Abstract
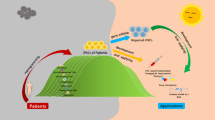
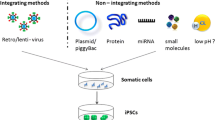
In the field of regenerative medicine, the development of induced pluripotent stem (iPS) cells may represent a potential strategy to overcome the limitations of human embryonic stem cells (ESCs). iPS cells have the potential to mimic human disease, since they carry the genome of the donor. Hypothetically, with iPS cell technology it is possible to screen patients for a genetic cause of disease (genetic mutation), develop cell lines, reprogram them back to iPS cells, finally differentiate them into one or more cell types that develop the disease. Although the creation of multiple lineages with iPS cells can seem limitless, a number of challenges need to be addressed in order to effectively use these cell lines for disease modeling. These include the low efficiency of iPS cell generation without genetic alterations, the possibility of tumor formation in vivo, the random integration of retroviral-based delivery vectors into the genome, and unregulated growth of the remaining cells that are partially reprogrammed and refractory to differentiation. The establishment of protein or RNA-based reprogramming strategies will help generate human iPS cells without permanent genetic alterations. Finally, direct reprogramming strategies can provide rapid production of models of human “diseases in a dish”, without first passing the cells through a pluripotent state, so avoiding the challenges of time-consumming and labor-intensive iPS cell line generation. This review will overview methods to develop iPS cells, current strategies for direct reprogramming, and main applications of iPS cells as human disease model, focusing on human cardiovascular diseases, with the aim to be a potential information resource for biomedical scientists and clinicians who exploit or intend to exploit iPS cell technology in a range of applications.



Similar content being viewed by others
References
Abrams, D. J., Perkin, M. A., Skinner, J. R. (2010). Long QT syndrome. Praxis (Bern 1994) 99, 854–858.
Ambasudhan, R., Talantova, M., Coleman, R., Yuan, X., Zhu, S., Lipton, S. A., et al. (2011). Direct reprogramming of adult human fibroblasts to functional neurons under defined conditions. Cell Stem Cell, 9, 113–118.
Axelrod, F. B. (2004). Familial dysautonomia. Muscle and Nerve, 29, 352–363.
Bartel, D. P. (2004). MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell, 116, 281–297.
Bentires-Alj, M., Kontaridis, M. I., & Neel, B. G. (2006). Stops along the RAS pathway in human genetic disease. Nature Medicine, 12, 283–285.
Bibikova, M., Carroll, D., Segal, D. J., Trautman, J. K., Smith, J., Kim, Y. G., et al. (2001). Stimulation of homologous recombination through targeted cleavage by chimeric nucleases. Molecular and Cellular Biology, 21, 289–297.
Bibikova, M., Golic, M., Golic, K. G., & Carroll, D. (2002). Targeted chromosomal cleavage and mutagenesis in Drosophila using zinc-finger nucleases. Genetics, 161, 1169–1175.
Brunet, E., Simsek, D., Tomishima, M., DeKelver, R., Choi, V. M., Gregory, P., et al. (2009). Chromosomal translocations induced at specified loci in human stem cells. Proceedings of the National Academy of Sciences of the United States of America, 106, 10620–10625.
Caiazzo, M., Dell’anno, M. T., Dvoretskova, E., Lazarevic, D., Taverna, S., Leo, D., et al. (2011). Direct generation of functional dopaminergic neurons from mouse and human fibroblasts. Nature, 476, 224–227.
Callaway, E. (2011). Cells snag top modelling job. Nature, 469, 279.
Carey, B. W., Markoulaki, S., Hanna, J., Saha, K., Gao, Q., Mitalipova, M., et al. (2009). Reprogramming of murine and human somatic cells using a single polycistronic vector. Proceedings of the National Academy of Sciences of the United States of America, 106, 157–162.
Carvajal-Vergara, X., Sevilla, A., D’Souza, S. L., Ang, Y. S., Schaniel, C., Lee, D. F., et al. (2010). Patient-specific induced pluripotent stem-cell-derived models of LEOPARD syndrome. Nature, 465, 808–812.
Choi, J., Costa, M. L., Mermelstein, C. S., Chagas, C., Holtzer, S., & Holtzer, H. (1990). MyoD converts primary dermal fibroblasts, chondroblasts, smooth muscle, and retinal pigmented epithelial cells into striated mononucleated myoblasts and multinucleated myotubes. Proceedings of the National Academy of Sciences of the United States of America, 87, 7988–7992.
Colman, A. (2008). Induced pluripotent stem cells and human disease. Cell Stem Cell, 3, 236–237.
Coppin, B. D., & Temple, I. K. (1997). Multiple lentigines syndrome (LEOPARD syndrome or progressive cardiomyopathic lentiginosis). Journal of Medical Genetics, 34, 582–586.
Curcic-Stojkovic, O., Nikolic, L., Obradovic, D., Krstic, A., & Radic, A. (1978). Noonan’s syndrome. (Male Turner’s syndrome, Turner-like syndrome). Med Pregl, 31, 299–303.
DeKelver, R. C., Choi, V. M., Moehle, E. A., Paschon, D. E., Hockemeyer, D., Meijsing, S. H., et al. (2010). Functional genomics, proteomics, and regulatory DNA analysis in isogenic settings using zinc finger nuclease-driven transgenesis into a safe harbor locus in the human genome. Genome Research, 20, 1133–1142.
Dimos, J. T., Rodolfa, K. T., Niakan, K. K., Weisenthal, L. M., Mitsumoto, H., Chung, W., et al. (2008). Induced pluripotent stem cells generated from patients with ALS can be differentiated into motor neurons. Science, 321, 1218–1221.
Ebert, A. D., Yu, J., Rose, F. F, Jr, Mattis, V. B., Lorson, C. L., Thomson, J. A., et al. (2009). Induced pluripotent stem cells from a spinal muscular atrophy patient. Nature, 457, 277–280.
***EMD Millipore. (2008). ENStem-A adherent human neural progenitors: A new source of primary human neural cells. Application Note.
Esteban, M. A., Wang, T., Qin, B., Yang, J., Qin, D., Cai, J., et al. (2010). Vitamin C enhances the generation of mouse and human induced pluripotent stem cells. Cell Stem Cell, 6, 71–79.
Esteban, M. A., Xu, J., Yang, J., Peng, M., Qin, D., Li, W., et al. (2009). Generation of induced pluripotent stem cell lines from Tibetan miniature pig. Journal of Biological Chemistry, 284, 17634–17640.
Hardouin, S. N., & Nagy, A. (2000). Mouse models for human disease. Clinical Genetics, 57, 237–244.
Hedley, P. L., Jorgensen, P., Schlamowitz, S., Wangari, R., Moolman-Smook, J., Brink, P. A., et al. (2009). The genetic basis of long QT and short QT syndromes: A mutation update. Human Mutation, 30, 1486–1511.
Hockemeyer, D., Soldner, F., Beard, C., Gao, Q., Mitalipova, M., DeKelver, R. C., et al. (2009). Efficient targeting of expressed and silent genes in human ESCs and iPSCs using zinc-finger nucleases. Nature Biotechnology, 27, 851–857.
Huangfu, D., Maehr, R., Guo, W., Eijkelenboom, A., Snitow, M., Chen, A. E., et al. (2008). Induction of pluripotent stem cells by defined factors is greatly improved by small-molecule compounds. Nature Biotechnology, 26, 795–797.
Ichida, J. K., Blanchard, J., Lam, K., Son, E. Y., Chung, J. E., Egli, D., et al. (2009). A small-molecule inhibitor of tgf-Beta signaling replaces sox2 in reprogramming by inducing nanog. Cell Stem Cell, 5, 491–503.
Ieda, M., Fu, J. D., Delgado-Olguin, P., Vedantham, V., Hayashi, Y., Bruneau, B. G., et al. (2010). Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell, 142, 375–386.
Itzhaki, I., Maizels, L., Huber, I., Zwi-Dantsis, L., Caspi, O., Winterstern, A., et al. (2011). Modelling the long QT syndrome with induced pluripotent stem cells. Nature, 471, 225–229.
Izumikawa, M., Minoda, R., Kawamoto, K., Abrashkin, K. A., Swiderski, D. L., Dolan, D. F., et al. (2005). Auditory hair cell replacement and hearing improvement by Atoh1 gene therapy in deaf mammals. Nature Medicine, 11, 271–276.
Jackson, S. P., & Bartek, J. (2009). The DNA-damage response in human biology and disease. Nature, 461, 1071–1078.
Kaji, K., Norrby, K., Paca, A., Mileikovsky, M., Mohseni, P., & Woltjen, K. (2009). Virus-free induction of pluripotency and subsequent excision of reprogramming factors. Nature, 458, 771–775.
Kim, J. B., Greber, B., Arauzo-Bravo, M. J., Meyer, J., Park, K. I., Zaehres, H., et al. (2009). Direct reprogramming of human neural stem cells by OCT4. Nature, 461, 649–653.
Koch, P., Kokaia, Z., Lindvall, O., & Brustle, O. (2009). Emerging concepts in neural stem cell research: Autologous repair and cell-based disease modelling. Lancet Neurology, 8, 819–829.
Krizhanovsky, V., & Lowe, S. W. (2009). Stem cells: The promises and perils of p53. Nature, 460, 1085–1086.
Lee, G., Papapetrou, E. P., Kim, H., Chambers, S. M., Tomishima, M. J., Fasano, C. A., et al. (2009). Modelling pathogenesis and treatment of familial dysautonomia using patient-specific iPSCs. Nature, 461, 402–406.
Lombardo, A., Genovese, P., Beausejour, C. M., Colleoni, S., Lee, Y. L., Kim, K. A., et al. (2007). Gene editing in human stem cells using zinc finger nucleases and integrase-defective lentiviral vector delivery. Nature Biotechnology, 25, 1298–1306.
Madonna, R., Shelat, H., De Caterina, R., & Geng, Y. (2011a). Aquaporin-1 is required for vascular development of human induced pluripotent stem cells following exposure to glucose-induced hyperosmolarity. Circulation AOS.701.01 (Abstract Oral Session).
****Madonna, R., Shelat, H., De Caterina, R., and Geng, Y. J. (2011b). Vascularization in human induced pluripotent stem cells under hyperosmolarity induced by high glucose. J Am Coll Cardiol (abstr).
Madonna, R., Shelath, H., & Geng, Y. J. (2010). Aquaporin-associated vascular development of human induced pluripotent stem cells stimulated with high levels of glucose. Circulation, 122, A21260.
Miyoshi, N., Ishii, H., Nagano, H., Haraguchi, N., Dewi, D. L., Kano, Y., et al. (2011). Reprogramming of mouse and human cells to pluripotency using mature microRNAs. Cell Stem Cell, 8, 633–638.
Moretti, A., Bellin, M., Welling, A., Jung, C. B., Lam, J. T., Bott-Flugel, L., et al. (2010). Patient-specific induced pluripotent stem-cell models for long-QT syndrome. New England Journal of Medicine, 363, 1397–1409.
Nakajima, T., Furukawa, T., Tanaka, T., Katayama, Y., Nagai, R., Nakamura, Y., et al. (1998). Novel mechanism of HERG current suppression in LQT2: Shift in voltage dependence of HERG inactivation. Circulation Research, 83, 415–422.
Nobelprize.org. (2007). The nobel prize in physiology or medicine 2007.
Okita, K., Hong, H., Takahashi, K., & Yamanaka, S. (2010). Generation of mouse-induced pluripotent stem cells with plasmid vectors. Nature Protocols, 5, 418–428.
Pabo, C. O., Peisach, E., & Grant, R. A. (2001). Design and selection of novel Cys2His2 zinc finger proteins. Annual Review of Biochemistry, 70, 313–340.
Pang, Z. P., Yang, N., Vierbuchen, T., Ostermeier, A., Fuentes, D. R., Yang, T. Q., et al. (2011). Induction of human neuronal cells by defined transcription factors. Nature, 476, 220–223.
Park, I. H., Arora, N., Huo, H., Maherali, N., Ahfeldt, T., Shimamura, A., et al. (2008). Disease-specific induced pluripotent stem cells. Cell, 134, 877–886.
Pfisterer, U., Kirkeby, A., Torper, O., Wood, J., Nelander, J., Dufour, A., et al. (2011). Direct conversion of human fibroblasts to dopaminergic neurons. Proceedings of the National Academy of Sciences of the United States of America, 108, 10343–10348.
Prior, T. W. (2007). Spinal muscular atrophy diagnostics. Journal of Child Neurology, 22, 952–956.
Rao, M., & Condic, M. L. (2008). Alternative sources of pluripotent stem cells: Scientific solutions to an ethical dilemma. Stem Cells and Development, 17, 1–10.
Raya, A., Rodriguez-Piza, I., Guenechea, G., Vassena, R., Navarro, S., Barrero, M. J., et al. (2009). Disease-corrected haematopoietic progenitors from Fanconi anaemia induced pluripotent stem cells. Nature, 460, 53–59.
Rosenthal, N., & Brown, S. (2007). The mouse ascending: perspectives for human-disease models. Nature Cell Biology, 9, 993–999.
Sartipy, P., Bjorquist, P., Strehl, R., & Hyllner, J. (2007). The application of human embryonic stem cell technologies to drug discovery. Drug Discovery Today, 12, 688–699.
Sinkkonen, L., Hugenschmidt, T., Berninger, P., Gaidatzis, D., Mohn, F., Artus-Revel, C. G., et al. (2008). MicroRNAs control de novo DNA methylation through regulation of transcriptional repressors in mouse embryonic stem cells. Nature Structural & Molecular Biology, 15, 259–267.
Soldner, F., Hockemeyer, D., Beard, C., Gao, Q., Bell, G. W., Cook, E. G., et al. (2009). Parkinson’s disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell, 136, 964–977.
Somers, A., Jean, J. C., Sommer, C. A., Omari, A., Ford, C. C., Mills, J. A., et al. (2010). Generation of transgene-free lung disease-specific human induced pluripotent stem cells using a single excisable lentiviral stem cell cassette. Stem Cells, 28, 1728–1740.
Stadtfeld, M., Nagaya, M., Utikal, J., Weir, G., & Hochedlinger, K. (2008). Induced pluripotent stem cells generated without viral integration. Science, 322, 945–949.
Stewart, R., Yang, C., Anyfantis, G., Przyborski, S., Hole, N., Strachan, T., et al. (2008). Silencing of the expression of pluripotent driven-reporter genes stably transfected into human pluripotent cells. Regenerative Medicine, 3, 505–522.
Sul, J. Y., Wu, C. W., Zeng, F., Jochems, J., Lee, M. T., Kim, T. K., et al. (2009). Transcriptome transfer produces a predictable cellular phenotype. Proceedings of the National Academy of Sciences of the United States of America, 106, 7624–7629.
Szabo, E., Rampalli, S., Risueno, R. M., Schnerch, A., Mitchell, R., Fiebig-Comyn, A., et al. (2010). Direct conversion of human fibroblasts to multilineage blood progenitors. Nature, 468, 521–526.
Takahashi, K., & Yamanaka, S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126, 663–676.
Tanaka, T., Tohyama, S., Murata, M., Nomura, F., Kaneko, T., Chen, H., et al. (2009). In vitro pharmacologic testing using human induced pluripotent stem cell-derived cardiomyocytes. Biochemical and Biophysical Research Communications, 385, 497–502.
Tay, Y., Zhang, J., Thomson, A. M., Lim, B., & Rigoutsos, I. (2008). MicroRNAs to Nanog, Oct4 and Sox2 coding regions modulate embryonic stem cell differentiation. Nature, 455, 1124–1128.
Thomas, K. R., Folger, K. R., & Capecchi, M. R. (1986). High frequency targeting of genes to specific sites in the mammalian genome. Cell, 44, 419–428.
Vierbuchen, T., Ostermeier, A., Pang, Z. P., Kokubu, Y., Sudhof, T. C., & Wernig, M. (2011). Direct conversion of fibroblasts to functional neurons by defined factors. Nature, 463, 1035–1041.
Warren, L., Manos, P. D., Ahfeldt, T., Loh, Y. H., Li, H., Lau, F., et al. (2010). Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell, 7, 618–630.
Wellner, U., Schubert, J., Burk, U. C., Schmalhofer, O., Zhu, F., Sonntag, A., et al. (2009). The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nature Cell Biology, 11, 1487–1495.
Wernig, M., Zhao, J. P., Pruszak, J., Hedlund, E., Fu, D., Soldner, F., et al. (2008). Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson’s disease. Proceedings of the National Academy of Sciences of the United States of America, 105, 5856–5861.
Woltjen, K., Michael, I. P., Mohseni, P., Desai, R., Mileikovsky, M., Hamalainen, R., et al. (2009). piggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature, 458, 766–770.
Xie, H., Ye, M., Feng, R., & Graf, T. (2004). Stepwise reprogramming of B cells into macrophages. Cell, 117, 663–676.
Xu, N., Papagiannakopoulos, T., Pan, G., Thomson, J. A., & Kosik, K. S. (2009). MicroRNA-145 regulates OCT4, SOX2, and KLF4 and represses pluripotency in human embryonic stem cells. Cell, 137, 647–658.
Ye, Z., Zhan, H., Mali, P., Dowey, S., Williams, D. M., Jang, Y. Y., et al. (2009). Human-induced pluripotent stem cells from blood cells of healthy donors and patients with acquired blood disorders. Blood, 114, 5473–5480.
Yoshizaki, S., Nishi, M., Kondo, A., Kojima, Y., Yamamoto, N., & Ryo, A. (2010). Vaccination with human induced pluripotent stem cells creates an antigen-specific immune response against HIV-1 gp160. Frontiers in Microbiology, 2, 27.
Yu, J., Hu, K., Smuga-Otto, K., Tian, S., Stewart, R., Slukvin, I. I., et al. (2009). Human induced pluripotent stem cells free of vector and transgene sequences. Science, 324, 797–801.
Yu, J., Vodyanik, M. A., Smuga-Otto, K., Antosiewicz-Bourget, J., Frane, J. L., Tian, S., et al. (2007). Induced pluripotent stem cell lines derived from human somatic cells. Science, 318, 1917–1920.
Zhao, T., Zhang, Z. N., Rong, Z., & Xu, Y. (2011). Immunogenicity of induced pluripotent stem cells. Nature, 474, 212–215.
Zou, J., Maeder, M. L., Mali, P., Pruett-Miller, S. M., Thibodeau-Beganny, S., Chou, B. K., et al. (2009). Gene targeting of a disease-related gene in human induced pluripotent stem and embryonic stem cells. Cell Stem Cell, 5, 97–110.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Madonna, R. Human-Induced Pluripotent Stem Cells: In Quest of Clinical Applications. Mol Biotechnol 52, 193–203 (2012). https://doi.org/10.1007/s12033-012-9504-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-012-9504-0