Abstract
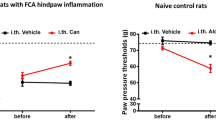
Activation of cannabinoid receptor-2 (CB2) results in β-endorphin release from keratinocytes, which then acts on primary afferent neurons to inhibit nociception. However, the underlying mechanism is still unknown. The CB2 receptor is generally thought to couple to Gi/o to inhibit cAMP production, which cannot explain the peripheral stimulatory effects of CB2 receptor activation. In this study, we found that in a keratinocyte cell line, the Gβγ subunits from Gi/o, but not Gαs, were involved in CB2 receptor activation-induced β-endorphin release. Inhibition of MAPK kinase, but not PLC, abolished CB2 receptor activation-induced β-endorphin release. Also, CB2 receptor activation significantly increased intracellular Ca2+. Treatment with BAPTA-AM or thapsigargin blocked CB2 receptor activation-induced β-endorphin release. Using a rat model of inflammatory pain, we showed that the MAPK kinase inhibitor PD98059 abolished the peripheral effect of the CB2 receptor agonist on nociception. We thus present a novel mechanism of CB2 receptor activation-induced β-endorphin release through Gi/o-Gβγ-MAPK-Ca2+ signaling pathway. Our data also suggest that stimulation of MAPK contributes to the peripheral analgesic effect of CB2 receptor agonists.






Similar content being viewed by others
References
Howlett AC (2005) Cannabinoid receptor signaling. Handb Exp Pharmacol 168:53–79
Munro S, Thomas KL, Abu-Shaar M (1993) Molecular characterization of a peripheral receptor for cannabinoids. Nature 365(6441):61–65. doi:10.1038/365061a0
Guindon J, Hohmann AG (2008) Cannabinoid CB2 receptors: a therapeutic target for the treatment of inflammatory and neuropathic pain. Br J Pharmacol 153(2):319–334. doi:10.1038/sj.bjp.0707531
Anand P, Whiteside G, Fowler CJ, Hohmann AG (2009) Targeting CB2 receptors and the endocannabinoid system for the treatment of pain. Brain Res Rev 60(1):255–266. doi:10.1016/j.brainresrev.2008.12.003
Ibrahim MM, Porreca F, Lai J, Albrecht PJ, Rice FL, Khodorova A, Davar G, Makriyannis A (2005) CB2 cannabinoid receptor activation produces antinociception by stimulating peripheral release of endogenous opioids. Proc Natl Acad Sci U S A 102(8):3093–3098. doi:10.1073/pnas.0409888102
Katritch V, Cherezov V, Stevens RC (2012) Diversity and modularity of G protein-coupled receptor structures. Trends Pharmacol Sci 33(1):17–27. doi:10.1016/j.tips.2011.09.003
Birnbaumer L (1992) Receptor-to-effector signaling through G proteins: roles for beta gamma dimers as well as alpha subunits. Cell 71(7):1069–1072
Mizuta K, Mizuta F, Xu D, Masaki E, Panettieri RA Jr, Emala CW (2011) Gi-coupled gamma-aminobutyric acid-B receptors cross-regulate phospholipase C and calcium in airway smooth muscle. Am J Respir Cell Mol Biol 45(6):1232–1238. doi:10.1165/rcmb.2011-0088OC
Surve CR, Lehmann D, Smrcka AV (2014) A chemical biology approach demonstrates G protein betagamma subunits are sufficient to mediate directional neutrophil chemotaxis. J Biol Chem 289(25):17791–17801. doi:10.1074/jbc.M114.576827
Berridge MJ, Irvine RF (1989) Inositol phosphates and cell signalling. Nature 341(6239):197–205. doi:10.1038/341197a0
Rittner HL, Labuz D, Richter JF, Brack A, Schafer M, Stein C, Mousa SA (2007) CXCR1/2 ligands induce p38 MAPK-dependent translocation and release of opioid peptides from primary granules in vitro and in vivo. Brain Behav Immun 21(8):1021–1032. doi:10.1016/j.bbi.2007.05.002
Glass M, Felder CC (1997) Concurrent stimulation of cannabinoid CB1 and dopamine D2 receptors augments cAMP accumulation in striatal neurons: evidence for a Gs linkage to the CB1 receptor. J Neurosci 17(14):5327–5333
Bonhaus DW, Chang LK, Kwan J, Martin GR (1998) Dual activation and inhibition of adenylyl cyclase by cannabinoid receptor agonists: evidence for agonist-specific trafficking of intracellular responses. J Pharmacol Exp Ther 287(3):884–888
Zimmermann M (1983) Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16(2):109–110
Su TF, Zhang LH, Peng M, Wu CH, Pan W, Tian B, Shi J, Pan HL et al (2011) Cannabinoid CB2 receptors contribute to upregulation of beta-endorphin in inflamed skin tissues by electroacupuncture. Mol Pain 7:98. doi:10.1186/1744-8069-7-98
Hargreaves K, Dubner R, Brown F, Flores C, Joris J (1988) A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 32(1):77–88
Khan GM, Chen SR, Pan HL (2002) Role of primary afferent nerves in allodynia caused by diabetic neuropathy in rats. Neuroscience 114(2):291–299
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53(1):55–63
Smrcka AV (2008) G protein betagamma subunits: central mediators of G protein-coupled receptor signaling. Cell Mol Life Sci 65(14):2191–2214. doi:10.1007/s00018-008-8006-5
Lehmann DM, Seneviratne AM, Smrcka AV (2008) Small molecule disruption of G protein beta gamma subunit signaling inhibits neutrophil chemotaxis and inflammation. Mol Pharmacol 73(2):410–418. doi:10.1124/mol.107.041780
Muto Y, Nagao T, Urushidani T (1997) The putative phospholipase C inhibitor U73122 and its negative control, U73343, elicit unexpected effects on the rabbit parietal cell. J Pharmacol Exp Ther 282(3):1379–1388
Alessi DR, Cuenda A, Cohen P, Dudley DT, Saltiel AR (1995) PD 098059 is a specific inhibitor of the activation of mitogen-activated protein kinase kinase in vitro and in vivo. J Biol Chem 270(46):27489–27494
Cabot PJ, Carter L, Gaiddon C, Zhang Q, Schafer M, Loeffler JP, Stein C (1997) Immune cell-derived beta-endorphin. Production, release, and control of inflammatory pain in rats. J Clin Invest 100(1):142–148. doi:10.1172/JCI119506
Chen XP, Yang W, Fan Y, Luo JS, Hong K, Wang Z, Yan JF, Chen X et al (2010) Structural determinants in the second intracellular loop of the human cannabinoid CB1 receptor mediate selective coupling to G(s) and G(i). Br J Pharmacol 161(8):1817–1834. doi:10.1111/j.1476-5381.2010.01006.x
Khodorova A, Navarro B, Jouaville LS, Murphy JE, Rice FL, Mazurkiewicz JE, Long-Woodward D, Stoffel M et al (2003) Endothelin-B receptor activation triggers an endogenous analgesic cascade at sites of peripheral injury. Nat Med 9(8):1055–1061
Wintzen M, Yaar M, Burbach JP, Gilchrest BA (1996) Proopiomelanocortin gene product regulation in keratinocytes. J Investig Dermatol 106(4):673–678
Funasaka Y, Chakraborty AK, Yodoi J, Ichihashi M (2001) The effect of thioredoxin on the expression of proopiomelanocortin-derived peptides, the melanocortin 1 receptor and cell survival of normal human keratinocytes. J Investig Dermatol Symp Proc 6(1):32–37. doi:10.1046/j.0022-202x.2001.00002.x
Geng X, Du XN, Rusinova R, Liu BY, Li F, Zhang X, Chen XJ, Logothetis DE et al (2009) Specificity of Gbetagamma signaling depends on Galpha subunit coupling with G-protein-sensitive K(+) channels. Pharmacology 84(2):82–90. doi:10.1159/000227772
Chan JS, Lee JW, Ho M, Wong Y (2000) Preactivation permits subsequent stimulation of phospholipase C by Gi coupled receptor. Mol Pharmacol 57:700–708
Faure M, Voyno-Yasenetskaya TA, Bourne HR (1994) cAMP and beta gamma subunits of heterotrimeric G proteins stimulate the mitogen-activated protein kinase pathway in COS-7 cells. J Biol Chem 269(11):7851–7854
Correa F, Hernangomez M, Mestre L, Loria F, Spagnolo A, Docagne F, Di Marzo V, Guaza C (2010) Anandamide enhances IL-10 production in activated microglia by targeting CB(2) receptors: roles of ERK1/2, JNK, and NF-kappaB. Glia 58(2):135–147. doi:10.1002/glia.20907
Kim SY, Han Y, Oh M, Kim W, Oh K, Lee SC, Bae K, Han B (2014) DUSP4 regulates neuronal differentiation and calcium homeostasis by modulating ERK1/2 phosphorylation. Stem Cells Dev. doi:10.1089/scd.2014.0434
Zhuang ZY, Gerner P, Woolf CJ, Ji RR (2005) ERK is sequentially activated in neurons, microglia, and astrocytes by spinal nerve ligation and contributes to mechanical allodynia in this neuropathic pain model. Pain 114(1–2):149–159. doi:10.1016/j.pain.2004.12.022
Liu CY, Lu ZY, Li N, Yu LH, Zhao YF, Ma B (2015) The role of large-conductance, calcium-activated potassium channels in a rat model of trigeminal neuropathic pain. Cephalalgia 35(1):16–35. doi:10.1177/0333102414534083
Acknowledgments
This work was supported by a grant from the National Natural Science Foundation of China (#81173328, #31300598), funds for the Doctoral Program of Higher Education of China (20120142110021), and the Fundamental Research Funds for the Central Universities, HUST:2010JC065.
Conflict of Interest
The authors declare no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, F., Zhang, LH., Su, TF. et al. Signaling Mechanism of Cannabinoid Receptor-2 Activation-Induced β-Endorphin Release. Mol Neurobiol 53, 3616–3625 (2016). https://doi.org/10.1007/s12035-015-9291-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9291-2