Abstract
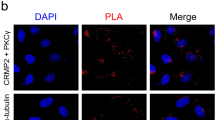
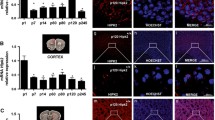
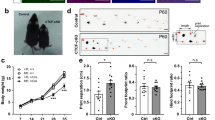
Purkinje cell dendritic development is severely compromised after chronic activation of protein kinase C (PKC). In a recent transgenic mouse model of spinocerebellar ataxia 14, the ser361-to-gly (S361G) mutation of the protein kinase C gamma (PKCγ) gene was expressed in Purkinje cells. Purkinje cells from these mutant mice in organotypic slice cultures have the same stunted dendritic tree as Purkinje cells after pharmacological activation of PKC. Because the transgene is exclusively present in Purkinje cells, cerebellar tissue from these mice is an attractive starting material for searching genes which might be interacting with PKCγ in Purkinje cells for inducing the stunted dendritic growth. We have performed a microarray analysis and identified several candidate genes with an increased messenger RNA (mRNA) expression in the PKCγ-S361G transgenic Purkinje cells. Out of these candidates, we have further studied carbonic anhydrase 8 (CA8). We show here that CA8 mRNA and protein expression is strongly induced in PKCγ-S361G transgenic Purkinje cells. Overexpression of CA8 in Purkinje cells in dissociated cultures strongly inhibited Purkinje cell dendritic development and produced a dendritic phenotype similar to PKCγ-S361G. There was no evidence for a direct binding of CA8 to either PKCγ or the type 1 IP3 receptor. Knockdown of CA8 with miRNA did not alter Purkinje cell dendritic development and did not protect Purkinje cells in dissociated cultures from the stunted dendritic growth induced by PKCγ-S361G or by PKC activation. Our results indicate that CA8 is a novel important regulator of Purkinje cell dendritic development and that its expression is controlled by PKCγ activity.







Similar content being viewed by others
References
Ito M, Yamaguchi K, Nagao S, Yamazaki T (2014) Long-term depression as a model of cerebellar plasticity. Prog Brain Res 210:1–30. doi:10.1016/B978-0-444-63356-9.00001-7
Metzger F, Kapfhammer JP (2000) Protein kinase C activity modulates dendritic differentiation of rat Purkinje cells in cerebellar slice cultures. Eur J Neurosci 12:1993–2005. doi:10.1046/j.1460-9568.2000.00086.x
Schrenk K, Kapfhammer JP, Metzger F (2003) Altered dendritic development of cerebellar Purkinje cells in slice cultures from protein kinase C gamma-deficient mice. Neuroscience 110:675–689. doi:10.1016/S0306-4522(01)00559-0
Kapfhammer JP (2004) Cellular and molecular control of dendritic growth and development of cerebellar Purkinje cells. Prog Histochem Cytochem 39:131–182
Gugger OS, Hartmann J, Birnbaumer L, Kapfhammer JP (2012) P/Q-type and T-type calcium channels, but not type 3 transient receptor potential cation channels, are involved in inhibition of dendritic growth after chronic metabotropic glutamate receptor type 1 and protein kinase C activation in cerebellar Purkinje cells. Eur J Neurosci 35:20–33. doi:10.1111/j.1460-9568.2011.07942.x
Becker EB, Oliver PL, Glitsch MD, Banks GT, Achilli F, Hardy A, Nolan PM, Fisher EM, Davies KE (2009) A point mutation in TRPC3 causes abnormal Purkinje cell development and cerebellar ataxia in Moonwalker mice. Proc Natl Acad Sci U S A 106:6706–6711. doi:10.1073/pnas.0810599106
Huang H, Nagaraja RY, Garside ML, Akemann W, Knöpfel T, Empson RM (2010) Contribution of plasma membrane Ca ATPase to cerebellar synapse function. World J Biol Chem 1:95–102. doi:10.4331/wjbc.v1.i5.95
Sherkhane P, Kapfhammer JP (2013) The plasma membrane Ca2 + -ATPase2 (PMCA2) is involved in the regulation of Purkinje cell dendritic growth in cerebellar organotypic slice cultures. Neural Plast 2013:321685. doi:10.1155/2013/321685
Pandolfo M, van de Warrenburg BP (2005) Spinocerebellar ataxia type 14: opening a new door in dominant ataxia research? Neurology 64:1113–1114. doi:10.1212/01.WNL.0000160013.10806.53
Chen DH, Raskind WH, Bird TD (2012) Spinocerebellar ataxia type 14. Handb Clin Neurol 103:555–559. doi:10.1016/B978-0-444-51892-7.00036-X
Verbeek DS, Knight MA, Harmison GG, Fischbeck KH, Howell BW (2005) Protein kinase C gamma mutations in spinocerebellar ataxia 14 increase kinase activity and alter membrane targeting. Brain 128:436–442. doi:10.1093/brain/awh378
Adachi N, Kobayashi T, Takahashi H, Kawasaki T, Shirai Y, Ueyama T, Matsuda T, Seki T, Sakai N, Saito N (2008) Enzymological analysis of mutant protein kinase Cgamma causing spinocerebellar ataxia type 14 and dysfunction in Ca2+ homeostasis. J Biol Chem 283:19854–19863. doi:10.1074/jbc.M801492200
Chen DH, Cimino PJ, Ranum LP, Zoghbi HY, Yabe I, Schut L, Margolis RL, Lipe HP, Feleke A, Matsushita M, Wolff J, Morgan C, Lau D, Fernandez M, Sasaki H, Raskind WH, Bird TD (2005) The clinical and genetic spectrum of spinocerebellar ataxia 14. Neurology 64:1258–1260. doi:10.1212/01
Ji J, Hassler ML, Shimobayashi E, Paka N, Streit R, Kapfhammer JP (2014) Increased protein kinase C gamma activity induces Purkinje cell pathology in a mouse model of spinocerebellar ataxia 14. Neurobiol Dis 70:1–11. doi:10.1016/j.nbd.2014.06.002
Jiao Y, Yan J, Zhao Y, Donahue LR, Beamer WG, Li X, Roe BA, Ledoux MS, Gu W (2005) Carbonic anhydrase-related protein VIII deficiency is associated with a distinctive lifelong gait disorder in waddles mice. Genetics 171:1239–1246. doi:10.1534/genetics.105.044487
Hirasawa M, Xu X, Trask RB, Maddatu TP, Johnson BA, Naggert JK, Nishina PM, Ikeda A (2007) Carbonic anhydrase related protein 8 mutation results in aberrant synaptic morphology and excitatory synaptic function in the cerebellum. Mol Cell Neurosci 35:161–170. doi:10.1016/j.mcn.2007.02.013
Wagner W, McCroskery S, Hammer JA 3rd (2011) An efficient method for the long-term and specific expression of exogenous cDNAs in cultured Purkinje neurons. J Neurosci Methods 200:95–105. doi:10.1016/j.jneumeth.2011.06.006
Kapfhammer JP (2010) Cerebellar slice cultures. In: Doering LC (ed) Protocols for neural cell culture, 4th edn. Humana Press, New York, pp 285–298
Tomomura M, Rice DS, Morgan JI, Yuzaki M (2001) Purification of Purkinje cells by fluorescence-activated cell sorting from transgenic mice that express green fluorescent protein. Eur J Neurosci 14:57–63. doi:10.1046/j.0953-816x.2001.01624.x
Sjöblom B, Elleby B, Wallgren K, Jonsson BH, Lindskog S (1996) Two point mutations convert a catalytically inactive carbonic anhydrase-related protein (CARP) to an active enzyme. FEBS Lett 398:322–325. doi:10.1016/S0014-5793(96)01263-X
Hirota J, Ando H, Hamada K, Mikoshiba K (2003) Carbonic anhydrase-related protein is a novel binding protein for inositol 1,4,5-trisphosphate receptor type 1. Biochem J 372:435–241. doi:10.1042/BJ20030110
Banerjee S, Hasan G (2005) The InsP3 receptor: its role in neuronal physiology and neurodegeneration. Bioessays 27:1035–1047. doi:10.1002/bies.20298
Lamont MG, Weber JT (2015) Mice deficient in carbonic anhydrase type 8 exhibit motor dysfunctions and abnormal calcium dynamics in the somatic region of cerebellar granule cells. Behav Brain Res 286:11–16. doi:10.1016/j.bbr.2015.02.035
Aspatwar A, Tolvanen ME, Ortutay C, Parkkila S (2014) Carbonic anhydrase related proteins: molecular biology and evolution. Subcell Biochem 75:135–156. doi:10.1007/978-94-007-7359-2_8
Kaya N, Aldhalaan H, Al-Younes B, Colak D, Shuaib T, Al-Mohaileb F, Al-Sugair A, Nester M, Al-Yamani S, Al-Bakheet A, Al-Hashmi N, Al-Sayed M, Meyer B, Jungbluth H, Al-Owain M (2011) Phenotypical spectrum of cerebellar ataxia associated with a novel mutation in the CA8 gene, encoding carbonic anhydrase (CA) VIII. Am J Med Genet B Neuropsychiatr Genet 156B:826–834. doi:10.1002/ajmg.b.31227
Türkmen S, Guo G, Garshasbi M, Hoffmann K, Alshalah AJ, Mischung C, Kuss A, Humphrey N, Mundlos S, Robinson PN (2009) CA8 mutations cause a novel syndrome characterized by ataxia and mild mental retardation with predisposition to quadrupedal gait. PLoS Genet 2009, e1000487. doi:10.1371/journal.pgen.100048
Höftberger R, Sabater L, Velasco F, Ciordia R, Dalmau J, Graus F (2014) Carbonic anhydrase-related protein VIII antibodies and paraneoplastic cerebellar degeneration. Neuropathol Appl Neurobiol 40:650–653. doi:10.1111/nan.12118
van de Leemput J, Chandran J, Knight MA, Holtzclaw LA, Scholz S, Cookson MR, Houlden H, Gwinn-Hardy K, Fung HC, Lin X, Hernandez D, Simon-Sanchez J, Wood NW, Giunti P, Rafferty I, Hardy J, Storey E, Gardner RJ, Forrest SM, Fisher EM, Russell JT, Cai H, Singleton AB (2007) Deletion at ITPR1 underlies ataxia in mice and spinocerebellar ataxia 15 in humans. PLoS Genet 3, e108. doi:10.1371/journal.pgen.0030108
Schorge S, van de Leemput J, Singleton A, Houlden H, Hardy J (2010) Human ataxias: a genetic dissection of inositol triphosphate receptor (ITPR1)-dependent signaling. Trends Neurosci 33:211–219. doi:10.1016/j.tins.2010.02.005
Zhuang GZ, Keeler B, Grant J, Bianchi L, Fu ES, Zhang YP, Erasso DM, Cui JG, Wiltshire T, Li Q, Hao S, Sarantopoulos KD, Candiotti K, Wishnek SM, Smith SB, Maixner W, Diatchenko L, Martin ER, Levitt RC (2015) Carbonic anhydrase-8 regulates inflammatory pain by inhibiting the ITPR1-cytosolic free calcium pathway. PLoS One 10:e0118273. doi:10.1371/journal.pone.0118273
Vermassen E, Parys JB, Mauger JP (2004) Subcellular distribution of the inositol 1,4,5-trisphosphate receptors: functional relevance and molecular determinants. Biol Cell 96:3–17. doi:10.1016/j.biolcel.2003.11.004
Acknowledgments
We thank Dr. John A. Hammer III (National Heart, Lung and Blood Institute, NIH, Bethesda, MD, USA) for providing the pL7-mGFP plasmid and Markus Saxer for technical assistance. Dr. Phillipe Demougin from the Life Science Training Facility (LSTF) of the University of Basel performed the microarray experiments. This work was supported by the University of Basel and the Swiss National Science Foundation (31003A-116624).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 70 kb)
Rights and permissions
About this article
Cite this article
Shimobayashi, E., Wagner, W. & Kapfhammer, J.P. Carbonic Anhydrase 8 Expression in Purkinje Cells Is Controlled by PKCγ Activity and Regulates Purkinje Cell Dendritic Growth. Mol Neurobiol 53, 5149–5160 (2016). https://doi.org/10.1007/s12035-015-9444-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9444-3