Abstract
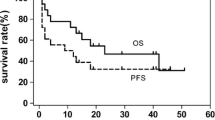
Optimal treatment strategies for localized extranodal natural killer/T cell lymphoma (ENKTL) have not been fully defined. We retrospectively compared the efficacy and safety of combined gemcitabine, l-asparaginase, and oxaliplatin (GELOX) (n = 38), continuous infusion of etoposide, vincristine and doxorubicin, with cyclophosphamide and prednisone (EPOCH) (n = 54), or combined cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) (n = 135) as induction chemotherapy in patients who were newly diagnosed with stage I/II ENKTL. After induction chemotherapy, the complete response (CR) rate and overall response rate (ORR) for the GELOX group were significantly higher than those in the EPOCH group (68.4 vs. 42.6 %, P = 0.011 for CR and 86.8 vs. 68.5 %, P = 0.038 for ORR). Both EPOCH and GELOX groups can attain much higher CR rates than CHOP group (CR rate was 31.8 %, P < 0.05). The 3-year overall survival (OS) and progression-free survival (PFS) rate were significantly better in GELOX group than in EPOCH or CHOP group (87.0 vs. 54.0 vs. 54.0 % for OS, P < 0.05; 72.0 vs. 50.0 vs. 43.0 % for PFS, P < 0.05). However, no significant differences were found between EPOCH and CHOP groups in OS or PFS (P = 0.765 for OS, and 0.421 for PFS). The safety profiles were acceptable in all three groups. In conclusion, GELOX is superior to EPOCH or CHOP in the treatment of patients with stage I/II ENKTL. Further clinical trials of ENKTL should use asparaginase-based regimens as the standard chemotherapy.

Similar content being viewed by others
References
Chan JK, Quintanilla-Martinez L, Ferry JA, Peh S-C. Extranodal NK/T-cell lymphoma, nasal type. In: Swerdlow SH, Campo E, Harris NL, et al., editors. WHO classification of tumours of haematopoietic and lymphoid tissues. Lyon: IARC; 2008. p. 285–8.
Vose J, Armitage J, Weisenburger D. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol. 2008;26:4124–30.
Li YX, Liu QF, Fang H, et al. Variable clinical presentations of nasal and Waldeyer ring natural killer/T-cell lymphoma. Clin Cancer Res. 2009;15(8):2905–12.
Huang MJ, Jiang Y, Liu WP, Li ZP, Li M, Zhou L, et al. Early or up-front radiotherapy improved survival of localized extranodal NK/T-cell lymphoma, nasal-type in the upper aerodigestive tract. Int J Radiat Oncol Biol Phys. 2008;70:166–74.
Kim GE, Cho JH, Yang WI, et al. Angiocentric lymphoma of the head and neck: patterns of systemic failure after radiation treatment. J Clin Oncol. 2000;18:54–63.
Wang B, Li XQ, Ma X, Hong X, Lu H, Guo Y. Immunohistochemical expression and clinical significance of P-glycoprotein in previously untreated extranodal NK/T-cell lymphoma, nasal type. Am J Hematol. 2008;83:795–9.
Lai G-M, Chen Y-N, Mickley LA, et al. P-glycoprotein expression and schedule dependence of adriamycin cytotoxicity in human colon carcinoma cell lines. Int J Cancer. 1991;49:696–703.
Huang H, Lin Z, Lin X, Cai Q, Xia Z, Jiang W. Long-term outcomes of patients with newly diagnosed extranodal natural killer/T-cell lymphoma treated by etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin regimen: a single-institution experience. Leuk Lymphoma. 2011;52:1041–8.
Yamaguchi M, Kwong YL, Kim WS, et al. Phase II study of SMILE chemotherapy for newly diagnosed stage IV, relapsed, or refractory extranodal natural killer (NK)/T-cell lymphoma, nasal type: the NK-Cell Tumor Study Group study. J Clin Oncol. 2011;29(33):4410–6.
Jaccard A, Gachard N, Marin B, et al. Efficacy of l-asparaginase with methotrexate and dexamethasone (AspaMetDex regimen) in patients with refractory or relapsing extranodal NK/T-cell lymphoma, a phase 2 study. Blood. 2011;117(6):1834–9.
Wang L, Wang ZH, Chen XQ, Li YJ, Wang KF, Xia YF, Xia ZJ. First-line combination of gemcitabine, oxaliplatin, and l-asparaginase (GELOX) followed by involved-field radiation therapy for patients with stage IE/IIE extranodal natural killer/T-cell lymphoma. Cancer. 2013;119:348–55.
Wang L, Xia ZJ, Huang HQ, Lu Y, Zhang YJ. Cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) in the treatment of stage IE/IIE extranodal natural killer/T cell lymphoma, nasal type: 13-year follow-up in 135 patients. Int J Hematol. 2012;96(5):617–23.
El Gnaoui T, Dupuis J, Belhadj K, et al. Rituximab, gemcitabine and oxaliplatin: an effective salvage regimen for patients with relapsed or refractory B-cell lymphoma not candidates for high-dose therapy. Ann Oncol. 2007;18(8):1363–8.
Ando M, Sugimoto K, Kitoh T, et al. Selective apoptosis of natural killer-cell tumours by l-asparaginase. Br J Haematol. 2005;130(6):860–8.
Lin N, Song Y, Zheng W, Tu M, Xie Y, Wang X, et al. A prospective phase II study of l-asparaginase-CHOP plus radiation in newly diagnosed extranodal NK/T-cell lymphoma, nasal type. J Hematol Oncol. 2013;6(1):44.
Yamaguchi M, Tobinai K, Oguchi M, Ishizuka N, Kobayashi Y, Isobe Y, et al. Phase I/II study of concurrent chemoradiotherapy for localized nasal natural killer/T-cell lymphoma: Japan Clinical Oncology Group Study JCOG0211. J Clin Oncol. 2009;27(33):5594–600.
Acknowledgments
We thank all of the physicians at Sun Yat-sen University Cancer Center for allowing us to include their patients. We also appreciate the cooperation of all the pathologists at Sun Yat-sen University Cancer Center for their support. This work received grant support from Young Teachers’ Cultivation Project of Sun Yat-sen University (No. 12ykpy54) and National Natural Science Foundation of China (No. 81272620).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liang, W., Wei-da, W., Zhong-jun, X. et al. Combination of gemcitabine, l-asparaginase, and oxaliplatin (GELOX) is superior to EPOCH or CHOP in the treatment of patients with stage IE/IIE extranodal natural killer/T cell lymphoma: a retrospective study in a cohort of 227 patients with long-term follow-up. Med Oncol 31, 860 (2014). https://doi.org/10.1007/s12032-014-0860-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0860-4