Abstract
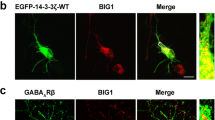
GABAA receptors (GABAARs) mediate the majority of fast synaptic inhibition. Trafficking regulation and protein–protein interactions that maintain the appropriate number of GABAARs at the cell surface are considered to be important mechanisms for controlling the strength of synaptic inhibition. Here, we report that BIG1, a brefeldin A (BFA)-inhibited guanine nucleotide-exchange factor (GEF) which has a known role in vesicle trafficking, is a new binding partner of GABAARs. Treatment of neurons with BFA, an uncompetitive inhibitor of BIG1 GEF activity, or depletion of BIG1 by small RNA interference (siRNA) significantly decreased GABAARs at the neuronal surface and suppressed GABA-gated influx of chloride ions. Over-expression of HA-tagged BIG1-E793K, a dominant-negative mutant, also significantly decreased GABAARs at the neuronal surface, but had no effect on the total amount of GABAARs. Inhibition of GABAAR endocytosis by muscimol increased both GABAARs and BIG1 at the neuronal surface in a time-dependent fashion, and this increase could be abolished by bicuculline. Finally, depletion of BIG1 by siRNA inhibited the muscimol-stimulated increase of GABAARs. Those data suggest an important function of BIG1 in trafficking of GABAARs to the cell surface through its GEF activity. Thus, we identify an important role of BIG1 in modulating GABA-gated Cl− influx through the regulation of cell surface expression of GABAARs.





Similar content being viewed by others
References
Jacob TC, Moss SJ, Jurd R (2008) GABA(A) receptor trafficking and its role in the dynamic modulation of neuronal inhibition. Nat Rev Neurosci 9(5):331–343
Vithlani M, Terunuma M, Moss SJ (2011) The dynamic modulation of GABA(A) receptor trafficking and its role in regulating the plasticity of inhibitory synapses. Physiol Rev 91(3):1009–1022
Fritschy JM (2008) Epilepsy, E/I balance and GABA(A) receptor plasticity. Front Mol Neurosci 1:5
Goodkin HP, Joshi S, Mtchedlishvili Z, Brar J, Kapur J (2008) Subunit-specific trafficking of GABA(A) receptors during status epilepticus. J Neurosci 28(10):2527–2538
Kalueff AV, Nutt DJ (2007) Role of GABA in anxiety and depression. Depress Anxiety 24(7):495–517
Charych EI, Liu F, Moss SJ, Brandon NJ (2009) GABA(A) receptors and their associated proteins: implications in the etiology and treatment of schizophrenia and related disorders. Neuropharmacology 57(5–6):481–495
Kumar S, Fleming RL, Morrow AL (2004) Ethanol regulation of gamma-aminobutyric acid A receptors: genomic and nongenomic mechanisms. Pharmacol Ther 101(3):211–226
Knabl J, Witschi R, Hosl K, Reinold H, Zeilhofer UB, Ahmadi S, Brockhaus J, Sergejeva M, Hess A, Brune K, Fritschy JM, Rudolph U, Mohler H, Zeilhofer HU (2008) Reversal of pathological pain through specific spinal GABAA receptor subtypes. Nature 451(7176):330–334
Saliba RS, Michels G, Jacob TC, Pangalos MN, Moss SJ (2007) Activity-dependent ubiquitination of GABA(A) receptors regulates their accumulation at synaptic sites. J Neurosci 27(48):13341–13351
Leil TA, Chen ZW, Chang CS, Olsen RW (2004) GABAA receptor-associated protein traffics GABAA receptors to the plasma membrane in neurons. J Neurosci 24(50):11429–11438
Chen ZW, Chang CS, Leil TA, Olcese R, Olsen RW (2005) GABAA receptor-associated protein regulates GABAA receptor cell-surface number in Xenopus laevis oocytes. Mol Pharmacol 68(1):152–159
Kittler JT, Rostaing P, Schiavo G, Fritschy JM, Olsen R, Triller A, Moss SJ (2001) The subcellular distribution of GABARAP and its ability to interact with NSF suggest a role for this protein in the intracellular transport of GABA(A) receptors. Mol Cell Neurosci 18(1):13–25
Goto H, Terunuma M, Kanematsu T, Misumi Y, Moss SJ, Hirata M (2005) Direct interaction of N-ethylmaleimide-sensitive factor with GABA(A) receptor beta subunits. Mol Cell Neurosci 30(2):197–206
Mizokami A, Kanematsu T, Ishibashi H, Yamaguchi T, Tanida I, Takenaka K, Nakayama KI, Fukami K, Takenawa T, Kominami E, Moss SJ, Yamamoto T, Nabekura J, Hirata M (2007) Phospholipase C-related inactive protein is involved in trafficking of gamma2 subunit-containing GABA(A) receptors to the cell surface. J Neurosci 27(7):1692–1701
Kanematsu T, Fujii M, Mizokami A, Kittler JT, Nabekura J, Moss SJ, Hirata M (2007) Phospholipase C-related inactive protein is implicated in the constitutive internalization of GABAA receptors mediated by clathrin and AP2 adaptor complex. J Neurochem 101(4):898–905
Keller CA, Yuan X, Panzanelli P, Martin ML, Alldred M, Sassoe-Pognetto M, Luscher B (2004) The gamma2 subunit of GABA(A) receptors is a substrate for palmitoylation by GODZ. J Neurosci 24(26):5881–5891
Fang C, Deng L, Keller CA, Fukata M, Fukata Y, Chen G, Luscher B (2006) GODZ-mediated palmitoylation of GABA(A) receptors is required for normal assembly and function of GABAergic inhibitory synapses. J Neurosci 26(49):12758–12768
Beck M, Brickley K, Wilkinson HL, Sharma S, Smith M, Chazot PL, Pollard S, Stephenson FA (2002) Identification, molecular cloning, and characterization of a novel GABAA receptor-associated protein, GRIF-1. J Biol Chem 277(33):30079–30090
Lin YF, Xu X, Cape A, Li S, Li XJ (2010) Huntingtin-associated protein-1 deficiency in orexin-producing neurons impairs neuronal process extension and leads to abnormal behavior in mice. J Biol Chem 285(21):15941–15949
Kittler JT, Thomas P, Tretter V, Bogdanov YD, Haucke V, Smart TG, Moss SJ (2004) Huntingtin-associated protein 1 regulates inhibitory synaptic transmission by modulating gamma-aminobutyric acid type A receptor membrane trafficking. Proc Natl Acad Sci U S A 101(34):12736–12741
Farhan H, Reiterer V, Kriz A, Hauri HP, Pavelka M, Sitte HH, Freissmuth M (2008) Signal-dependent export of GABA transporter 1 from the ER-Golgi intermediate compartment is specified by a C-terminal motif. J Cell Sci 121(Pt 6):753–761
Charych EI, Yu W, Miralles CP, Serwanski DR, Li X, Rubio M, De Blas AL (2004) The brefeldin A-inhibited GDP/GTP exchange factor 2, a protein involved in vesicular trafficking, interacts with the beta subunits of the GABA receptors. J Neurochem 90(1):173–189
Moss J, Vaughan M (1999) Activation of toxin ADP-ribosyltransferases by eukaryotic ADP-ribosylation factors. Mol Cell Biochem 193(1–2):153–157
Morinaga N, Tsai SC, Moss J, Vaughan M (1996) Isolation of a brefeldin A-inhibited guanine nucleotide-exchange protein for ADP ribosylation factor (ARF) 1 and ARF3 that contains a Sec7-like domain. Proc Natl Acad Sci U S A 93(23):12856–12860
Lowery J, Szul T, Styers M, Holloway Z, Oorschot V, Klumperman J, Sztul E (2013) The Sec7 guanine nucleotide exchange factor GBF1 regulates membrane recruitment of BIG1 and BIG2 guanine nucleotide exchange factors to the trans-Golgi network (TGN). J Biol Chem 288(16):11532–11545
Sheen VL, Ganesh VS, Topcu M, Sebire G, Bodell A, Hill RS, Grant PE, Shugart YY, Imitola J, Khoury SJ, Guerrini R, Walsh CA (2004) Mutations in ARFGEF2 implicate vesicle trafficking in neural progenitor proliferation and migration in the human cerebral cortex. Nat Genet 36(1):69–76
Shen X, Meza-Carmen V, Puxeddu E, Wang G, Moss J, Vaughan M (2008) Interaction of brefeldin A-inhibited guanine nucleotide-exchange protein (BIG) 1 and kinesin motor protein KIF21A. Proc Natl Acad Sci U S A 105(48):18788–18793
Cuitino L, Godoy JA, Farias GG, Couve A, Bonansco C, Fuenzalida M, Inestrosa NC (2010) Wnt-5a modulates recycling of functional GABAA receptors on hippocampal neurons. J Neurosci 30(25):8411–8420
Czondor K, Ellwanger K, Fuchs YF, Lutz S, Gulyas M, Mansuy IM, Hausser A, Pfizenmaier K, Schlett K (2009) Protein kinase D controls the integrity of Golgi apparatus and the maintenance of dendritic arborization in hippocampal neurons. Mol Biol Cell 20(7):2108–2120
Shen X, Hong MS, Moss J, Vaughan M (2007) BIG1, a brefeldin A-inhibited guanine nucleotide-exchange protein, is required for correct glycosylation and function of integrin beta1. Proc Natl Acad Sci U S A 104(4):1230–1235
Hoffman JF, Geibel JP (2005) Fluorescent imaging of Cl− in Amphiuma red blood cells: how the nuclear exclusion of Cl− affects the plasma membrane potential. Proc Natl Acad Sci U S A 102(3):921–926
Feng Y, Yu S, Lasell TK, Jadhav AP, Macia E, Chardin P, Melancon P, Roth M, Mitchison T, Kirchhausen T (2003) Exo1: a new chemical inhibitor of the exocytic pathway. Proc Natl Acad Sci U S A 100(11):6469–6474
Chun J, Shapovalova Z, Dejgaard SY, Presley JF, Melancon P (2008) Characterization of class I and II ADP-ribosylation factors (Arfs) in live cells: GDP-bound class II Arfs associate with the ER-Golgi intermediate compartment independently of GBF1. Mol Biol Cell 19(8):3488–3500
Anders N, Jurgens G (2008) Large ARF guanine nucleotide exchange factors in membrane trafficking. Cell Mol Life Sci 65(21):3433–3445
Peyroche A, Paris S, Jackson CL (1996) Nucleotide exchange on ARF mediated by yeast Gea1 protein. Nature 384(6608):479–481
Shinotsuka C, Yoshida Y, Kawamoto K, Takatsu H, Nakayama K (2002) Overexpression of an ADP-ribosylation factor-guanine nucleotide exchange factor, BIG2, uncouples brefeldin A-induced adaptor protein-1 coat dissociation and membrane tubulation. J Biol Chem 277(11):9468–9473
Kittler JT, Delmas P, Jovanovic JN, Brown DA, Smart TG, Moss SJ (2000) Constitutive endocytosis of GABAA receptors by an association with the adaptin AP2 complex modulates inhibitory synaptic currents in hippocampal neurons. J Neurosci 20(21):7972–7977
Porcher C, Hatchett C, Longbottom RE, McAinch K, Sihra TS, Moss SJ, Thomson AM, Jovanovic JN (2011) Positive feedback regulation between gamma-aminobutyric acid type A (GABA(A)) receptor signaling and brain-derived neurotrophic factor (BDNF) release in developing neurons. J Biol Chem 286(24):21667–21677
Eshaq RS, Stahl LD, Stone R 2nd, Smith SS, Robinson LC, Leidenheimer NJ (2010) GABA acts as a ligand chaperone in the early secretory pathway to promote cell surface expression of GABAA receptors. Brain Res 1346:1–13
Marandi N, Konnerth A, Garaschuk O (2002) Two-photon chloride imaging in neurons of brain slices. Pflugers Arch 445(3):357–365
Coskun T, Baumgartner HK, Chu S, Montrose MH (2002) Coordinated regulation of gastric chloride secretion with both acid and alkali secretion. Am J Physiol Gastrointest Liver Physiol 283(5):G1147–1155
Fukura H, Komiya Y, Igarashi M (1996) Signaling pathway downstream of GABAA receptor in the growth cone. J Neurochem 67(4):1426–1434
Dallwig R, Deitmer JW, Backus KH (1999) On the mechanism of GABA-induced currents in cultured rat cortical neurons. Pflugers Arch 437(2):289–297
Schwartz RD, Yu X (1995) Optical imaging of intracellular chloride in living brain slices. J Neurosci Methods 62(1–2):185–192
Frech MJ, Deitmer JW, Backus KH (1999) Intracellular chloride and calcium transients evoked by gamma-aminobutyric acid and glycine in neurons of the rat inferior colliculus. J Neurobiol 40(3):386–396
Bevensee MO, Apkon M, Boron WF (1997) Intracellular pH regulation in cultured astrocytes from rat hippocampus. II. Electrogenic Na/HCO3 cotransport. J Gen Physiol 110(4):467–483
Acknowledgments
The anti-BIG1 antibody and BIG1 plasmids were kindly provided by Dr. Martha Vaughan and Dr. Joel Moss from the Cardiovascular-Pulmonary Branch of the National Heart, Lung, and Blood Institute, National Institutes of Health. This study was supported by National Natural Science Foundation of China (nos. 31070924 and 81173056) and the Research Fund for the Doctoral Program of Higher Education of China (no. 20100171110052).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
The blank control was analyzed with the vehicle (Krebs-HEPES) in cultured hippocampal neurons (9 DIV) labeled with the Cl– sensitive dye MQAE. Live cell images were taken each 5 seconds. (AVI 3407 kb)
Cl– influx stimulated by GABA (100 mM) in cultured hippocampal neurons (9 DIV) labeled with MQAE was analyzed by a confocal microscope (Zeiss 710). Live cell images were taken each 5 seconds. (AVI 957 kb)
Cultured hippocampal neurons (9 DIV) were treated with 5 μg/ml BFA for 5 min, then subjected to the MQAE paradigm to monitor GABA stimulated (100 mM) Cl– influx. Live cell images were taken each 5 seconds. (AVI 5107 kb)
Cultured hippocampal neurons (9 DIV) were treated with 5 μg/ml BFA for 30 min, then subjected to the MQAE paradigm to monitor GABA stimulated (100 mM) Cl– influx. Live cell images were taken each 5 seconds. (AVI 1599 kb)
Cultured hippocampal neurons (9 DIV) were treated with 100 μM bicuculine for 15 min, and then subjected to the MQAE paradigm to monitor GABA stimulated (100 mM) Cl– influx. Live cell images were taken each 5 seconds. (AVI 1693 kb)
The blank control was analyzed with the vehicle (Krebs-HEPES) in SHSY-5Y cells transfected with negative control siRNA (NC). Live cell images were taken each 5 seconds. (AVI 61 kb)
Cl– influx stimulated by GABA (100 mM) in SHSY-5Y cells transfected with negative control siRNA (NC) was analyzed by a confocal microscope (Zeiss 710). Live cell images were taken each 5 seconds. (AVI 55 kb)
Cl– influx stimulated by GABA (100 mM) in SHSY-5Y cells transfected with BIG1 siRNA (G05) was analyzed by a confocal microscope (Zeiss 710). Live cell images were taken each 5 seconds. (AVI 57 kb)
Fig. S1
Cellular localization of BIG1 and GM130. Hippocampal neurons were fixed and permeabilized, followed by incubation with anti-BIG1 and anti-GM130 antibodies. Images were acquired using a laser scanning confocal microscopy (scale bar, 20 μm). (JPEG 69 kb)
Fig. S2
Effect of BFA treatment on GABA A Rs expression in neurons. Cultured hippocampal neurons (9 DIV) were treated with vehicle (DMSO), or with 5 μg/ml BFA for 30 min. The neurons were fixed, permeabilized, and reacted with anti-GABAARβ2,3 antibodies. Images were acquired using a laser scanning confocal microscopy (scale bar, 20 μm). Fluorescent intensity was quantified with ImageJ software (NIH) in a minimum of 20 cells per slide. The experiments were repeated at least three times. (JPEG 142 kb)
Fig. S3
Effect of BIG1 depletion on GABA A Rs expression in neurons. Hippocampal neurons (7 DIV) transfected with Cy3 labeled NC siRNA or BIG1 (G05) siRNA were fixed, permeabilized, and reacted with anti-GABAARβ2,3 antibodies. Images were acquired using a laser scanning confocal microscopy (Scale bar, 20 μm). Fluorescent intensity was quantified with ImageJ software (NIH) in a minimum of 20 cells per slide. The experiments were repeated at least three times. (JPEG 249 kb)
Fig. S4
Effect of muscimol, bicuculine or BFA on GABA A Rs expression in neurons. Hippocampal neurons (9 DIV) treated with vehicle (DMSO), muscimol alone (50 μM for 15 min; Musc), or bicuculline (100 μM for 10 min; Bic), or bicuculline (100 μM) plus muscimol (50 Μm for 15 min, Musc + Bic,), or BFA (5 μg/ml) plus muscimol (50 μM for 30 min, Musc + BFA). The neurons were fixed, permeabilized, and reacted with anti-GABAARβ2,3 antibodies. Images were acquired using a laser scanning confocal microscopy (Scale bar, 20 μm). Fluorescent intensity was quantified with ImageJ software (NIH) in a minimum of 20 cells per slide. The experiments were repeated at least three times. (JPEG 570 kb)
Rights and permissions
About this article
Cite this article
Li, C., Chen, S., Yu, Y. et al. BIG1, a Brefeldin A-Inhibited Guanine Nucleotide-Exchange Factor, Is Required for GABA-Gated Cl– Influx Through Regulation of GABAA Receptor Trafficking. Mol Neurobiol 49, 808–819 (2014). https://doi.org/10.1007/s12035-013-8558-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-013-8558-8