Abstract
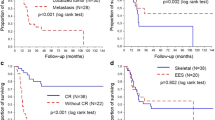
The aim of this study was to evaluate prognostic factors, survival rate and the efficacy of the treatment modalities used in patients with Ewing sarcoma family of tumors (ESFT). Data of patients with ESFTs followed up at different cancer centers in Turkey between 2001 and 2010 were retrospectively analyzed. The median age of 114 patients was 26 years (range 14–66). The median follow-up was 20 months (range 1–118 months). Tumor size was between 1.5 and 14 cm (median 8 cm). Eighty-six percent of patients had localized disease at presentation, and 14 % had metastatic disease. Local therapy was surgery alone in 31 % of patients, surgery combined with radiotherapy in 41 % and radiotherapy alone in 18 %. Approximately 70 % of patients were treated with vincristine, doxorubicin, cyclophosphamide and actinomycin-D, alternating with ifosfamide and etoposide every 3 weeks. In patients with localized disease at presentation, the 5-year disease-free survival and overall survival were 60 and 65 %, respectively. At univariate analysis, patients with tumor size ≥8 cm, high serum lactate dehydrogenase, metastasis at presentation, poor histological response to chemotherapy and positive surgical margin had significantly worse event-free survival. The significant predictors of worse overall survival at univariate analysis were tumor size ≤8 cm, high lactate dehydrogenase, metastasis at presentation, poor histological response to chemotherapy, radiotherapy only as local treatment and positive surgical margin. ESFTs are aggressive tumors with a high incidence of local recurrence and distant metastasis. Multimodality treatment consisting of adequate surgical resection, aggressive chemotherapy (VAC alternating with IE) and radiotherapy is recommended for patients with ESFTs.





Similar content being viewed by others
References
Cotterill SJ, Parker L, Malcolm AJ, et al. Incidence and survival for cancer in children and young adults in the North of England, 1968–1995: a report from the Northern Region Young Persons’ Malignant Disease Registry. Br J Cancer. 2000;83:397–403.
Aurias A, Rimbaut C, Buffe D, et al. Translocation involving chromosome 22 in Ewing’s sarcoma. A cytogenetic study of four fresh tumors. Cancer Genet Cytogenet. 1984;12:21–5.
Turc-Carel C, Philip I, Berger MP, et al. Chromosome study of Ewing’s sarcoma (ES) cell lines. Consistency of a reciprocal translocation t(11;22)(q24;q12). Cancer Genet Cytogenet. 1984;12:1–19.
Cangir A, Vietti TJ, Gehan EA, et al. Ewing’s sarcoma metastatic at diagnosis. Results and comparisons of two intergroup Ewing’s sarcoma studies. Cancer. 1990;66:887–93.
Bernstein ML, Devidas M, Lafreniere D, et al. Intensive therapy with growth factor support for patients with Ewing tumor metastatic at diagnosis: pediatric Oncology Group/Children’s Cancer Group Phase II Study 9457—a report from the Children’s Oncology Group. J Clin Oncol. 2006;24:152–9.
Leavey PJ, Collier AB. Ewing sarcoma: prognostic criteria, outcomes and future treatment. Expert Rev Anticancer Ther. 2008;8:617–24.
Bacci G, Ferrari S, Bertoni F, et al. Prognostic factors in nonmetastatic Ewing’s sarcoma of bone treated with adjuvant chemotherapy: analysis of 359 patients at the Istituto Ortopedico Rizzoli. J Clin Oncol. 2000;18:4–11.
Bacci G, Forni C, Longhi A, et al. Long-term outcome for patients with nonmetastatic Ewing’s sarcoma treated with adjuvant and neoadjuvant chemotherapies. 402 patients treated at Rizzoli between 1972 and 1992. Eur J Cancer. 2004;40:73–83.
Cotterill SJ, Ahrens S, Paulussen M, et al. Prognostic factors in Ewing’s tumor of bone: analysis of 975 patients from the European Intergroup Cooperative Ewing’s Sarcoma Study Group. J Clin Oncol. 2000;18:3108–14.
Paulussen M, Ahrens S, Craft AW, et al. Ewing’s tumors with primary lung metastases: survival analysis of 114 (European Intergroup) Cooperative Ewing’s Sarcoma Studies patients. J Clin Oncol. 1998;16:3044–52.
Pinkerton CR, Bataillard A, Guillo S, et al. Treatment strategies for metastatic Ewing’s sarcoma. Eur J Cancer. 2001;37:1338–44.
Schuck A, Ahrens S, Paulussen M, et al. Local therapy in localized Ewing tumors: results of 1058 patients treated in the CESS 81, CESS 86, and EICESS 92 trials. Int J Radiat Oncol Biol Phys. 2003;55:168–77.
Bernstein M, Kovar H, Paulussen M, et al. Ewing’s sarcoma family of tumors: current management. Oncologist. 2006;11:503–19.
Grier HE, Krailo MD, Tarbell NJ, et al. Addition of ifosfamide and etoposide to standard chemotherapy for Ewing’s sarcoma and primitive neuroectodermal tumor of bone. N Engl J Med. 2003;348:694–701.
Nesbit ME Jr, Gehan EA, Burgert EO Jr, et al. Multimodal therapy for the management of primary, nonmetastatic Ewing’s sarcoma of bone: a longterm follow-up of the First Intergroup study. J Clin Oncol. 1990;8:1664–74.
Paulussen M, Craft AW, Lewis I, et al. Results of the EICESS-92 Study: two randomized trials of Ewing’s sarcoma treatment—cyclophosphamide compared with ifosfamide in standard-risk patients and assessment of benefit of etoposide added to standard treatment in high-risk patients. J Clin Oncol. 2008;26:4385–93.
Ferrari S. Sundby Hall K, Luksch R et al. Nonmetastatic Ewing family tumors: high-dose chemotherapy with stem cell rescue in poor responder patients. Results of the Italian Sarcoma Group/Scandinavian Sarcoma Group III protocol. Ann Oncol. 2011;22(5):1221–7.
EURO-E.W.I.N.G 99 Treatment Manual. http://euro-ewing.klinikum.uni-muenster. de/(9 Nov 2009, date last accessed).
van der Woude HJ, Bloem JL, Hogendoorn PC. Preoperative evaluation and monitoring chemotherapy in patients with high-grade osteogenic and Ewing’s sarcoma: review of current imaging modalities. Skeletal Radiol. 1998;27:57–71.
van der Woude HJ, Bloem JL, Verstraete KL, et al. Osteosarcoma and Ewing’s sarcoma after neoadjuvant chemotherapy: value of dynamic MR imaging in detecting viable tumor before surgery. AJR Am J Roentgenol. 1995;165:593–8.
Shapeero LG, Vanel D. Imaging evaluation of the response of high-grade osteosarcoma and Ewing sarcoma to chemotherapy with emphasis on dynamic contrast-enhanced magnetic resonance imaging. Semin Musculoskelet Radiol. 2000;4:137–46.
Ladenstein R, Pötschger U, Le Deley MC, et al. Primary disseminated multifocal Ewing sarcoma: results of the Euro-EWING 99 trial. J Clin Oncol. 2010;28(20):3284–91.
Haeusler J, Ranft A, Boelling T, et al. The value of local treatment in patients with primary, disseminated, multifocal Ewing sarcoma (PDMES). Cancer. 2010;116(2):443–50.
Stahl M, Ranft A, Paulussen M, et al. Risk of recurrence and survival after relapse in patients with Ewing sarcoma. Pediatr Blood Cancer. 2011;57(4):549–53.
Ferrari S, del Prever AB, Palmerini E. Response to high-dose ifosfamide in patients with advanced/recurrent Ewing sarcoma. Pediatr Blood Cancer. 2009;52(5):581–4.
Wagner LM, McAllister N, Goldsby RE, et al. Temozolomide and intravenous irinotecan for treatment of advanced Ewing sarcoma. Pediatr Blood Cancer. 2007;48:132–9.
Hunold A, Weddeling N, Paulussen M, et al. Topotecan and cyclophosphamide in patients with refractory or relapsed Ewing tumors. Pediatr Blood Cancer. 2006;47:795–800.
Juergens H, Daw NC, Geoerger B, et al. Preliminary efficacy of the anti-insulin like growth factor type 1 receptor antibody figitumumab in patients with refractory Ewing sarcoma. J Clin Oncol. 2011;29(34):4534–40.
Pappo AS, Patel SR, Crowley J, et al. R1507, a monoclonal antibody to the insulin-like growth factor 1 receptor, in patients with recurrent or refractory Ewing sarcoma family of tumors: results of a phase II Sarcoma Alliance for Research through Collaboration study. J Clin Oncol. 2011;29(34):4541–7.
Siegel RD, Ryan LM, Antman KH. Adults with Ewing’s sarcoma: an analysis of 16 patients at the Dana-Farber Cancer Institute. Am J Clin Oncol. 1988;11:614–7.
Angervall L, Enzinger FM. Extraskeletal neoplasm resembling Ewing’s sarcoma. Cancer. 1975;36:240–51.
Smith MA, Gurney JG, Ries LA. Cancer in adolescents 15 to 19 years old. Cancer incidence and survival among children and adolescents: United States SEER Program 1975–1995. NIH Pub. No. 99-4649. Bethesda, MD, 1999.
Grier HE, Krailo Mark D. Addition of ifosfamide and etoposide to standard chemotherapy for Ewing’s sarcoma and primitive neuroectodermal tumor of bone. N Engl J Med. 2003;348:694–701.
Ahmad R, Mayol BR, Davis M. Extraskeletal Ewing’s sarcoma. Cancer. 1999;85:725–31.
Baldini EH, Demetri GD, Christopher DM, Fletcher CDM. Adults with Ewing’s sarcoma/primitive neuroectodermal tumor: adverse effect of older age and primary extraosseous disease on outcome. Ann Surg. 1999;230:79–86.
Martin RCG II, Brennan MF. Adult soft tissue ewing sarcoma or primitive neuroectodermal tumors. Arch Surg. 2003;138:281–5.
Ramachandran V, Mathew KG, Ganapathy Ramanan S. A single institution experience of combined modality management of extra skeletal Ewings sarcoma. World J Surg Oncol. 2007;5:3.
El Weshi A, Allam A, Ajarim D. Extraskeletal Ewing’s sarcoma family of tumours in adults: analysis of 57 patients from a single institution. Clin Oncol. 2010;22:374–81.
Rud NP, Reiman HM, Pritchard DJ. Extraosseous Ewing’s sarcoma. A study of 42 cases. Cancer. 1989;64:1548–53.
Sinkovics JG, Plager C, Ayala AG. Ewing sarcoma: its course and treatment in 50 adults patients. Oncology. 1980;37:114–9.
Prestidge BR, Donaldson SS. Treatment results among adults with childhood tumors: a 20-year experience. Int J Radiat Oncol Biol Phys. 1989;17:507–14.
Raney RB, Asmar L, Newton WA Jr. Ewing’s sarcoma of soft tissues in childhood: a report from the Intergroup Rhabdomyosarcoma Study, 1972–1991. J Clin Oncol. 1997;15:574–82.
Kinsella TJ, Triche TJ, Dickman PS. Extraskeletal Ewing’s sarcoma: result of combined modality treatment. J Clin Onclogy. 1983;1:489–95.
Sienbenrock KA, Nascimento AG, Rock MG. Comparison of soft tissue Ewing’s sarcoma and peripheral neuroectodermal tumor. Clin Orthop. 1996;329:288–99.
Verrill MW, Judson IR, Wiltshaw E. The use of paediatric chemotherapy protocols at full dose is both a rational and feasible treatment strategy in adults with Ewing’s family tumours. Ann Oncol. 1997;8:1099–105.
Smeland S, Wiebe T, Brosjo O. Chemotherapy in Ewing’s sarcoma. The Scandinavian Sarcoma Group experience. Acta Orthop Scand. 2004;75:87–91.
Bacci G, Ferrari S, Longhi A. Prognostic significance of serum LDH in Ewing’s sarcoma of bone. Oncol Rep. 1999;6:807–11.
Picci P, Rougraff BT, Bacci G. Prognostic significance of histopathologic response to chemotherapy in nonmetastatic Ewing’s sarcoma of the extremities. J Clin Oncol. 1993;11:1763–9.
Zagar TM, Triche TJ, Kinsella TJ. Extraosseous Ewing’s sarcoma: 25 years later. J Clin Oncol. 2008;26:4230–2.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arpaci, E., Yetisyigit, T., Seker, M. et al. Prognostic factors and clinical outcome of patients with Ewing’s sarcoma family of tumors in adults: multicentric study of the Anatolian Society of Medical Oncology. Med Oncol 30, 469 (2013). https://doi.org/10.1007/s12032-013-0469-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0469-z