Abstract
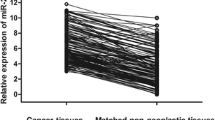
Recently, accumulating evidence indicates that dysregulation of miRNAs is associated with the initiation and progression of cancer. MiR-101 has been reported down-regulated in various types of cancer. The aim of this study was to investigate the expression profile of miR-101 and its target gene Mcl-1 in NSCLC and to assess their clinical significance. QRT-PCR was used in the detection of miR-101 and Mcl-1 mRNA expression both in NSCLC tissue and in adjacent normal lung tissue. Immunohistochemistry and Western blot analysis were used in the detection of Mcl-1 protein expression. The clinicopathological implications of these molecules were analyzed statistically. Survival analysis was performed to assess prognostic significance. Down-regulation of miR-101 was associated with overexpression of Mcl-1 mRNA in NSCLC tissue when compared with corresponding normal tissue, with a negative correlation (r = −0.724, P < 0.01). MiR-101 expression was significantly associated with pathological stage (P = 0.004) and lymph node involvement (P = 0.012). Overexpression of Mcl-1 was associated with pathological grade (P = 0.022) and lymph node involvement (P = 0.017). A comparison of survival curves of low versus high expressers of miR-101 and Mcl-1 revealed a highly significant difference in NSCLC (P < 0.05), which suggests that reduced expression of miR-101 versus overexpression of Mcl-1 is associated with a poorer prognosis. Our results suggest that down-regulation of miR-101 may result in enhanced expression of Mcl-1 in NSCLC, which consequently favored tumor progression. MiR-101 and Mcl-1 may play important roles as biomarkers for prognosis and therapeutic targets in NSCLC.


Similar content being viewed by others
References
Govindan R, Page N, Morgensztern D, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol. 2006;24:4539–44.
Weiss GJ, Bunn PA Jr, Camidge DR. From radiotherapy to targeted therapy: 20 years in the management of non-small-cell lung cancer. Oncology. 2006;20:1515–24.
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–97.
Bartels CL, Tsongalis GJ. MicroRNAs: novel biomarkers for human cancer. Clin Chem. 2009;55:623–31.
Cho WC. Role of miRNAs in lung cancer. Expert Rev Mol Diagn. 2009;9:773–6.
Varambally S, Cao Q, Mani RS, et al. Genomic loss of microRNA-101 leads to overexpression of histone methyltransferase EZH2 in cancer. Science. 2008;322:1695–9.
Su H, Yang JR, Xu T, et al. MicroRNA-101, down-regulated in hepatocellular carcinoma, promotes apoptosis and suppresses tumorigenicity. Cancer Res. 2009;69:1135–42.
Friedman JM, Liang G, Liu CC, et al. The putative tumor suppressor microRNA-101 modulates the cancer epigenome by repressing the polycomb group protein EZH2. Cancer Res. 2009;69:2623–9.
Clohessy JG, Zhuang J, de Boer J, et al. Mcl-1 interacts with truncated Bid and inhibits its induction of cytochrome c release and its role in receptor-mediated apoptosis. J Biol Chem. 2006;281:5750–9.
Cuconati A, Mukherjee C, Perez D, White E. DNA damage response and MCL-1 destruction initiate apoptosis in adenovirus-infected cells. Genes Dev. 2003;17:2922–32.
Wang HJ, Ruan HJ, He XJ, et al. MicroRNA-101 is down-regulated in gastric cancer and involved in cell migration and invasion. Eur J Cancer. 2010;46:2295–303.
Inagawa S, Itabashi M, Adachi S, et al. Expression and prognostic roles of beta catenin in hepatocellular carcinoma: correlation with tumor progression and postoperative survival. Clin Cancer Res. 2002;8:450–6.
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108.
Calin GA, Dumitru CD, Shimizu M, et al. Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci USA. 2002;99:15524–9.
Scott GK, Goga A, Bhaumik D, et al. Coordinate suppression of ERBB2 and ERBB3 by enforced expression of micro-RNA miR-125a or miR-125b. J Biol Chem. 2007;282:1479–86.
Takamizawa J, Konishi H, Yanagisawa K, et al. Reduced expression of the let-7 microRNAs in human lung cancers in association with shortened postoperative survival. Cancer Res. 2004;64:3753–6.
Asangani IA, Rasheed SA, Nikolova DA, et al. MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene. 2008;27:2128–36.
Smits M, Nilsson J, Mir SE, et al. miR-101 is down-regulated in glioblastoma resulting in EZH2-induced proliferation, migration, and angiogenesis. Oncotarget. 2010;1:710–20.
Strillacci A, Griffoni C, Sansone P, et al. MiR-101 downregulation is involved in cyclooxygenase-2 overexpression in human colon cancer cells. Exp Cell Res. 2009;315:1439–47.
Cho-Vega JH, Rassidakis GZ, Admirand JH, et al. MCL-1 expression in B-cell non-Hodgkin’s lymphomas. Hum Pathol. 2004;35:1095–100.
Allen TD, Zhu CQ, Jones KD, et al. Interaction between MYC and MCL1 in the genesis and outcome of non-small-cell lung cancer. Cancer Res. 2011;71:2212–21.
Acknowledgments
This work was supported by the National Natural Scientific Foundation of China Grant (No. 81170064) and the Natural Science Foundation of Jiangsu Province Grant (No. BK2011658).
Conflict of interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Liang Luo and Ting Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Luo, L., Zhang, T., Liu, H. et al. MiR-101 and Mcl-1 in non-small-cell lung cancer: expression profile and clinical significance. Med Oncol 29, 1681–1686 (2012). https://doi.org/10.1007/s12032-011-0085-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-0085-8