Abstract
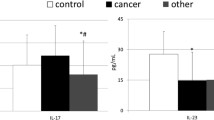
To investigate the expression of interleukin 17 (IL-17) in serum and tumor tissues, and evaluate the correlation between IL-17 and clinical characteristics from patients with gastric cancer. From January 2009 to January 2010, total 50 patients with gastric cancer and 50 healthy controls were enrolled. The patients included 32 males and 18 females with average age of (62.05 ± 11.98) years. The concentration of serum IL-17 was detected by ELISA. Intratumoral IL-17 expression and microvessel density (MVD) were examined by immunohistochemical staining. Compared with healthy controls, patients with gastric cancer had higher levels of IL-17 in serum (p < 0.01) and cancer tissues. High expression of IL-17 was associated with high MVD (p < 0.01). IL-17 in cancer tissues was associated with the clinical stage of the tumors (p < 0.01) and lymph node metastasis (p < 0.05). No statistically significant correlation between the serum IL-17 and the clinical pathological features was found. Increased expression of IL-17 was seen in patient with gastric cancer. IL-17 may be involved in the progression of gastric cancer by promoting angiogenesis in tumor microenvironment.




Similar content being viewed by others
References
Chen WQ. Estimation of cancer incidence and mortality in China in 2004–2005. Zhonghua Zhong Liu Za Zhi. 2009;31:664–8.
Bornschein J, Kandulski A, Selgrad M, Malfertheiner P. From gastric inflammation to gastric cancer. Dig Dis. 2010;28:609–14.
Tsai HF, Hsu PN. Interplay between helicobacter pylori and immune cells in immune pathogenesis of gastric inflammation and mucosal pathology. Cell Mol Immunol. 2010;7:255–9.
Gaffen SL. An overview of IL-17 function and signaling. Cytokine. 2008;43:402–7.
Michel ML, Mendes-da-Cruz D, Keller AC, Lochner M, Schneider E, Dy M, et al. Critical role of ROR-gammat in a new thymic pathway leading to IL-17-producing invariant NKT cell differentiation. Proc Natl Acad Sci USA. 2008;105:19845–50.
Ciric B, El-behi M, Cabrera R, Zhang GX, Rostami A. IL-23 drives pathogenic IL-17-producing CD8+ T cells. J Immunol. 2009;182:5296–305.
O’Brien RL, Roark CL, Born WK. IL-17-producing gammadelta T cells. Eur J Immunol. 2009;39:662–6.
Murugaiyan G, Saha B. Protumor vs antitumor functions of IL-17. J Immunol. 2009;183:4169–75.
Yu JJ, Gaffen SL. Interleukin-17: a novel inflammatory cytokine that bridges innate and adaptive immunity. Front Biosci. 2008;13:170–7.
Derhovanessian E, Adams V, Hähnel K, Groeger A, Pandha H, Ward S, et al. Pretreatment frequency of circulating IL-17+ CD4+ T-cells, but not Tregs, correlates with clinical response to whole-cell vaccination in prostate cancer patients. Int J Cancer. 2009;125:1372–9.
Zhang JP, Yan J, Xu J, Pang XH, Chen MS, Li L, et al. Increased intratumoral IL-17-producing cells correlate with poor survival in hepatocellular carcinoma patients. J Hepatol. 2009;50:980–9.
Miyahara Y, Odunsi K, Chen W, Peng G, Matsuzaki J, Wang RF. Generation and regulation of human CD4+ IL-17-producing T cells in ovarian cancer. Proc Natl Acad Sci USA. 2008;105:15505–10.
Wägsäter D, Löfgren S, Hugander A, Dimberg J. Expression of interleukin-17 in human colorectal cancer. Anticancer Res. 2006;26:4213–6.
Peng SF, Wang SJ, Chen JG, Dai XL, Shi Y, Li YZ, et al. Detection and significance of transcription factors and cytokines of Th17/Treg cells in peripheral blood in the gastric cancer patients. Zhonghua Zhong Liu Za Zhi. 2010;32:185–9.
Suzuki S, Dobashi Y, Hatakeyama Y, Tajiri R, Fujimura T, Heldin CH, et al. Clinicopathological significance of platelet-derived growth factor (PDGF)-B and vascular endothelial growth factor-A expression, PDGF receptor-β phosphorylation, and microvessel density in gastric cancer. BMC Cancer. 2010;10:659.
Iordache S, Saftoiu A, Georgescu CV, Ramboiu S, Gheonea DI, Filip M, et al. Vascular endothelial growth factor expression and microvessel density—two useful tools for the assessment of prognosis and survival in gastric cancer patients. J Gastrointestin Liver Dis. 2010;19:135–9.
Maruyama T, Kono K, Mizukami Y, Kawaguchi Y, Mimura K, Watanabe M, et al. Distribution of Th17 cells and FoxP3(+) regulatory T cells in tumor-infiltrating lymphocytes, tumor-draining lymph nodes and peripheral blood lymphocytes in patients with gastric cancer. Cancer Sci. 2010;101:1947–54.
Zhang B, Rong G, Wei H, Zhang M, Bi J, Ma L, et al. The prevalence of Th17 cells in patients with gastric cancer. Biochem Biophys Res Commun. 2008;374:533–7.
Iida T, Iwahashi M, Katsuda M, Ishida K, Nakamori M, Nakamura M, et al. Tumor-infiltrating CD4+ Th17 cells produce IL-17 in tumor microenvironment and promote tumor progression in human gastric cancer. Oncol Rep. 2011;25:1271–7.
Wang L, Yi T, Zhang W, Pardoll DM, Yu H. IL-17 enhances tumor development in carcinogen-induced skin cancer. Cancer Res. 2010;70:10112–20.
Numasaki M, Fukushi J, Ono M, Narula SK, Zavodny PJ, Kudo T, et al. Interleukin-17 promotes angiogenesis and tumor growth. Blood. 2003;101:2620–7.
Alexandrakis MG, Pappa CA, Miyakis S, Sfiridaki A, Kafousi M, Alegakis A, et al. Serum interleukin-17 and its relationship to angiogenic factors in multiple myeloma. Eur J Intern Med. 2006;17:412–6.
Liu J, Duan Y, Cheng X, Chen X, Xie W, Long H, et al. IL-17 is associated with poor prognosis and promotes angiogenesis via stimulating VEGF production of cancer cells in colorectal carcinoma. Biochem Biophys Res Commun. 2011;407:348–54.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meng, X.Y., Zhou, C.H., Ma, J. et al. Expression of interleukin-17 and its clinical significance in gastric cancer patients. Med Oncol 29, 3024–3028 (2012). https://doi.org/10.1007/s12032-012-0273-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-012-0273-1