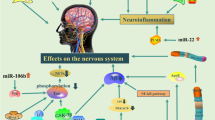
Abstract
The multifactorial aspect of neurodegenerative diseases has posed challenges in terms of understanding various mechanistic cues behind these ailments. The fact that single microRNA (miRNA) molecules can regulate multiple genes and associated pathways makes these molecules interesting for studies within the area of age-associated neurodegenerative diseases. miRNAs are endogenous, evolutionarily conserved, 20–23 nucleotide non-coding RNAs, which were first discovered in Caenorhabditis elegans. They play a key role in gene regulation and are known to be deregulated in many disease conditions. Steady regulations of miRNAs are required for normal biological processes. One of the crucial miRNA molecules let-7 is highly conserved and is known to be required for development and viability. It acts as a regulator for oncogenes and insulin-PI3K-mTOR pathway genes. Upregulation of let-7 impairs glucose homeostasis and results in degeneration of neurons, while its downregulation leads to cancer. Maturation of let-7 in cancer subjects is inhibited by lin-28, an RNA-binding protein inhibitor. This highlights the importance of let-7 miRNAs in various diseases and developmental processes. This article provides an overview on the functions of let-7 and its probable association with various neurodegenerative diseases.



Similar content being viewed by others
References
Meza-Sosa KF, Valle-Garcia D, Pedraza-Alva G, Perez-Martinez L (2012) Role of microRNAs in central nervous system development and pathology. J Neurosci Res 90(1):1–12. doi:10.1002/jnr.22701
Brenner JL, Kemp BJ, Abbott AL (2012) The mir-51 family of microRNAs functions in diverse regulatory pathways in Caenorhabditis elegans. PLoS One 7(5):e37185. doi:10.1371/journal.pone.0037185
Clark AM, Goldstein LD, Tevlin M, Tavare S, Shaham S, Miska EA (2010) The microRNA miR-124 controls gene expression in the sensory nervous system of Caenorhabditis elegans. Nucleic Acids Res 38(11):3780–3793. doi:10.1093/nar/gkq083
Roush S, Slack FJ (2008) The let-7 family of microRNAs. Trends Cell Biol 18(10):505–516. doi:10.1016/j.tcb.2008.07.007
Ambros V, Bartel B, Bartel DP, Burge CB, Carrington JC, Chen X, Dreyfuss G, Eddy SR et al (2003) A uniform system for microRNA annotation. RNA 9(3):277–279
Thum T (2012) MicroRNA therapeutics in cardiovascular medicine. EMBO Mol Med 4(1):3–14. doi:10.1002/emmm.201100191
Lanford RE, Hildebrandt-Eriksen ES, Petri A, Persson R, Lindow M, Munk ME, Kauppinen S, Orum H (2010) Therapeutic silencing of microRNA-122 in primates with chronic hepatitis C virus infection. Science 327(5962):198–201. doi:10.1126/science.1178178
Chen C, Ridzon DA, Broomer AJ, Zhou Z, Lee DH, Nguyen JT, Barbisin M, Xu NL et al (2005) Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res 33(20):e179. doi:10.1093/nar/gni178
Leidinger P, Backes C, Deutscher S, Schmitt K, Mueller SC, Frese K, Haas J, Ruprecht K et al (2013) A blood based 12-miRNA signature of Alzheimer disease patients. Genome Biol 14(7):R78. doi:10.1186/gb-2013-14-7-r78
Niwa R, Zhou F, Li C, Slack FJ (2008) The expression of the Alzheimer’s amyloid precursor protein-like gene is regulated by developmental timing microRNAs and their targets in Caenorhabditis elegans. Dev Biol 315(2):418–425. doi:10.1016/j.ydbio.2007.12.044
Reinhart BJ, Slack FJ, Basson M, Pasquinelli AE, Bettinger JC, Rougvie AE, Horvitz HR, Ruvkun G (2000) The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature 403(6772):901–906. doi:10.1038/35002607
Slack FJ, Basson M, Liu Z, Ambros V, Horvitz HR, Ruvkun G (2000) The lin-41 RBCC gene acts in the C. elegans heterochronic pathway between the let-7 regulatory RNA and the LIN-29 transcription factor. Mol Cell 5(4):659–669
Sokol NS, Xu P, Jan YN, Ambros V (2008) Drosophila let-7 microRNA is required for remodeling of the neuromusculature during metamorphosis. Genes Dev 22(12):1591–1596. doi:10.1101/gad.1671708
Boyerinas B, Park SM, Hau A, Murmann AE, Peter ME (2010) The role of let-7 in cell differentiation and cancer. Endocr Relat Cancer 17(1):F19–F36. doi:10.1677/ERC-09-0184
Dimmeler S, Nicotera P (2013) MicroRNAs in age-related diseases. EMBO Mol Med 5(2):180–190. doi:10.1002/emmm.201201986
Yao J, Hennessey T, Flynt A, Lai E, Beal MF, Lin MT (2010) MicroRNA-related cofilin abnormality in Alzheimer’s disease. PLoS One 5(12):e15546. doi:10.1371/journal.pone.0015546
Ma L, Wei L, Wu F, Hu Z, Liu Z, Yuan W (2013) Advances with microRNAs in Parkinson’s disease research. Drug Des Dev Ther 7:1103–1113. doi:10.2147/DDDT.S48500
Wang X, Cao L, Wang Y, Wang X, Liu N, You Y (2012) Regulation of let-7 and its target oncogenes (review). Oncol Lett 3(5):955–960. doi:10.3892/ol.2012.609
Yun J, Frankenberger CA, Kuo WL, Boelens MC, Eves EM, Cheng N, Liang H, Li WH et al (2011) Signalling pathway for RKIP and Let-7 regulates and predicts metastatic breast cancer. EMBO J 30(21):4500–4514. doi:10.1038/emboj.2011.312
Zhu H, Shyh-Chang N, Segre AV, Shinoda G, Shah SP, Einhorn WS, Takeuchi A, Engreitz JM et al (2011) The Lin28/let-7 axis regulates glucose metabolism. Cell 147(1):81–94. doi:10.1016/j.cell.2011.08.033
Lunn JS, Sakowski SA, Hur J, Feldman EL (2011) Stem cell technology for neurodegenerative diseases. Ann Neurol 70(3):353–361. doi:10.1002/ana.22487
Ambegaokar SS, Roy B, Jackson GR (2010) Neurodegenerative models in Drosophila: polyglutamine disorders, Parkinson disease, and amyotrophic lateral sclerosis. Neurobiol Dis 40(1):29–39. doi:10.1016/j.nbd.2010.05.026
Tzatsos A, Bardeesy N (2008) Ink4a/Arf regulation by let-7b and Hmga2: a genetic pathway governing stem cell aging. Cell Stem Cell 3(5):469–470. doi:10.1016/j.stem.2008.10.008
Ewald CY, Li C (2012) Caenorhabditis elegans as a model organism to study APP function. Exp Brain Res 217(3–4):397–411. doi:10.1007/s00221-011-2905-7
Hornsten A, Lieberthal J, Fadia S, Malins R, Ha L, Xu X, Daigle I, Markowitz M et al (2007) APL-1, a Caenorhabditis elegans protein related to the human beta-amyloid precursor protein, is essential for viability. Proc Natl Acad Sci U S A 104(6):1971–1976. doi:10.1073/pnas.0603997104
Ewald CY, Raps DA, Li C (2012) APL-1, the Alzheimer’s Amyloid precursor protein in Caenorhabditis elegans, modulates multiple metabolic pathways throughout development. Genetics 191(2):493–507. doi:10.1534/genetics.112.138768
Walsh DM, Minogue AM, Sala Frigerio C, Fadeeva JV, Wasco W, Selkoe DJ (2007) The APP family of proteins: similarities and differences. Biochem Soc Trans 35(Pt 2):416–420. doi:10.1042/BST0350416
Wiese M, Antebi A, Zheng H (2010) Intracellular trafficking and synaptic function of APL-1 in Caenorhabditis elegans. PloS One 5(9). doi:10.1371/journal.pone.0012790
Williamson J, Goldman J, Marder KS (2009) Genetic aspects of Alzheimer disease. Neurologist 15(2):80–86. doi:10.1097/NRL.0b013e318187e76b
Wollen KA (2010) Alzheimer’s disease: the pros and cons of pharmaceutical, nutritional, botanical, and stimulatory therapies, with a discussion of treatment strategies from the perspective of patients and practitioners. Altern Med Rev J Clin Ther 15(3):223–244
Liu T, Xu ZZ, Park CK, Berta T, Ji RR (2010) Toll-like receptor 7 mediates pruritus. Nat Neurosci 13(12):1460–1462. doi:10.1038/nn.2683
Lehmann SM, Kruger C, Park B, Derkow K, Rosenberger K, Baumgart J, Trimbuch T, Eom G et al (2012) An unconventional role for miRNA: let-7 activates Toll-like receptor 7 and causes neurodegeneration. Nat Neurosci 15(6):827–835. doi:10.1038/nn.3113
Chen PS, Su JL, Cha ST, Tarn WY, Wang MY, Hsu HC, Lin MT, Chu CY et al (2011) miR-107 promotes tumor progression by targeting the let-7 microRNA in mice and humans. J Clin Invest 121(9):3442–3455. doi:10.1172/JCI45390
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39(6):889–909
Lim KL, Lim TM (2003) Molecular mechanisms of neurodegeneration in Parkinson’s disease: clues from Mendelian syndromes. IUBMB Life 55(6):315–322. doi:10.1080/1521654031000153030
Asikainen S, Rudgalvyte M, Heikkinen L, Louhiranta K, Lakso M, Wong G, Nass R (2010) Global microRNA expression profiling of Caenorhabditis elegans Parkinson’s disease models. J Mol Neurosci: MN 41(1):210–218. doi:10.1007/s12031-009-9325-1
Gehrke S, Imai Y, Sokol N, Lu B (2010) Pathogenic LRRK2 negatively regulates microRNA-mediated translational repression. Nature 466(7306):637–641. doi:10.1038/nature09191
Junn E, Mouradian MM (2012) MicroRNAs in neurodegenerative diseases and their therapeutic potential. Pharmacol Ther 133(2):142–150. doi:10.1016/j.pharmthera.2011.10.002
Bicchi I, Morena F, Montesano S, Polidoro M, Martino S (2013) MicroRNAs and molecular mechanisms of neurodegeneration. Genes 4(2):244–263. doi:10.3390/genes4020244
Blokhuis AM, Groen EJ, Koppers M, van den Berg LH, Pasterkamp RJ (2013) Protein aggregation in amyotrophic lateral sclerosis. Acta Neuropathol 125(6):777–794. doi:10.1007/s00401-013-1125-6
Lagier-Tourenne C, Polymenidou M, Cleveland DW (2010) TDP-43 and FUS/TLS: emerging roles in RNA processing and neurodegeneration. Hum Mol Genet 19(R1):R46–R64. doi:10.1093/hmg/ddq137
Chesebro B (2003) Introduction to the transmissible spongiform encephalopathies or prion diseases. Br Med Bull 66:1–20
Saba R, Goodman CD, Huzarewich RL, Robertson C, Booth SA (2008) A miRNA signature of prion induced neurodegeneration. PLoS One 3(11):e3652. doi:10.1371/journal.pone.0003652
Bilen J, Liu N, Burnett BG, Pittman RN, Bonini NM (2006) MicroRNA pathways modulate polyglutamine-induced neurodegeneration. Mol Cell 24(1):157–163. doi:10.1016/j.molcel.2006.07.030
Marti E, Pantano L, Banez-Coronel M, Llorens F, Minones-Moyano E, Porta S, Sumoy L, Ferrer I et al (2010) A myriad of miRNA variants in control and Huntington’s disease brain regions detected by massively parallel sequencing. Nucleic Acids Res 38(20):7219–7235. doi:10.1093/nar/gkq575
Kocerha J, Xu Y, Prucha MS, Zhao D, Chan AW (2014) microRNA-128a dysregulation in transgenic Huntington’s disease monkeys. Mol Brain 7:46. doi:10.1186/1756-6606-7-46
Feng J, Sun G, Yan J, Noltner K, Li W, Buzin CH, Longmate J, Heston LL et al (2009) Evidence for X-chromosomal schizophrenia associated with microRNA alterations. PLoS One 4(7):e6121. doi:10.1371/journal.pone.0006121
Acknowledgements
The authors thank Director CDRI for infrastructural support. AN is supported by CSIR NWP BSC0118 (EpiHeD). CDRI Communication no. for this publication is: 8946.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shamsuzzama, Kumar, L., Haque, R. et al. Role of MicroRNA Let-7 in Modulating Multifactorial Aspect of Neurodegenerative Diseases: an Overview. Mol Neurobiol 53, 2787–2793 (2016). https://doi.org/10.1007/s12035-015-9145-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9145-y