Abstract
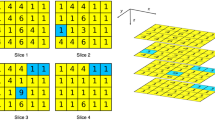
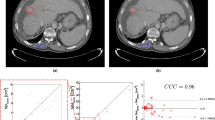
Texture analysis (TA) can provide quantitative features from medical imaging that can be correlated to clinical endpoints. The challenges relevant to robustness of radiomics features have been analyzed by many researchers, as it seems to be influenced by acquisition and reconstruction protocols. Delta-texture analysis (D-TA), conversely, consist in the analysis of TA feature variations at different acquisition times, usually before and after a therapy. Aim of this study was to investigate the influence of different CT scanners and acquisition parameters in the robustness of TA and D-TA. We scanned a commercial phantom (CIRS model 467, Gammex, Middleton, WI, USA), that is used for the calibration of electron density, two times by varying the disposition of plugs, using three different scanners. After the segmentation, we extracted TA features with LifeX and calculated TA features and D-TA features, defined as the variation of each TA parameters extracted from the same position by varying the plugs with the formula (Y–X)/X. The robustness of TA and D-TA features were then tested with intraclass coefficient correlation (ICC) analysis. The reliability of TA parameters across different scans, with different acquisition parameters and ROI positions has shown poor reliability in 12/37 and moderate reliability in the remaining 25/37, with no parameters showing good reliability. The reliability of D-TA, conversely, showed poor reliability in 10/37 parameters, moderate reliability in 10/37 parameters, and good reliability in 17/37 parameters. The comparison between TA and D-TA ICCs showed a significant difference for the whole group of parameters (p:0.004) and for the subclasses of GLCM parameters (p:0.033), whereas for the other subclasses of matrices (GLRLM, NGLDM, GLZLM, Histogram), the difference was not significant. D-TA features seem to be more robust than TA features. These findings reinforce the potentiality for using D-TA features for early assessment of treatment response and for developing tailored therapies. More work is needed in a clinical setting to confirm the results of the present study.



Similar content being viewed by others
References
Ganeshan B, Panayiotou E, Burnand K, Dizdarevic S, Miles K. Tumour heterogeneity in non-small cell lung carcinoma assessed by CT texture analysis: a potential marker of survival. Eur Radiol. 2012;22(4):796–802.
Ganeshan B, Abaleke S, Young RC, Chatwin CR, Miles KA. Texture analysis of non-small cell lung cancer on unenhanced computed tomography: initial evidence for a relationship with tumour glucose metabolism and stage. Cancer Imaging. 2010;10:137–43.
Ganeshan B, Miles KA, Young RC, Chatwin CR. Hepatic enhancement in colorectal cancer: texture analysis correlates with hepatic hemodynamics and patient survival. Acad Radiol. 2007;14(12):1520–30.
Nardone V, Reginelli A, Scala F, Carbone SF, Mazzei MA, Sebaste L, et al. Magnetic-resonance-imaging texture analysis predicts early progression in rectal cancer patients undergoing neoadjuvant chemoradiation. Gastroenterol Res Pract. 2019;2019:8505798.
Russo U, Sabatino V, Nizzoli R, Tiseo M, Cappabianca S, Reginelli A, et al. Transthoracic computed tomography-guided lung biopsy in the new era of personalized medicine. Future Oncol. 2019;15(10):1125–34.
Bianconi F, Palumbo I, Fravolini ML, Chiari R, Minestrini M, Brunese L, et al. Texture analysis on [(18)F]FDG PET/CT in non-small-cell lung cancer: correlations between PET features, CT features, and histological types. Mol Imaging Biol. 2019;21(6):1200–9.
Alobaidli S, McQuaid S, South C, Prakash V, Evans P, Nisbet A. The role of texture analysis in imaging as an outcome predictor and potential tool in radiotherapy treatment planning. Br J Radiol. 2014;87(1042):20140369.
Mattonen SA, Tetar S, Palma DA, Louie AV, Senan S, Ward AD. Imaging texture analysis for automated prediction of lung cancer recurrence after stereotactic radiotherapy. J Med Imaging. 2015;2(4):041010.
Mattonen SA, Palma DA, Haasbeek CJ, Senan S, Ward AD. Early prediction of tumor recurrence based on CT texture changes after stereotactic ablative radiotherapy (SABR) for lung cancer. Med Phys. 2014;41(3):033502.
Coroller TP, Agrawal V, Narayan V, Hou Y, Grossmann P, Lee SW, et al. Radiomic phenotype features predict pathological response in non-small cell lung cancer. Radiother Oncol. 2016;119(3):480–6.
Nardone V, Tini P, Carbone SF, Grassi A, Biondi M, Sebaste L, et al. Bone texture analysis using CT-simulation scans to individuate risk parameters for radiation-induced insufficiency fractures. Osteoporos Int. 2017;28(6):1915–23.
Nardone V, Tini P, Nioche C, Biondi M, Sebaste L, Mazzei MA, et al. Texture analysis of parotid gland as a predictive factor of radiation induced xerostomia: a subset analysis. Radiother Oncol. 2016;122:321.
Nardone V, Tini P, Croci S, Carbone SF, Sebaste L, Carfagno T, et al. 3D bone texture analysis as a potential predictor of radiation-induced insufficiency fractures. Quant Imaging Med Surg. 2018;8(1):14–24.
Nardone V, Tini P, Nioche C, Mazzei MA, Carfagno T, Battaglia G, et al. Texture analysis as a predictor of radiation-induced xerostomia in head and neck patients undergoing IMRT. Radiol Med (Torino). 2018;123(6):415–23.
Nardone V, Reginelli A, Scala F. Magnetic-resonance-imaging texture analysis predicts early progression in rectal cancer patients undergoing neoadjuvant chemoradiation. Gastroenterol Res Pract. 2019;2019:8505798.
Dinapoli N, Barbaro B, Gatta R, Chiloiro G, Casa C, Masciocchi C, et al. Magnetic resonance, vendor-independent, intensity histogram analysis predicting pathologic complete response after radiochemotherapy of rectal cancer. Int J Radiat Oncol Biol Phys. 2018;102(4):765–74.
Nardone V, Nanni S, Pastina P, Vinciguerra C, Cerase A, Correale P, et al. Role of perilesional edema and tumor volume in the prognosis of non-small cell lung cancer (NSCLC) undergoing radiosurgery (SRS) for brain metastases. Strahlenther Onkol. 2019;195(8):734–44.
Yip SSF, Coroller TP, Sanford NN, Huynh E, Mamon H, Aerts HJWL, et al. Use of registration-based contour propagation in texture analysis for esophageal cancer pathologic response prediction. Phys Med Biol. 2016;61(2):906–22.
Balagurunathan Y, Kumar V, Gu Y, Kim J, Wang H, Liu Y, et al. Test–retest reproducibility analysis of lung CT image features. J Digit Imaging. 2014;27(6):805–23.
Avanzo M, Stancanello J, El Naqa I. Beyond imaging: the promise of radiomics. Physica Medica Eur J Med Phys. 2017;38:122–39.
Biondi M, Vanzi E, De Otto G, Carbone SF, Nardone V, Banci BF. Effects of CT FOV displacement and acquisition parameters variation on texture analysis features. Phys Med Biol. 2018;63(23):235021.
Reginelli A, Capasso R, Petrillo M, Rossi C, Faella P, Grassi R, et al. Looking for lepidic component inside invasive adenocarcinomas appearing as CT solid solitary pulmonary nodules (SPNs): CT morpho-densitometric features and 18-FDG PET findings. Biomed Res Int. 2019;2019:7683648.
Macchi M, Belfiore MP, Floridi C, Serra N, Belfiore G, Carmignani L, et al. Radiofrequency versus microwave ablation for treatment of the lung tumours: LUMIRA (lung microwave radiofrequency) randomized trial. Med Oncol. 2017;34(5):96.
Scialpi M, Cappabianca S, Rotondo A, Scalera GB, Barberini F, Cagini L, et al. Pulmonary congenital cystic disease in adults. Spiral computed tomography findings with pathologic correlation and management. Radiol Med. 2010;115(4):539–50.
Reginelli A, Vanzulli A, Sgrazzutti C, Caschera L, Serra N, Raucci A, et al. Vascular microinvasion from hepatocellular carcinoma: CT findings and pathologic correlation for the best therapeutic strategies. Med Oncol. 2017;34(5):93.
Bera K, Velcheti V, Madabhushi A. Novel quantitative imaging for predicting response to therapy: techniques and clinical applications. Am Soc Clin Oncol Educ Book. 2018;38:1008–18.
Lee G, Bak SH, Lee HY, Choi JY, Park H, Lee SH, et al. Measurement variability in treatment response determination for non-small cell lung cancer: improvements using radiomics. J Thorac Imaging. 2019;34(2):103–15.
Mazzei MA, Nardone V, Di Giacomo L, Bagnacci G, Gentili F, Tini P, et al. The role of delta radiomics in gastric cancer. Quant Imaging Med Surg. 2018;8(7):719–21.
van Dijk LV, Langendijk JA, Zhai TT, Vedelaar TA, Noordzij W, Steenbakkers R, et al. Delta-radiomics features during radiotherapy improve the prediction of late xerostomia. Sci Rep. 2019;9(1):12483.
Boldrini L, Cusumano D, Chiloiro G, Casa C, Masciocchi C, Lenkowicz J, et al. Delta radiomics for rectal cancer response prediction with hybrid 0.35 T magnetic resonance-guided radiotherapy (MRgRT): a hypothesis-generating study for an innovative personalized medicine approach. Radiol Med (Torino). 2019;124(2):145–53.
Reginelli A, Silvestro G, Fontanella G, Sangiovanni A, Conte M, Nuzzo I, et al. Validation of DWI in assessment of radiotreated bone metastases in elderly patients. Int J Surg. 2016;33(Suppl 1):S148–S153153.
Nioche C, Orlhac F, Boughdad S, Reuze S, Soussan M, Robert C, et al. A freeware for tumor heterogeneity characterization in PET, SPECT, CT, MRI and US to accelerate advances in radiomics. J Nucl Med. 2017;58(supplement 1):1316.
Watson PF, Petrie A. Method agreement analysis: a review of correct methodology. Theriogenology. 2010;73(9):1167–79.
Portney LG, Watkins MP. Foundations of clinical research: applications to practice. 2nd ed. Upper Saddle River, NJ: Prentice Hall Health; 2000.
Bamberg F. Quantitative imaging and imaging biomarkers: the search for generalizability in radiology. J Thorac Imaging. 2018;33(2):69–70.
Raunig DL, McShane LM, Pennello G, Gatsonis C, Carson PL, Voyvodic JT, et al. Quantitative imaging biomarkers: a review of statistical methods for technical performance assessment. Stat Methods Med Res. 2015;24(1):27–67.
Zhao B, Tan Y, Tsai WY, Qi J, Xie C, Lu L, et al. Reproducibility of radiomics for deciphering tumor phenotype with imaging. Sci Rep. 2016;6:23428.
Ganeshan B, Miles KA. Quantifying tumour heterogeneity with CT. Cancer Imaging. 2013;13(1):140–9.
Shafiq-Ul-Hassan M, Zhang GG, Latifi K, Ullah G, Hunt DC, Balagurunathan Y, et al. Intrinsic dependencies of CT radiomic features on voxel size and number of gray levels. Med Phys. 2017;44(3):1050–62.
Larue R, van Timmeren JE. Influence of gray level discretization on radiomic feature stability for different CT scanners, tube currents and slice thicknesses: a comprehensive phantom study. Acta Oncol. 2017;56(11):1544–53.
Mackin D, Fave X, Zhang L, Fried D, Yang J, Taylor B, et al. Measuring computed tomography scanner variability of radiomics features. Investig Radiol. 2015;50(11):757–65.
Fave X, Mackin D, Yang J, Zhang J, Fried D, Balter P, et al. Can radiomics features be reproducibly measured from CBCT images for patients with non-small cell lung cancer? Med Phys. 2015;42(12):6784–97.
Plautz TE, Zheng C, Noid G, Li XA. Time stability of delta-radiomics features and the impact on patient analysis in longitudinal CT images. Med Phys. 2019;46(4):1663–766.
Acknowledgements
Thanks to the Research Program “Valere” supported by University of Campania “L.Vavitelli”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nardone, V., Reginelli, A., Guida, C. et al. Delta-radiomics increases multicentre reproducibility: a phantom study. Med Oncol 37, 38 (2020). https://doi.org/10.1007/s12032-020-01359-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-020-01359-9