Abstract
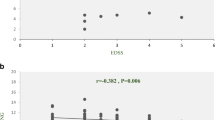
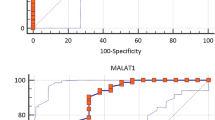
Long non-coding RNAs (lncRNAs) have been reported to participate in the pathogenesis of several complex disorders such as immune-related disorders. Multiple sclerosis (MS) as an inflammatory disorder of the central nervous system has been associated with aberrant expression of several lncRNAs. In the current study, we assessed expression of NF-κB-, autophagy-, and obesity-associated lncRNAs/genes and two inflammatory cytokines in the peripheral blood of MS patients and healthy controls. LNC-MKI67IP was down-regulated in total MS patients compared with total controls (P value < 0.0001). However, when comparing its expression in a gender-based manner, no significant difference was found between patients and controls. Expression of HNF1A-AS1 was significantly lower in total MS patients and patients of both sexes when compared with the matched controls (P value < 0.0001; P value = 0.03; P value = 0.004, respectively). Expression of LINC00305 had a similar pattern to HNF1A-AS1 (P value < 0.0001; P value = 0.005; P value < 0.0001, respectively). Expressions of other assessed NF-κB associated lncRNAs were not different between cases and controls. Expression of IL-1B was significantly lower in total MS patients compared with total controls (P value < 0.0001). Such decreased expression was detected in female patients compared with female controls as well (P value < 0.0001). Expression of IL-6 was not different between cases and controls. Expression of CEBPA was higher in total MS patients compared with controls (P value = 0.047). However, when dividing study participants into male and female subgroups, no significant difference was detected between cases and gender-matched controls. There were no significant difference in the expression of any assessed autophagy-associated lncRNAs between cases and controls. ATG5, CEBPA, HNF1A-AS1, IL-1B, LINC00305, and LNC-MKI67IP could differentiate disease status with diagnostic power values of 0.781, 0.582, 0.744, 0.76, 0.926, and 0.703, respectively. Expression levels of ADINR and CHAST were correlated with age of MS patients and disease duration, respectively (r = 0.33, P < 0.0001; r = 0.34, P < 0.0001, respectively). The present study highlighted the role of a number lncRNAs in the pathogenesis of MS and supported the previous data regarding aberrant expression of these transcripts in this immune-related disorder.





Similar content being viewed by others
References
Ambele MA, Dessels C, Durandt C, Pepper MS (2016) Genome-wide analysis of gene expression during adipogenesis in human adipose-derived stromal cells reveals novel patterns of gene expression during adipocyte differentiation. Stem Cell Res 16:725–734
Baldwin AS (2012) Regulation of cell death and autophagy by IKK and NF-κB: critical mechanisms in immune function and cancer. Immunol Rev 246:327–345
Bao C, Li Y, Huan L, Zhang Y, Zhao F, Wang Q, Liang L, Ding J, Liu L, Chen T, Li J, Yao M, Huang S, He X (2015) NF-kappaB signaling relieves negative regulation by miR-194 in hepatocellular carcinoma by suppressing the transcription factor HNF-1alpha. Sci Signal 8:ra75
Beauchemin P, Carruthers R (2016) MS arising during Tocilizumab therapy for rheumatoid arthritis. Mult Scler 22:254–256
Burm SM, Peferoen LA, Zuiderwijk-Sick EA, Haanstra KG, T Hart BA, Van der Valk P, Amor S, Bauer J, Bajramovic JJ (2016) Expression of IL-1beta in rhesus EAE and MS lesions is mainly induced in the CNS itself. J Neuroinflammation 13:138
Campbell IL, Erta M, Lim SL, Frausto R, May U, Rose-John S, Scheller J, Hidalgo J (2014) Trans-signaling is a dominant mechanism for the pathogenic actions of interleukin-6 in the brain. J Neurosci 34:2503–2513
Danikowski KM, Jayaraman S, Prabhakar BS (2017) Regulatory T cells in multiple sclerosis and myasthenia gravis. J Neuroinflammation 14:117–117
Davignon JL, Hayder M, Baron M, Boyer JF, Constantin A, Apparailly F, Poupot R, Cantagrel A (2013) Targeting monocytes/macrophages in the treatment of rheumatoid arthritis. Rheumatology (Oxford) 52:590–598
Ding CH, Yin C, Chen SJ, Wen LZ, Ding K, Lei SJ, Liu JP, Wang J, Chen KX, Jiang HL, Zhang X, Luo C, Xie WF (2018) The HNF1alpha-regulated lncRNA HNF1A-AS1 reverses the malignancy of hepatocellular carcinoma by enhancing the phosphatase activity of SHP-1. Mol Cancer 17:63
Fenoglio C, Oldoni E, Serpente M, De Riz MA, Arcaro M, D'Anca M, Pietroboni AM, Calvi A, Lecchi E, Goris A, Mallants K, Dubois B, Comi C, Cantello R, Scarpini E, Galimberti D (2018) LncRNAs expression profile in peripheral blood mononuclear cells from multiple sclerosis patients. J Neuroimmunol 324:129–135
Ganji M, Sayad A, Omrani MD, Arsang-Jang S, Mazdeh M, Taheri M (2019) Expression analysis of long non-coding RNAs and their target genes in multiple sclerosis patients. Neurol Sci 40:801–811
Gao J, Chen F, Hua M, Guo J, Nong Y, Tang Q, Zhong F, Qin L (2018) Knockdown of lncRNA MIR31HG inhibits cell proliferation in human HaCaT keratinocytes. Biol Res 51:30
Ghafouri-Fard S, Taheri M (2020) A comprehensive review of non-coding RNAs functions in multiple sclerosis. Eur J Pharmacol 173127
Gianfrancesco MA, Barcellos LF (2016) Obesity and multiple sclerosis susceptibility: a review. J Neurol Neuromed 1:1–5
Giralt M, Ramos R, Quintana A, Ferrer B, Erta M, Castro-Freire M, Comes G, Sanz E, Unzeta M, Pifarré P (2013) Induction of atypical EAE mediated by transgenic production of IL-6 in astrocytes in the absence of systemic IL-6. Glia 61:587–600
Gu Z, Hou Z, Zheng L, Wang X, Wu L, Zhang C (2018) LncRNA DICER1-AS1 promotes the proliferation, invasion and autophagy of osteosarcoma cells via miR-30b/ATG5. Biomed Pharmacother 104:110–118
Guerrero-Garcia JJ, Carrera-Quintanar L, Lopez-Roa RI, Marquez-Aguirre AL, Rojas-Mayorquin AE, Ortuno-Sahagun D (2016) Multiple sclerosis and obesity: possible roles of adipokines. Mediat Inflamm 2016:4036232
Hill AA, Anderson-Baucum EK, Kennedy AJ, Webb CD, Yull FE, Hasty AH (2015) Activation of NF-κB drives the enhanced survival of adipose tissue macrophages in an obesogenic environment. Molec Metabol 4:665–677
Hosseini A, Teimuri S, Ehsani M, Rasa SMM, Etemadifar M, Nasr Esfahani MH, Megraw TL, Ghaedi K (2019) LncRNAs associated with multiple sclerosis expressed in the Th1 cell lineage. J Cell Physiol 234:22153–22162
Krawczyk M, Emerson BM (2014) p50-associated COX-2 extragenic RNA (PACER) activates COX-2 gene expression by occluding repressive NF-kappaB complexes. Elife 3:e01776
Leibowitz SM, Yan J (2016) NF-kappaB pathways in the pathogenesis of multiple sclerosis and the therapeutic implications. Front Mol Neurosci 9:84
Li J, Liu C (2019) Coding or noncoding, the converging concepts of RNAs. Front Genet 10:496
Li Q, Verma IM (2002) NF-kappaB regulation in the immune system. Nat Rev Immunol 2:725–734
Li XY, Yu MQ, Han L, Chen L, Zhang DX, Zhou GN, Zhao Q, Sun TT (2019) LINC00305 represses miR-124 expression to trigger inflammatory insults in the presence of lipopolysaccharide. Artificial Cells Nanomed Biotechnol 47:2352–2360
Liang P, Le W (2015) Role of autophagy in the pathogenesis of multiple sclerosis. Neurosci Bull 31:435–444
Lin Z, Ge J, Wang Z, Ren J, Wang X, Xiong H, Gao J, Zhang Y, Zhang Q (2017a) Let-7e modulates the inflammatory response in vascular endothelial cells through ceRNA crosstalk. Sci Rep 7:42498
Lin ZW, Ge JF, Wang Z, Ren JW, Wang XW, Xiong H, Gao J, Zhang Y, Zhang QY (2017b) Let-7e modulates the inflammatory response in vascular endothelial cells through ceRNA crosstalk. Sci Rep 7
Liu B, Sun L, Liu Q, Gong C, Yao Y, Lv X, Lin L, YaO H, Su F, Li D, Zeng M, Song E (2015) A cytoplasmic NF-kappaB interacting long noncoding RNA blocks IkappaB phosphorylation and suppresses breast cancer metastasis. Cancer Cell 27:370–381
Liu T, Zhang L, Joo D, Sun S-C (2017) NF-κB signaling in inflammation. Signal Trans Target Ther 2:1–9
Rothaug M, Becker-Pauly C, Rose-John S (2016) The role of interleukin-6 signaling in nervous tissue. Biochimica et Biophysica Acta (BBA)-Molecular Cell Res 1863:1218–1227
Sigdel KR, Cheng A, Wang Y, Duan LH, Zhang YL (2015) The emerging functions of long noncoding RNA in immune cells: autoimmune diseases. J Immunol Res
Smith KJ, McDonald WI (1999) The pathophysiology of multiple sclerosis: the mechanisms underlying the production of symptoms and the natural history of the disease. Philos Trans R Soc Lond Ser B Biol Sci 354:1649–1673
Viereck J, Kumarswamy R, Foinquinos A, Xiao K, Avramopoulos P, Kunz M, Dittrich M, Maetzig T, Zimmer K, Remke J, Just A, Fendrich J, Scherf K, Bolesani E, Schambach A, Weidemann F, Zweigerdt R, De Windt LJ, Engelhardt S, Dandekar T, Batkai S, Thum T (2016) Long noncoding RNA Chast promotes cardiac remodeling. Sci Transl Med 8:326ra22
Xiao T, Liu L, Li H, Sun Y, Luo H, Li T, Wang S, Dalton S, ZhaO RC, Chen R (2015) Long noncoding RNA ADINR regulates adipogenesis by transcriptionally activating C/EBPalpha. Stem Cell Rep 5:856–865
Yan J, Greer JM (2008) NF-kappa B, a potential therapeutic target for the treatment of multiple sclerosis. CNS Neurol Disord Drug Targets 7:536–557
Zhang DD, Wang WT, Xiong J, Xie XM, Cui SS, ZhaO ZG, Li MJ, Zhang ZQ, Hao DL, Zhao X, Li YJ, Wang J, Chen HZ, Lv X, Liu DP (2017) Long noncoding RNA LINC00305 promotes inflammation by activating the AHRR-NF-kappaB pathway in human monocytes. Sci Rep 7:46204
Acknowledgments
The current study was supported by a grant from Shahid Beheshti University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Safa, A., Arsang-Jang, S., Taheri, M. et al. Dysregulation of NF-κB-Associated lncRNAs in Multiple Sclerosis Patients. J Mol Neurosci 71, 80–88 (2021). https://doi.org/10.1007/s12031-020-01628-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01628-2