Abstract
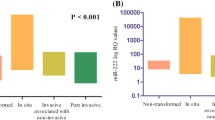
MicroRNAs play essential role in breast carcinoma progression and invasion. Our principal goals were to assess clinicopathological and prognostic correlations of microRNA-21 (miR-21) expression levels in a group of 39 Serbian breast cancer patients with invasive lobular (ILC), ductal (IDC), or mixed (ILC–IDC) breast carcinomas and in order to discover the role of miR-21 in potential novel form of stratification of the patients with different estrogen receptor (ER) and progesterone receptor (PR) status. MiR-21 expression levels were measured by stem-loop real-time RT-PCR using TaqMan technology. ER, PR, human epidermal growth factor 2 receptor (Her-2), and proliferative index (Ki-67) were evaluated by immunohistochemistry. MiR-21 levels do not vary among ILC, IDC, and ILC–IDC subgroups. MiR-21 expression levels varied significantly in the age, tumor size, Ki-67, and different grade (p = 0.030, p = 0.036, p = 0.027 and p = 0.032, respectively) subgroups. ER+ and PR+ showed higher miR-21 levels than their negative receptor status paired groups ER− and PR− with p = 0.012 and p = 0.018, respectively. MiR-21 positively correlated with ER and PR status (p = 0.018, ρ = 0.379 and p = 0.034, ρ = 0.345, respectively). Our findings suggest that miR-21 emulates transitional form of expression and that the levels of expression might be useful for stratification of the patients with different receptor status with the purpose to seek for new therapy approaches especially for the patients with the lack of response to conventional endocrine therapy.



Similar content being viewed by others
References
Pan X, Wang Z-X, Wang R. MicroRNA-21: a novel therapeutic target in human cancer. Cancer Biol Ther. 2010;10(12):1224–32. doi:10.4161/cbt.10.12.14252.
Shenouda SK, Alahari SK. MicroRNA function in cancer: oncogene or a tumor suppressor? Cancer Metast Rev. 2009;28(3–4):369–78. doi:10.1007/s10555-009-9188-5.
Huang G-L, Zhang X-H, Guo G-L, Huang K-T, Yang K-Y, Shen X, You J, Hu X-Q. Clinical significance of miR-21 expression in breast cancer: SYBR-Green I-based real-time RT-PCR study of invasive ductal carcinoma. Oncol Rep. 2009. doi:10.3892/or_00000270.
Song B, Wang C, Liu J, Wang X, Lv L, Wei L, Xie L, Zheng Y, Song X. MicroRNA-21 regulates breast cancer invasion partly by targeting tissue inhibitor of metalloproteinase 3 expression. J Exp Clin Cancer Res. 2010;29(1):29. doi:10.1186/1756-9966-29-29.
Lee JA, Lee HY, Lee ES, Kim I, Bae JW. Prognostic implications of MicroRNA-21 overexpression in invasive ductal carcinomas of the breast. Int J Breast Cancer. 2011;14(4):269–75. doi:10.4048/jbc.2011.14.4.269.
Qi L, Bart J, Tan L, Platteel I, Sluis T, Huitema S, Harms G, Fu L, Hollema H, Berg A. Expression of miR-21 and its targets (PTEN, PDCD4, TM1) in flat epithelial atypia of the breast in relation to ductal carcinoma in situ and invasive carcinoma. BMC Cancer. 2009;9(1):163. doi:10.1186/1471-2407-9-163.
Yoshinaga H, Matsuhashi S, Fujiyama C, Masaki Z. Novel human PDCD4 (H731) gene expressed in proliferative cells is expressed in the small duct epithelial cells of the breast as revealed by an anti-H731 antibody. Pathol Int. 1999;49:1067–77. doi:10.1186/bcr1285.
Yang Y, Chaerkady R, Beer MA, Mendell JT, Pandey A. Identification of miR-21 targets in breast cancer cells using a quantitative proteomic approach. Proteomics. 2009;9(5):1374–84. doi:10.1002/pmic.200800551.
Span PN, Lindberg RLP, Manders P, Tjan-Heijnen VCG, Heuvel JJTM, Beex LVAM, et al. Tissue inhibitors of metalloproteinase expression in human breast cancer: TIMP-3 is associated with adjuvant endocrine therapy success. J Pathol. 2004;202(4):395–402. doi:10.1002/path.1528.
Mylona E, Magkou C, Giannopoulou I, Agrogiannis G, Markaki S, Keramopoulos A, Nakopoulou L. Expression of tissue inhibitor of matrix metalloproteinases (TIMP)-3 protein in invasive breast carcinoma: Relation to tumor phenotype and clinical outcome. Breast Cancer Res. 2006;8(5):R57. doi:10.1186/bcr1607.
Li J, Zhang Y, Zhang W, Jia S, Tian R, Kang Y, Ma Y, Li D. Genetic heterogeneity of breast cancer metastasis may be related to miR-21 regulation of TIMP-3 in translation. Int J Sur Oncol. 2013;2013:1–7. doi:10.1155/2013/875078.
Zhu S, Si ML, Wu H, Mo YY. MicroRNA-21 targets the tumor suppressor gene tropomyosin 1 (TPM1). J Biol Chem. 2007;282(19):14328–36. doi:10.1074/jbc.M611393200.
Lopez-Camarillo C, Fonseca-Sánchez MA, Flores-Pérez A, Marchat LA, Arechaga-Ocampo E, Azuara-Liceaga E, et al. Functional roles of microRNAs in cancer: microRNomes and oncomiRs connection. In: Lopez-Camarillo C, Marchat LA, Arechaga-Ocampo E, Azuara-Liceaga E, Perez Plasencia C, Fuentes-Mera L, et al., editors. Oncogenomics and cancer proteomics—novel approaches in biomarkers discovery and therapeutic targets in cancer. InTech, p. 72–89, 2013; 9(1):53–9. doi:10.1111/j.1743-7563.2012.01548.x.
Rask L, Balslev EVA, JØRgensen S, Eriksen J, Flyger H, MØLler S, HØGdall E, Litman T, Schnack Nielsen B. High expression of miR-21 in tumor stroma correlates with increased cancer cell proliferation in human breast cancer. Apmis. 2011;119(10):663–73. doi:10.1111/j.1600-0463.2011.02782.x.
Esquela-Kerscher A, Slack FJ. Oncomirs—microRNAs with a role in cancer. Nat Rev Cancer. 2006;6(4):259–69. doi:10.1038/nrc1840.
Schwarz DS, Hutvagner G, Du T, Xu Z, Aronin N, Zamore PD. Asymmetry in the assembly of the RNAi enzyme complex. Cell. 2003;115(2):199–208.
Lowery AJ, Miller N, Devaney A, McNeill RE, Davoren PA, Lemetre C, Benes V, Schmidt S, Blake J, Ball G, Kerin MJ. MicroRNA signatures predict oestrogen receptor, progesterone receptor and HER2/neu receptor status in breast cancer. Breast Cancer Res. 2009;11(3):R27. doi:10.1186/bcr2257.
Verkooijen HM, Fioretta Gr, Vlastos G, Morabia A, Schubert H, Sappino A-P, et al. Important increase of invasive lobular breast cancer incidence in Geneva, Switzerland. Int Cancer. 2003;104(6):778–81. doi:10.1002/ijc.11032.
Petrović N, Mandušić V, Stanojević B, Lukić S, Todorović L, Roganović J, et al. The difference in miR-21 expression levels between invasive and non-invasive breast cancers emphasizes its role in breast cancer invasion. Med Oncol. 2014;31(3):867. doi:10.1007/s12032-014-0867-x.
Harrell JC, Dye WW, Harvell DM, Pinto M, Jedlicka P, Sartorius CA, Horwitz KB. Estrogen insensitivity in a model of estrogen receptor positive breast cancer lymph node metastasis. Cancer Res. 2007;67(21):10582–91.
Yamashita H, Nishio M, Kobayashi S, Ando Y, Sugiura H, Zhang Z, Hamaguchi M, Mita K, Fujii Y, Iwase H. Phosphorylation of estrogen receptor α serine 167 is predictive of response to endocrine therapy and increases postrelapse survival in metastatic breast cancer. Breast Cancer Res. 2005;7(5):R753. doi:10.1186/bcr1285.
Ng CH, Pathy NB, Taib NA, Mun KS, Rhodes A, Yip CH. The estrogen receptor negative-progesterone receptor positive breast carcinoma is a biological entity and not a technical artifact. Asian Pac J Cancer Prev. 2012;13(4):1111–3. doi:10.7314/apjcp.2012.13.4.1111.
Leake R. Immunohistochemical detection of steroid receptors in breast cancer: a working protocol. J Clin Pathol. 2000;53(8):634–5. doi:10.1136/jcp.53.8.634.
Sauter G, Lee J, Bartlett JMS, Slamon DJ, Press MF. Guidelines for human epidermal growth factor receptor 2 testing: biologic and methodologic considerations. J Clin Oncol. 2009;27(8):1323–33. doi:10.1200/jco.2007.14.8197.
Kim YJ, Hwang SJ, Bae YC, Jung JS. miR-21 regulates adipogenic differentiation through the modulation of TGF-β signaling in mesenchymal stem cells derived from human adipose tissue. Stem Cells. 2009;27(12):3093–102. doi:10.1002/stem.235.
Qian B, Katsaros D, Lu L, Preti M, Durando A, Arisio R, Mu L, Yu H. High miR-21 expression in breast cancer associated with poor disease-free survival in early stage disease and high TGF-β1. Breast Cancer Res Treat. 2008;117(1):131–40. doi:10.1007/s10549-008-0219-7.
Wickramasinghe NS, Manavalan TT, Dougherty SM, Riggs KA, Li Y, Klinge CM. Estradiol downregulates miR-21 expression and increases miR-21 target gene expression in MCF-7 breast cancer cells. Nucl Acids Res. 2009;37(8):2584–95. doi:10.1093/nar/gkp117.
Iorio MV, Ferracin M, Liu CG, Veronese A, Spizzo R, Sabbioni S, Magri E, Pedriali M, Fabbri M, Campiglio M, Menard S, Palazzo JP, Rosenberg A, Musiani P, Volinia S, Nenci I, Calin GA, Querzoli P, Negrini M, Croce CM. MicroRNA gene expression deregulation in human breast cancer. Cancer Res. 2005;65(16):7065–70. doi:10.1158/0008-5472.CAN-05-1783.
Huang TH, Wu F, Loeb GB, Hsu R, Heidersbach A, Brincat A, Horiuchi D, Lebbink RJ, Mo YY, Goga A, McManus MT. Up-regulation of miR-21 by HER2/neu signaling promotes cell invasion. J Biol Chem. 2009;284(27):18515–24. doi:10.1074/jbc.M109.006676.
Mattie MD, Benz CC, Bowers J, Sensinger K, Wong L, Scott GK, Fedele V, Ginzinger D, Getts R, Haqq C. Optimized high-throughput microRNA expression profiling provides novel biomarker assessment of clinical prostate and breast cancer biopsies. Mol Cancer. 2006;5(1):24. doi:10.1186/1476-4598-5-24.
Arpino G, Weiss H, Lee AV, Schiff R, De Placido S, Osborne CK, Elledge RM. Estrogen receptor-positive, progesterone receptor-negative breast cancer: association with growth factor receptor expression and tamoxifen resistance. J Nat Cancer Inst. 2005;97(17):1254–61. doi:10.1093/jnci/dji249.
Acknowledgments
This work was supported by the Ministry of Education and Science, Republic of Serbia, Grant ON173049 (Nina Petrović, Vesna Mandušić, Boban Stanojević, Lidija Todorović, Silvana Lukić, Bogomir Dimitrijević).
Conflict of interest
All the authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Petrović, N., Mandušić, V., Dimitrijević, B. et al. Higher miR-21 expression in invasive breast carcinomas is associated with positive estrogen and progesterone receptor status in patients from Serbia. Med Oncol 31, 977 (2014). https://doi.org/10.1007/s12032-014-0977-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0977-5