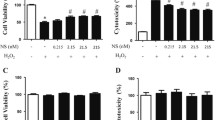
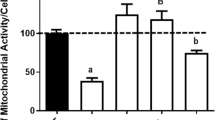
Abstract
Brain-derived neurotrophic factor (BDNF), in addition to its neurotrophic action, also possesses antioxidant activities. However, the underlying mechanisms remain to be fully defined. Sestrin2 is a stress-responsive gene implicated in the cellular defense against oxidative stress. Currently, the potential functions of sestrin2 in nervous system, in particular its correlation with neurotrophic factors, have not been well established. In this study, we hypothesized that BDNF may enhance sestrin2 expression to confer neuronal resistance against oxidative stress induced by 3-nitropropionic acid (3-NP), an irreversible mitochondrial complex II inhibitor, and characterized the molecular mechanisms underlying BDNF induction of sestrin2 in primary rat cortical cultures. We found that BDNF-mediated sestrin2 expression in cortical neurons required formation of nitric oxide (NO) with subsequent production of 3′,5′-cyclic guanosine monophosphate (cGMP) and activation of cGMP-dependent protein kinase (PKG). BDNF induced localization of nuclear factor-kappaB (NF-κB) subunits p65 and p50 into neuronal nuclei that required PKG activities. Interestingly, BDNF exposure led to formation of a protein complex containing at least PKG-1 and p65/p50, which bound to sestrin2 promoter with resultant upregulation of its protein products. Finally, BDNF preconditioning mitigated production of reactive oxygen species (ROS) as a result of 3-NP exposure; this antioxidative effect of BDNF was dependent upon PKG activity, NF-κB, and sestrin2. Taken together, our results indicated that BDNF enhances sestrin2 expression to confer neuronal resistance against oxidative stress induced by 3-NP through attenuation of ROS formation; furthermore, BDNF induction of sestrin2 requires activation of a pathway involving NO/PKG/NF-κB.








Similar content being viewed by others
References
Quintanilla RA, Johnson GV (2009) Role of mitochondrial dysfunction in the pathogenesis of Huntington’s disease. Brain Res Bull 80(4-5):242–247
Alston TA, Mela L, Bright HJ (1977) 3-Nitropropionate, the toxic substance of Indigofera, is a suicide inactivator of succinate dehydrogenase. Proc Natl Acad Sci U S A 74(9):3767–3771
Liot G, Bossy B, Lubitz S, Kushnareva Y, Sejbuk N, Bossy-Wetzel E (2009) Complex II inhibition by 3-NP causes mitochondrial fragmentation and neuronal cell death via an NMDA- and ROS-dependent pathway. Cell Death Differ 16(6):899–909
Beal MF, Brouillet E, Jenkins BG, Ferrante RJ, Kowall NW, Miller JM, Storey E, Srivastava R et al (1993) Neurochemical and histologic characterization of striatal excitotoxic lesions produced by the mitochondrial toxin 3-nitropropionic acid. J Neurosci 13(10):4181–4192
Brouillet E, Jacquard C, Bizat N, Blum D (2005) 3-Nitropropionic acid: a mitochondrial toxin to uncover physiopathological mechanisms underlying striatal degeneration in Huntington’s disease. J Neurochem 95(6):1521–1540
Wu CL, Hwang CS, Yang DI (2009) Protective effects of brain-derived neurotrophic factor against neurotoxicity of 3-nitropropionic acid in rat cortical neurons. Neurotoxicology 30(4):718–726
Baydyuk M, Xu B (2014) BDNF signaling and survival of striatal neurons. Front Cell Neurosci 8:254
Bothwell M (2014) NGF, BDNF, NT3, and NT4. Handb Exp Pharmacol 220:3–15
Mattson MP, Lovell MA, Furukawa K, Markesbery WR (1995) Neurotrophic factors attenuate glutamate-induced accumulation of peroxides, elevation of intracellular Ca2+ concentration, and neurotoxicity and increase antioxidant enzyme activities in hippocampal neurons. J Neurochem 65(4):1740–1751
Lee B, Cao R, Choi YS, Cho HY, Rhee AD, Hah CK, Hoyt KR, Obrietan K (2009) The CREB/CRE transcriptional pathway: protection against oxidative stress-mediated neuronal cell death. J Neurochem 108(5):1251–1265
Wu CL, Yin JH, Hwang CS, Chen SD, Yang DY, Yang DI (2012) c-Jun-dependent sulfiredoxin induction mediates BDNF protection against mitochondrial inhibition in rat cortical neurons. Neurobiol Dis 46(2):450–462
Chan SH, Wu CW, Chang AY, Hsu KS, Chan JY (2010) Transcriptional upregulation of brain-derived neurotrophic factor in rostral ventrolateral medulla by angiotensin II: significance in superoxide homeostasis and neural regulation of arterial pressure. Circ Res 107(9):1127–1139
Budanov AV, Shoshani T, Faerman A, Zelin E, Kamer I, Kalinski H, Gorodin S, Fishman A et al (2002) Identification of a novel stress-responsive gene Hi95 involved in regulation of cell viability. Oncogene 21(39):6017–6031
Essler S, Dehne N, Brune B (2009) Role of sestrin2 in peroxide signaling in macrophages. FEBS Lett 583(21):3531–3535
Sanli T, Linher-Melville K, Tsakiridis T, Singh G (2012) Sestrin2 modulates AMPK subunit expression and its response to ionizing radiation in breast cancer cells. PLoS One 7(2):e32035
Shin BY, Jin SH, Cho IJ, Ki SH (2012) Nrf2-ARE pathway regulates induction of Sestrin-2 expression. Free Radic Biol Med 53(4):834–841
Zhou D, Zhan C, Zhong Q, Li S (2013) Upregulation of sestrin-2 expression via P53 protects against 1-methyl-4-phenylpyridinium (MPP+) neurotoxicity. J Mol Neurosci 51(3):967–975
Kallenborn-Gerhardt W, Lu R, Syhr KM, Heidler J, von Melchner H, Geisslinger G, Bangsow T, Schmidtko A (2013) Antioxidant activity of sestrin 2 controls neuropathic pain after peripheral nerve injury. Antioxid Redox Signal 19(17):2013–2023
Wu CL, Chen SD, Yin JH, Hwang CS, Yang DI (2010) Erythropoietin and sonic hedgehog mediate the neuroprotective effects of brain-derived neurotrophic factor against mitochondrial inhibition. Neurobiol Dis 40(1):146–154
Ju TC, Yang YT, Yang DI (2004) Protective effects of S-nitrosoglutathione against neurotoxicity of 3-nitropropionic acid in rat. Neurosci Lett 362(3):226–231
Huang CY, Yang HI, Chen SD, Shaw FZ, Yang DI (2008) Protective effects of lipopolysaccharide preconditioning against nitric oxide neurotoxicity. J Neurosci Res 86(6):1277–1289
Patapoutian A, Reichardt LF (2001) Trk receptors: mediators of neurotrophin action. Curr Opin Neurobiol 11(3):272–280
Chang SH, Hwang CS, Yin JH, Chen SD, Yang DI (2015) Oncostatin M-dependent Mcl-1 induction mediated by JAK1/2-STAT1/3 and CREB contributes to bioenergetic improvements and protective effects against mitochondrial dysfunction in cortical neurons. Biochim Biophys Acta-Mol Cell Res (in press)
Tang CM, Hwang CS, Chen SD, Yang DI (2011) Neuroprotective mechanisms of minocycline against sphingomyelinase/ceramide toxicity: roles of Bcl-2 and thioredoxin. Free Radic Biol Med 50(6):710–721
Kurauchi Y, Hisatsune A, Isohama Y, Sawa T, Akaike T, Katsuki H (2013) Nitric oxide/soluble guanylyl cyclase signaling mediates depolarization-induced protection of rat mesencephalic dopaminergic neurons from MPP(+) cytotoxicity. Neuroscience 231:206–215
Astort F, Mercau M, Giordanino E, Degese MS, Caldareri L, Coso O, Cymeryng CB (2014) Nitric oxide sets off an antioxidant response in adrenal cells: involvement of sGC and Nrf2 in HO-1 induction. Nitric Oxide 37(1-10
Collier J, Vallance P (1989) Second messenger role for NO widens to nervous and immune systems. Trends Pharmacol Sci 10(11):427–431
Ju TC, Chen SD, Liu CC, Yang DI (2005) Protective effects of S-nitrosoglutathione against amyloid beta-peptide neurotoxicity. Free Radic Biol Med 38(7):938–949
Zhang XY, Wu XQ, Deng R, Sun T, Feng GK, Zhu XF (2013) Upregulation of sestrin 2 expression via JNK pathway activation contributes to autophagy induction in cancer cells. Cell Signal 25(1):150–158
He B, Weber GF (2003) Phosphorylation of NF-kappaB proteins by cyclic GMP-dependent kinase. A noncanonical pathway to NF-kappaB activation. Eur J Biochem 270(10):2174–2185
Budanov AV, Sablina AA, Feinstein E, Koonin EV, Chumakov PM (2004) Regeneration of peroxiredoxins by p53-regulated sestrins, homologs of bacterial AhpD. Science 304(5670):596–600
Eid AA, Lee DY, Roman LJ, Khazim K, Gorin Y (2013) Sestrin 2 and AMPK connect hyperglycemia to Nox4-dependent endothelial nitric oxide synthase uncoupling and matrix protein expression. Mol Cell Biol 33(17):3439–3460
Maiuri MC, Malik SA, Morselli E, Kepp O, Criollo A, Mouchel PL, Carnuccio R, Kroemer G (2009) Stimulation of autophagy by the p53 target gene Sestrin2. Cell Cycle 8(10):1571–1576
Lee J, Giordano S, Zhang J (2012) Autophagy, mitochondria and oxidative stress: cross-talk and redox signalling. Biochem J 441(2):523–540
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132(1):27–42
Gudi T, Lohmann SM, Pilz RB (1997) Regulation of gene expression by cyclic GMP-dependent protein kinase requires nuclear translocation of the kinase: identification of a nuclear localization signal. Mol Cell Biol 17(9):5244–5254
Bae SH, Sung SH, Oh SY, Lim JM, Lee SK, Park YN, Lee HE, Kang D et al (2013) Sestrins activate Nrf2 by promoting p62-dependent autophagic degradation of Keap1 and prevent oxidative liver damage. Cell Metab 17(1):73–84
Biteau B, Labarre J, Toledano MB (2003) ATP-dependent reduction of cysteine-sulphinic acid by S. cerevisiae sulphiredoxin. Nature 425(6961):980–984
Chang TS, Jeong W, Woo HA, Lee SM, Park S, Rhee SG (2004) Characterization of mammalian sulfiredoxin and its reactivation of hyperoxidized peroxiredoxin through reduction of cysteine sulfinic acid in the active site to cysteine. J Biol Chem 279(49):50994–51001
Puerta E, Hervias I, Barros-Minones L, Jordan J, Ricobaraza A, Cuadrado-Tejedor M, Garcia-Osta A, Aguirre N (2010) Sildenafil protects against 3-nitropropionic acid neurotoxicity through the modulation of calpain, CREB, and BDNF. Neurobiol Dis 38(2):237–245
Yu Z, Zhou D, Cheng G, Mattson MP (2000) Neuroprotective role for the p50 subunit of NF-kappaB in an experimental model of Huntington’s disease. J Mol Neurosci 15(1):31–44
Gupta S, Sharma B (2014) Pharmacological benefit of I(1)-imidazoline receptors activation and nuclear factor kappa-B (NF-kappaB) modulation in experimental Huntington’s disease. Brain Res Bull 102:57–68
Lorenz JE, Kallenborn-Gerhardt W, Lu R, Syhr KM, Eaton P, Geisslinger G, Schmidtko A (2014) Oxidant-induced activation of cGMP-dependent protein kinase I alpha mediates neuropathic pain after peripheral nerve injury. Antioxid Redox Signal 21(10):1504–1515
Acknowledgments
This study was supported by the National Science Council/Ministry of Science and Technology in Taiwan (NSC 101-2314-B-010-042MY2 and MOST 103-2314-B-010-013MY3 to Ding-I Yang), Ministry of Education in Taiwan-Aim for the Top University Plan (104AC-B5 to Ding-I Yang), Department of Health in Taipei City Government (10301-62-003 and 10401-62-022 to Ding-I Yang and Chi-Shin Hwang), and Cheng Hsin General Hospital (103F003C16 and CY10417 to Ding-I Yang and Jiu-Haw Yin).
Conflict of Interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Supplementary Figures: The entire Western blots shown in the present study, except β-actin, GAPDH, and histone3 that served as the respective loading controls for total proteins, cytosolic proteins, and nuclear proteins, are demonstrated in the corresponding supplementary figures. The numbers on the left side of each blot indicate the molecular weight markers in kDa. Because no immunoblots are presented in Fig. 6, there is no Suppl. Fig. 6.
Suppl. Fig. 1 and 2
(GIF 252 kb)
Suppl. Fig. 3 and 4
(GIF 273 kb)
Suppl. Fig. 5
(GIF 226 kb)
Suppl. Fig. 7
(GIF 229 kb)
Rights and permissions
About this article
Cite this article
Wu, CL., Chen, SD., Yin, JH. et al. Nuclear Factor-kappaB-Dependent Sestrin2 Induction Mediates the Antioxidant Effects of BDNF Against Mitochondrial Inhibition in Rat Cortical Neurons. Mol Neurobiol 53, 4126–4142 (2016). https://doi.org/10.1007/s12035-015-9357-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9357-1