Abstract
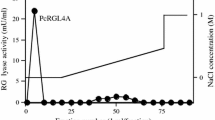
A thermostable, alkaline rhamnogalacturonan lyase (RG lyase) CtRGLf, of family 11 polysaccharide lyase from Clostridium thermocellum was cloned, expressed, purified and biochemically characterised. Both, the full-length CtRGLf (80 kDa) protein and its truncated derivative CtRGL (63.9 kDa) were expressed as soluble proteins and displayed maximum activity against rhamnogalacturonan I (RG I). CtRGLf showed maximum activity at 70 °C, while CtRGL at 60 °C. Both enzymes showed maximum activity at pH 8.5. CtRGLf and CtRGL do not show higher activity on substrates with high β-d-galactopyranose (d-Galp) substitution, this catalytic property deviates from that of some earlier characterised RG lyases which prefer substrates with high d-Galp substitution. The enzyme activity of CtRGLf and CtRGL was enhanced by 1.5 and 1.3 fold, respectively, in the presence of 3 mM of Ca2+ ions. The TLC analysis of the degraded products of RG I, released by the action of CtRGLf and CtRGL revealed the production of RG oligosaccharides as major products confirming their endolytic activity.




Similar content being viewed by others
References
Vincken, J. P., Schols, H. A., & Oomen, R. J. (2003). If homogalacturonan were a side chain of rhamnogalacturonan I. Implications for cell wall architecture. Plant Physiology, 132, 1781–1789.
Ridley, B. L., O’Neill, M. A., & Mohnen, D. (2001). Pectins: structure, biosynthesis, and oligogalacturonide-related signalling. Phytochemistry, 57, 929–967.
Matsunaga, T., Ishii, T., & Matsumoto, S. (2004). Occurrence of the primary cell wall polysaccharide rhamnogalacturonan II in pteridophytes, lycophytes and bryophytes. Implications for the evolution of vascular plants. Plant Physiology, 134, 339–351.
O’Neill, M. A., Warrenfeltz, D., & Kates, K. (1996). Rhamnogalacturonan-II, a pectic polysaccharide in the walls of growing plant cell, forms a dimer that is covalently cross-linked by borate ester in vitro conditions for the formation and hydrolysis of the dimer. Journal of Biological Chemistry, 271, 22923–22930.
Oomen, R. J., Doeswijk, V. H., & Bush, M. S. (2002). In muro fragmentation of the rhamnogalacturonan I backbone in potato (Solanum tuberosum L.) results in a reduction and altered location of the galactan and arabinan side-chains and abnormal periderm development. Plant J, 30, 403–413.
Ochiai, A., Itoh, T., & Kawamata, A. (2007). Plant cell wall degradation by saprophytic Bacillus subtilis strains: gene clusters responsible for rhamnogalacturonande polymerization. Applied and Environment Microbiology, 73, 3803–3813.
Pages, S., Valette, O., & Abdou, L. (2003). A rhamnogalacturonan lyase in the Clostridium cellulolyticum cellulosome. Journal of Bacteriology, 185, 4727–4733.
McKie, V. A., Vincken, J. P., & Voragen, A. G. (2001). A new family of rhamnogalacturonan lyases contains an enzyme that binds to cellulose. Biochemical Journal, 355, 167–177.
Koshland, D. E. (1953). Stereochemistry and the mechanism of enzymatic reactions. Biological Reviews, 28, 416–436.
Moran, F. S., Nasuno, S., & Starr, M. P. (1968). Extracellular and intracellular polygalacturonic acid trans-eliminase of Erwinia carotovora. Archives of Biochemistry and Biophysics, 123, 298–306.
Lombard, V., Golaconda, R. H., & Drula, E. (2014). The Carbohydrate-active enzymes database (CAZy) in 2013. Nucleic Acids Research, 42, D490–D495.
Koukiekolo, R., Cho, H. Y., & Kosugi, A. (2005). Degradation of corn fibre by Clostridium cellulovorans cellulases and hemicellulases and contribution of scaffolding protein CbpA. Applied and Environment Microbiology, 71, 3504–3511.
Aachary, A. A., & Prapulla, S. G. (2011). Xylooligosaccharides (XOS) as an emerging prebiotic: microbial synthesis, utilization, structural characterization, bioactive properties, and applications. Comprehensive Reviews in Food Science and Food Safety, 10, 2–16.
Kashyap, D. R., Vohra, P. K., & Tewari, R. (2001). Application of pectinases in the commercial sector: a review. Bioresource Technology, 77, 215–227.
Fontes, C. M., & Gilbert, H. J. (2010). Cellulosomes: highly efficient nanomachines designed to deconstruct plant cell wall complex carbohydrates. Annual Review of Biochemistry, 79, 655–681.
Lamed, R., Setter, E., & Bayer, E. A. (1983). Characterization of a cellulose-binding, cellulase-containing complex in Clostridium thermocellum. Journal of Bacteriology, 156, 828–836.
Bayer, E. A., Morag, E., & Lamed, R. (1994). The cellulosome- a treasure-trove for biotechnology. Trends in Biotechnology, 12, 379–386.
Das, S. P., Ravindran, R., & Ahmed, S. (2012). Bioethanol production involving recombinant C. thermocellum hydrolytic hemicellulase and fermentative microbes. Applied Biochemistry and Biotechnology, 167, 1475–1488.
Anbar, M., Gul, O., & Lamed, R. (2012). Improved thermostability of Clostridium thermocellum endoglucanase Cel8A by using consensus-guided mutagenesis. Applied and Environmental Microbiology, 78, 3458–3464.
Silva, I. R., Jers, C., & Otten, H. (2014). Design of thermostable rhamnogalacturonan lyase mutants from Bacillus licheniformis by combination of targeted single point mutations. Applied Microbiology and Biotechnology, 98, 4521–4531.
Tartof, K. D., & Hobbs, C. A. (1987). Improved media for growing plasmid and cosmid clones. Bethesda Research Laboratories Focus, 9, 12.
Tanabe, H., Kobayashi, Y., Matuo, Y., Nishi, N., & Wada, F. (1984). Isolation and Fundamental Properties of endo-Pectate Lyase pl-Isozymes from Erwinia carotovora. Agricultural and Biological Chemistry, 48(8), 2113–2120.
Zhang, Z., Xiao, Z., & Linhardt, R. J. (2009). Thin layer chromatography for the separation and analysis of acidic carbohydrates. Journal of Liquid Chromatography & Related Technologies, 32, 1711–1732.
Perlman, D., & Halvorson, H. O. (1983). A putative signal peptidase recognition site and sequence in eukaryotic and prokaryotic signal peptides. Journal of Molecular Biology, 167, 391–409.
Iwai, M., Yamada, H., Ikemoto, T., Matsumoto, S., Fujiwara, D., Takenaka, S., & Sakamoto, T. (2015). Biochemical characterization and overexpression of an endo-rhamnogalacturonan lyase from Penicillium chrysogenum. Molecular Biotechnology, 57(6), 539–548.
Li, S., Yang, X., Bao, M., Wu, Y., Yu, W., & Han, F. (2015). Family 13 carbohydrate-binding module of alginate lyase from Agarivorans sp. L11 enhances its catalytic efficiency and thermostability, and alters its substrate preference and product distribution. FEMS microbiology letters, 362(10), fnv054.
Ochiai, A., Itoh, T., Mikami, B., Hashimoto, W., & Murata, K. (2009). Structural determinants responsible for substrate recognition and mode of action in family 11 polysaccharide lyases. Journal of Biological Chemistry, 284(15), 10181–10189.
Laatu, M., & Condemine, G. (2003). Rhamnogalacturonate lyase RhiE is secreted by the out system in Erwinia chrysanthemi. Journal of Bacteriology, 185, 1642–1649.
Ames, G. L., Mimura, C. S., & Shyamala, V. (1990). Bacterial periplasmic permeases belong to a family of transport proteins operating from Escherichia coli to human: traffic ATPases. FEMS Microbiology Reviews, 6(4), 429–446.
Olano, E., Rimbach, G. H., Gibson, G. R., & Rastall, R. A. (2002). Pectin and pectic-oligosaccharides induce apoptosis in in vitro human colonic adenocarcinoma cells. Anticancer Research, 23(1A), 341–346.
Cheng, H., Zhang, Z., & Leng, J. (2013). The inhibitory effects and mechanisms of rhamnogalacturonan I pectin from potato on HT-29 colon cancer cell proliferation and cell cycle progression. International Journal of Food Science and Nutrition, 64, 36–43.
Acknowledgments
The authors thank Department of Science and Technology, Ministry of Science and Technology, New Delhi, Govt. of India and Fundação para a Ciência e a Tecnologia, Portugal for Indo-Portugal joint project grant (INT/Portugal/P-14/2013). Fellowship provided by Ministry of Human Resource Development, Govt. of India to AD is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Dhillon, A., Fernandes, V.O., Dias, F.M.V. et al. A New Member of Family 11 Polysaccharide Lyase, Rhamnogalacturonan Lyase (CtRGLf) from Clostridium thermocellum . Mol Biotechnol 58, 232–240 (2016). https://doi.org/10.1007/s12033-016-9921-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-016-9921-6