Abstract
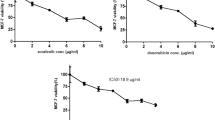
Accumulating evidences indicated that hypoxia-induced factors and COX-2 play a important role in tumorigenesis in various human cancer. Yet, the relationship between HIFs and COX-2 in human renal cancer remains unclear. The present study was to examine the role of HIFs and COX-2 in the invasiveness and the resistance to target agent in renal cancer cell line (786-O). In 786-O cells, hypoxia induced the increase in the protein expression of HIF1 and HIF2. We also demonstrate that hypoxia up-regulated the protein expression of COX-2 and Snail, but down-regulation of E-cadherin expression in 786-O cells promoted the invasiveness of 786-O cells and enhanced the resistance of 786-O cells to sorafenib. siRNA target to HIF1α, HIF2α and NS398, a selective inhibitor of COX-2, were used in this study. Only siRNA-HIF2α significantly suppressed the protein expression of HIF2 and COX-2, then decreased the invasive ability and resistance of 786-O cells to sorafenib under hypoxia. NS398 attenuated the increase in invasive cells number and the IC50 value of sorafenib induced by hypoxia. In conclusion, our results demonstrated that hypoxia promoted the invasiveness and resistance of 786-O cells to sorafenib via HIF2 and COX-2 and induced the activation of Snail/E-cadherin, suggesting that a signalling mechanism involving HIF2/COX2 modulates invasiveness and resistance to sorafenib in 786-O cells under hypoxia.






Similar content being viewed by others
References
McLaughlin JK, Lipworth L. Epidemiologic aspects of renal cell cancer. Semin Oncol. 2000;27(2):115–23.
Brian I, Rini SCC. Bernard escudier: renal cell carcinoma. Lancet. 2009;373:1119–32.
Poprach A, Pavlik T, Melichar B, Kubackova K, Bortlicek Z, Svoboda M, Lakomy R, Vyzula R, Kiss I, Dusek L, et al. Clinical and laboratory prognostic factors in patients with metastatic renal cell carcinoma treated with sunitinib and sorafenib after progression on cytokines. Urol Oncol. 2014;32(4):488–95.
Kubackova K, Bortlicek Z, Pavlik T, Melichar B, Linke Z, Pokorna P, Vyzula R, Prausova J, Buchler T, on behalf of the Czech Renal Cancer Cooperative G: Prognostic factors in renal cell carcinoma patients treated with sorafenib: results from the Czech registry. Target oncol. 2014. doi:10.1007/s11523-014-0343-8.
Wu H, Liang X, Fang Y, Qin X, Zhang Y, Liu J. Resveratrol inhibits hypoxia-induced metastasis potential enhancement by restricting hypoxia-induced factor-1 alpha expression in colon carcinoma cells. Biomed Pharmacother. 2008;62(9):613–21.
Fong GH, Takeda K. Role and regulation of prolyl hydroxylase domain proteins. Cell Death Differ. 2008;15(4):635–41.
Lisy K, Peet DJ. Turn me on: regulating HIF transcriptional activity. Cell Death Differ. 2008;15(4):642–9.
Bertout JA, Patel SA, Simon MC. The impact of O2 availability on human cancer. Nat Rev Cancer. 2008;8(12):967–75.
Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3(10):721–32.
Tian H, McKnight SL, Russell DW. Endothelial PAS domain protein 1 (EPAS1), a transcription factor selectively expressed in endothelial cells. Genes Dev. 1997;11(1):72–82.
Wiesener MS, Turley H, Allen WE, Willam C, Eckardt KU, Talks KL, Wood SM, Gatter KC, Harris AL, Pugh CW, et al. Induction of endothelial PAS domain protein-1 by hypoxia: characterization and comparison with hypoxia-inducible factor-1alpha. Blood. 1998;92(7):2260–8.
Raval RR, Lau KW, Tran MG, Sowter HM, Mandriota SJ, Li JL, Pugh CW, Maxwell PH, Harris AL, Ratcliffe PJ. Contrasting properties of hypoxia-inducible factor 1 (HIF-1) and HIF-2 in von Hippel–Lindau-associated renal cell carcinoma. Mol Cell Biol. 2005;25(13):5675–86.
Holmes MD, Chen WY, Schnitt SJ, Collins L, Colditz GA, Hankinson SE, Tamimi RM. COX-2 expression predicts worse breast cancer prognosis and does not modify the association with aspirin. Breast Cancer Res Treat. 2011;130(2):657–62.
Ji B, Liu Y, Zhang P, Wang Y, Wang G. COX-2 expression and tumor angiogenesis in thyroid carcinoma patients among northeast Chinese population-result of a single-center study. Int J Med Sci. 2012;9(3):237–42.
Teichert-Kuliszewska K, Kutryk MJ, Kuliszewski MA, Karoubi G, Courtman DW, Zucco L, Granton J, Stewart DJ. Bone morphogenetic protein receptor-2 signaling promotes pulmonary arterial endothelial cell survival: implications for loss-of-function mutations in the pathogenesis of pulmonary hypertension. Circ Res. 2006;98(2):209–17.
Franovic A, Gunaratnam L, Smith K, Robert I, Patten D, Lee S. Translational up-regulation of the EGFR by tumor hypoxia provides a nonmutational explanation for its overexpression in human cancer. Proc Natl Acad Sci USA. 2007;104(32):13092–7.
Charles C.Wykoff CP: The HIF pathway: implications for patterns of gene expression in cancer. The tumour microenvironment: causes and consequences of hypoxia and acidity Wiley, Chichester (NovartisFoundation Symposium 240) 2001.p. 212–231.
Kirkpatrick K, Ogunkolade W, Elkak A, Bustin S, Jenkins P, Ghilchik M, Mokbel K. The mRNA expression of cyclo-oxygenase-2 (COX-2) and vascular endothelial growth factor (VEGF) in human breast cancer. Curr Med Res Opin. 2002;18(4):237–41.
Cunha LL, Marcello MA, Nonogaki S, Morari EC, Soares FA, Vassallo J, Ward LS. CD8+ tumour-infiltrating lymphocytes and COX2 expression may predict relapse in differentiated thyroid cancer. Clin endocrinol. 2014:1–8. doi:10.1111/cen.12586.
Sena JA, Wang L, Hu CJ. BRG1 and BRM chromatin-remodeling complexes regulate the hypoxia response by acting as coactivators for a subset of hypoxia-inducible transcription factor target genes. Mol Cell Biol. 2013;33(19):3849–63.
Jorge L. Ruas LPaTP: role of CBP in regulating HIF-1-mediated activation of transcription. J Cell Sci. 2004;118:301–11.
Ratcliffe PJ. HIF-1 and HIF-2: working alone or together in hypoxia? J Clin Invest. 2007;117(4):862–5.
Kondo K, Klco J, Nakamura E, Lechpammer M, Kaelin WG Jr. Inhibition of HIF is necessary for tumor suppression by the von Hippel–Lindau protein. Cancer Cell. 2002;1:237–46.
Kondo K, Lechpammer M, Kaelin WG Jr. Inhibition of HIF2a is sufficient to suppress pVHL-defective tumor growth. PLoS Biol. 2003;1(3):439–44.
Daneau G, Boidot R, Martinive P, Feron O. Identification of cyclooxygenase-2 as a major actor of the transcriptomic adaptation of endothelial and tumor cells to cyclic hypoxia: effect on angiogenesis and metastases. Clin Cancer Res. 2010;16(2):410–9.
Shono T, Tofilon PJ, Bruner JM, Owolabi O, Lang FF. Cyclooxygenase-2 expression in human gliomas: prognostic significance and molecular correlations. Cancer Res. 2001;61(11):4375–81.
Harris RE, Casto BC, Harris ZM. Cyclooxygenase-2 and the inflammogenesis of breast cancer. World J Clin Oncol. 2014;5(4):677–92.
Koki AT LK, Harmon JM, Masferrer JL.: In: RE Harris, ed. Cyclooxygenase-2 (cox-2) and the inflammogenesis of cancer. Subcel biochem. 2007;42:93–126.
Harris RE. Cyclooxygenase-2 (cox-2) blockade in the chemoprevention of cancers of the colon, breast, prostate, and lung. Inflammopharmacology. 2009;17(2):55–67.
van Roy F, Berx G. The cell–cell adhesion molecule E-cadherin. Cell Mol Life Sci. 2008;65(23):3756–88.
Kalluri R, Weinberg RA. The basics of epithelial–mesenchymal transition. J Clin Invest. 2009;119(6):1420–8.
Dong C, Wu Y, Yao J, Wang Y, Yu Y, Rychahou PG, Evers BM, Zhou BP. G9a interacts with Snail and is critical for Snail-mediated E-cadherin repression in human breast cancer. J Clin Invest. 2012;122(4):1469–86.
Hou Z, Peng H, Ayyanathan K, Yan K-P, Langer EM, Longmore GD, Rauscher FJ 3rd. The LIM protein AJUBA recruits protein arginine methyltransferase 5 to mediate SNAIL-dependent transcriptional repression. Mol Cell Biol. 2008;28(10):3198–207.
Peinado H, Ballestar E, Esteller M, Cano A. Snail mediates E-cadherin repression by the recruitment of the Sin3A/histone deacetylase 1 (HDAC1)/HDAC2 complex. Mol Cell Biol. 2004;24(1):306–19.
Lin Y, Wu Y, Li J, Dong C, Ye X, Chi Y-I, Evers BM, Zhou BP. The SNAG domain of Snail1 functions as a molecular hook for recruiting lysine-specific demethylase 1. EMBO J. 2010;29(11):1803–16.
Dong C, Wu Y, Wang Y, Wang C, Kang T, Rychahou PG, Chi YI, Evers BM, Zhou BP. Interaction with Suv39H1 is critical for Snail-mediated E-cadherin repression in breast cancer. Oncogene. 2013;32(11):1351–62.
Adamski J, Price A, Dive C, Makin G. Hypoxia-induced cytotoxic drug resistance in osteosarcoma is independent of HIF-1Alpha. PLoS One. 2013;8(6):e65304.
Roberts DL, Williams KJ, Cowen RL, Barathova M, Eustace AJ, Brittain-Dissont S, Tilby MJ, Pearson DG, Ottley CJ, Stratford IJ, et al. Contribution of HIF-1 and drug penetrance to oxaliplatin resistance in hypoxic colorectal cancer cells. Br J Cancer. 2009;101(8):1290–7.
Song X, Liu X, Chi W, Liu Y, Wei L, Wang X, Yu J. Hypoxia-induced resistance to cisplatin and doxorubicin in non-small cell lung cancer is inhibited by silencing of HIF-1alpha gene. Cancer Chemother Pharmacol. 2006;58(6):776–84.
Johansson A, Rudolfsson SH, Kilter S, Bergh A. Targeting castration-induced tumour hypoxia enhances the acute effects of castration therapy in a rat prostate cancer model. BJU Int. 2011;107(11):1818–24.
Cui J, Guo Y-H, Zhang H-Y, Jiang L-L, Ma J-Q, Wang W-J, Wang M-C, Yang C-C, Nan K-J, Song L-P. Cyclooxygenase-2 inhibitor is a robust enhancer of anticancer agents against hepatocellular carcinoma multicellular spheroids. Onco Targets Ther. 2014;7:353–63.
Zhang H, Li Z, Wang K. Combining sorafenib with celecoxib synergistically inhibits tumor growth of non-small cell lung cancer cells in vitro and in vivo. Oncol Rep. 2014;31(4):1954–60.
Garcia M, Velez R, Romagosa C, Majem B, Pedrola N, Olivan M, Rigau M, Guiu M, Gomis RR, Morote J, et al. Cyclooxygenase-2 inhibitor suppresses tumour progression of prostate cancer bone metastases in nude mice. BJU Int. 2014;113(5b):E164–77.
Bocca C, Levolella M, Autelli R, Motta M, Mosso L, Torchio B, Bozzo F, Cannito S, Paternostro C, Colombatto S, Parola M, Miglietta A. Expression of Cox-2 in human breast cancer cells as a critical determinant of epithelial-to mesenchymal transition and invasiveness. Expert Opin Ther Targets. 2013;18(2):121–35.
Jiang J, Tang Y-L, Liang X-H. EMT: a new vision of hypoxia promoting cancer progression. Cancer Biol Ther. 2011;11(8):714–23.
Tiwari N, Gheldof A, Tatari M, Christofori G. EMT as the ultimate survival mechanism of cancer cells. Semin Cancer Biol. 2012;22(3):194–207.
Cannito S, Novo E, di Bonzo LV, Busletta C, Colombatto S, Parola M. Epithelial–mesenchymal transition: from molecular mechanisms, redox regulation to implications in human health and disease. Antioxid Redox Signal. 2010;12(12):1383–430.
Ichihara S, Yamada Y, Kawai Y, Osawa T, Furuhashi K, Duan Z, Ichihara G. Roles of oxidative stress and Akt signaling in doxorubicin cardiotoxicity. Biochem Biophys Res Commun. 2007;359(1):27–33.
Calzada MJ, del Peso L. Hypoxia-inducible factors and cancer. Clin Transl Oncol. 2007;9(5):278–89.
Murphy M, Carlson JA, Keough MP, Claffey KP, Signoretti S, Loda M. Hypoxia regulation of the cell cycle in malignant melanoma: putative role for the cyclin-dependent kinase inhibitor p27. J Cutan Pathol. 2004;31(7):477–82.
Scholten DJ 2nd, Timmer CM, Peacock JD, Pelle DW, Williams BO, Steensma MR. Down regulation of wnt signaling mitigates hypoxia-induced chemoresistance in human osteosarcoma cells. PLoS One. 2014;9(10):e111431.
Wang X, Zhang L, O’Neill A, Bahamon B, Alsop DC, Mier JW, Goldberg SN, Signoretti S, Atkins MB, Wood CG, et al. Cox-2 inhibition enhances the activity of sunitinib in human renal cell carcinoma xenografts. Br J Cancer. 2013;108(2):319–26.
Hida T, Kozaki K, Muramatsu H, et al. Cyclooxygenase-2 inhibitor induces apoptosis and enhances cytotoxicity of various anticancer agents in non-small cell lung cancer cell lines. Clin Cancer Res. 2000;6:2006–11.
Acknowledgments
We thank the staff of Life Science Institute laboratory for helping technical assistance.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, CX., Luo, CL. & Wu, XH. Hypoxia promotes 786-O cells invasiveness and resistance to sorafenib via HIF-2α/COX-2. Med Oncol 32, 419 (2015). https://doi.org/10.1007/s12032-014-0419-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0419-4