, Puryear CB, Govindarajan A, Deisseroth K, Tonegawa S (2012) Optogenetic stimulation of a hippocampal engram activates fear memory recall. Nature 484:381–385
Tanaka KZ, Pevzner A, Hamidi AB, Nakazawa Y, Graham J, Wiltgen BJ (2014) Cortical representations are reinstated by the hippocampus during memory retrieval. Neuron 84:347–354
Lu YM, Jia Z, Janus C, Henderson JT, Gerlai R, Wojtowicz JM, Roder JC (1997) Mice lacking metabotropic glutamate receptor 5 show impaired learning and reduced CA1 long-term potentiation (LTP) but normal CA3 LTP. J Neurosci 17:5196–5205
Thiels E, Urban NN, Gonzalez-Burgos GR, Kanterewicz BI, Barrionuevo G, Chu CT, Oury TD, Klann E (2000) Impairment of long-term potentiation and associative memory in mice that overexpress extracellular superoxide dismutase. J Neurosci 20:7631–7639
Plath N, Ohana O, Dammermann B, Errington ML, Schmitz D, Gross C, Mao X, Engelsberg A et al (2006) Arc/Arg3.1 is essential for the consolidation of synaptic plasticity and memories. Neuron 52:437–444
Acknowledgements
We thank the technical services provided by the Bio-image Core Facility of the National Core Facility Program for Biotechnology, Ministry of Science and Technology, Taiwan.
Funding
This work was supported by research grants from the Ministry of Science and Technology (MOST 106-2320-B-006-026-MY3) and the National Health Research Institute (NHRI-EX106-10613NI and NHRI-EX107-10713NI), Taiwan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experimental procedures complied with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee of National Cheng Kung University.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Tsai, TC., Huang, CC. & Hsu, KS. Infantile Amnesia Is Related to Developmental Immaturity of the Maintenance Mechanisms for Long-Term Potentiation. Mol Neurobiol 56, 907–919 (2019). https://doi.org/10.1007/s12035-018-1119-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1119-4