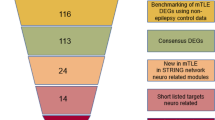
Abstract
In the epilepsy spectrum, temporal lobe epilepsy (TLE) is the most common and devastating focal and symptomatic epilepsy form in adults, where more than 30% of patients develop pharmacoresistance. It is not fully understood how the gene expression contributes to establishing an epileptic phenotype. Cerebrovascular remodeling directed by VEGF (vascular endothelial growth factor) signaling might modulate the synaptic neurotransmission in the epileptic brain. To address this question, the gene expression was profiled in biopsies of the temporal cortex from diagnosed patients with pharmacoresistant TLE that underwent surgical resection to seizure control. One hundred sixty-eight genes related to VEGF signaling and GABA and glutamate neurotransmissions were evaluated. Genes related to downstream signaling —phosphoinositide 3-kinase (PI3K), mitogen-activated protein kinases (MAPK), and Janus-activated kinase/signal transducer and activator of transcription (JAK/STAT) pathways— and neurotransmitters metabolism were evaluated too. Thirty-nine genes were upregulated. The genes encoding for G protein q polypeptide, serine racemase, gephyrin, and glutamate/cystine antiporter system xCT appeared as novel upregulated genes in the pharmacoresistant TLE. ClueGO, a Cytoscape plugin, was used to build a gene network associated using Gene Ontology (GO) terminology. Enrichment analysis by ClueGO retrieves that positive regulation of endothelial cell proliferation, nerve development, and neuronal apoptosis were over-represented categories. In conclusion, VEGF signaling is confirmed as a relevant mediator in the pharmacoresistant TLE. In addition, the enrichment analysis applied to differentially expressed genes suggests new pharmacological targets to be assessed in the treatment of pharmacoresistant TLE. Results make up an approximation to better understand the epileptic brain and complement the available data.



Similar content being viewed by others
Abbreviations
- TLE:
-
Temporal lobe epilepsy
- HS:
-
Hippocampal sclerosis
- FCD:
-
Focal cortical dysplasia
- GABA:
-
Gamma-aminobutyric acid
- MAPK:
-
Mitogen-activated protein kinase
- CREB:
-
cAMP-responsive element-binding protein
- RT:
-
Retrotranscription
- qPCR:
-
Quantitative PCR
- VEGF:
-
Vascular endothelial growth factor
- PI3K:
-
Phosphoinositide 3-kinase
- JAK/STAT:
-
Janus-activated kinase/signal transducer and activator of transcription
References
Arion D, Sabatini M, Unger T, Pastor J, Alonso-Nanclares L, Ballesteros-Yáñez I, García Sola R, Muñoz A, Mirnics K, DeFelipe J (2006) Correlation of transcriptome profile with electrical activity in temporal lobe epilepsy. Neurobiol Dis 22(2):374–387. https://doi.org/10.1016/j.nbd.2005.12.012
Aronica E, Boer K, Becker A, Redeker S, Spliet WGM, van Rijen PC et al (2008) Gene expression profile analysis of epilepsy-associated gangliogliomas. Neuroscience 151(1):272–292. https://doi.org/10.1016/j.neuroscience.2007.10.036
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G (2000) Gene ontology: tool for the unification of biology. Nat Genet 25:25–29. https://doi.org/10.1038/75556
Baburin I, Khom S, Timin E, Hohaus A, Sieghart W, Hering S (2008) Estimating the efficiency of benzodiazepines on GABA A receptors comprising γ1 or γ2 subunits. Br J Pharmacol 155(3):424–433. https://doi.org/10.1038/bjp.2008.271
Barkovich AJ, Guerrini R, Kuzniecky RI, Jackson GD, Dobyns WB (2012) A developmental and genetic classification for malformations of cortical development: update 2012. Brain 135(Pt 5):1348–1369. https://doi.org/10.1093/brain/aws019
Bartolomei F, Cosandier-Rimele D, McGonigal A, Aubert S, Régis J, Gavaret M, Wendling F, Chauvel P (2010) From mesial temporal lobe to temporoperisylvian seizures: a quantified study of temporal lobe seizure networks. Epilepsia 51(10):2147–2158. https://doi.org/10.1111/j.1528-1167.2010.02690.x
Bateman LM, Li CS, Seyal M (2008) Ictal hypoxemia in localization-related epilepsy: analysis of incidence, severity and risk factors. Brain 131(12):3239–3245. https://doi.org/10.1093/brain/awn277
Bindea G, Mlecnik B, Hackl H, Charoentong P, Tosolini M, Kirilovsky A, Fridman WH, Pagès F, Trajanoski Z, Galon J (2009) ClueGO: a Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 25(8):1091–1093. https://doi.org/10.1093/bioinformatics/btp101
Bindea G, Galon J, Mlecnik B (2013) CluePedia Cytoscape plugin: pathway insights using integrated experimental and in silico data. Bioinformatics 29(5):661–663. https://doi.org/10.1093/bioinformatics/btt019
Blair RD (2012) Temporal lobe epilepsy semiology. Epilepsy Research and Treatment 2012:751510. https://doi.org/10.1155/2012/751510
Blümcke I, Thom M, Aronica E, Armstrong DD, Vinters HV, Palmini A, Jacques TS, Avanzini G, Barkovich AJ, Battaglia G, Becker A, Cepeda C, Cendes F, Colombo N, Crino P, Cross JH, Delalande O, Dubeau F, Duncan J, Guerrini R, Kahane P, Mathern G, Najm I, Özkara Ç, Raybaud C, Represa A, Roper SN, Salamon N, Schulze-Bonhage A, Tassi L, Vezzani A, Spreafico R (2011) The clinicopathologic spectrum of focal cortical dysplasias: a consensus classification proposed by an ad hoc Task Force of the ILAE Diagnostic Methods Commission. Epilepsia 52(1):158–174
Blümcke I, Thom M, Aronica E, Armstrong DD, Bartolomei F, Bernasconi A, Bernasconi N, Bien CG, Cendes F, Coras R, Cross JH, Jacques TS, Kahane P, Mathern GW, Miyata H, Moshé SL, Oz B, Özkara Ç, Perucca E, Sisodiya S, Wiebe S, Spreafico R (2013) International consensus classification of hippocampal sclerosis in temporal lobe epilepsy: a Task Force report from the ILAE Commission on Diagnostic Methods. Epilepsia 54(7):1315–1329. https://doi.org/10.1111/epi.12220
Boer K, Troost D, Spliet WGM, Van Rijen PC, Gorter JA, Aronica E (2008) Cellular distribution of vascular endothelial growth factor A (VEGFA) and B (VEGFB) and VEGF receptors 1 and 2 in focal cortical dysplasia type IIB. Acta Neuropathol 115(6):683–696. https://doi.org/10.1007/s00401-008-0354-6
Bough KJ, Mott DD, Dingledine RJ (2004) Medial perforant path inhibition mediated by mGluR7 is reduced after status epilepticus. J Neurophysiol 92(3):1549–1557. https://doi.org/10.1152/jn.00315.2004
Choii G, Ko J (2015) Gephyrin: a central GABAergic synapse organizer. Exp Mol Med 47:e158. https://doi.org/10.1038/emm.2015.5
Chomczynski P, Sacchi N (2006) The single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction: twenty-something years on. Nat Protoc 1(2):581–585. https://doi.org/10.1038/nprot.2006.83
Croll SD, McCloskey DP, Nicoletti JN, & Scharfman HE (2005) VEGF as a novel seizure therapeutic: killing two birds with one stone. Growth Factors and Epilepsy. New York, NY: Nova sciences publishers, 145-162
Darstein M, Petralia RS, Swanson GT, Wenthold RJ, Heinemann SF (2003) Distribution of kainate receptor subunits at hippocampal mossy fiber synapses. J Neurosci 23(22):8013–8019
Drexel M, Kirchmair E, Sperk G (2012) Changes in the expression of GABAA receptor subunit mRNAs in parahippocampal areas after kainic acid-induced seizures. Front Neural Circuit 7:142. https://doi.org/10.3389/fncir.2013.00142
El Mestikawy S, Wallén-Mackenzie Å, Fortin GM, Descarries L, Trudeau LE (2011) From glutamate co-release to vesicular synergy: vesicular glutamate transporters. Nat Rev Neurosci 12:204–216. https://doi.org/10.1038/nrn2969
Farrant M, Nusser Z (2005) Variations on an inhibitory theme: phasic and tonic activation of GABA A receptors. Nat Rev Neurosci 6:215–229. https://doi.org/10.1038/nrn1625
Feast A, Martinian L, Liu J, Catarino CB, Thom M, Sisodiya SM (2012) Investigation of hypoxia-inducible factor-1α in hippocampal sclerosis: a postmortem study. Epilepsia 53(8):1349–1359. https://doi.org/10.1111/j.1528-1167.2012.03591.x
Fiest KM, Sajobi TT, Wiebe S (2014) Epilepsy surgery and meaningful improvements in quality of life: results from a randomized controlled trial. Epilepsia 55(6):886–892. https://doi.org/10.1111/epi.12625
González-Albo MC, Gómez-Utrero E, Sánchez A, Sola RG, DeFelipe J (2001) Changes in the colocalization of glutamate ionotropic receptor subunits in the human epileptic temporal lobe cortex. Exp Brain Res 138(3):398–402. https://doi.org/10.1007/s002210100720
Grabenstatter HL, Cogswell M, Cruz Del Angel Y, Carlsen J, Gonzalez MI, Raol YH et al (2014) Effect of spontaneous seizures on GABAA receptor α4 subunit expression in an animal model of temporal lobe epilepsy. Epilepsia 55(11):1826–1833. https://doi.org/10.1111/epi.12771
Griffith M, Griffith OL, Coffman AC, Weible JV, Mcmichael JF, Spies NC et al (2013) DGIdb: mining the druggable genome. Nat Methods 10(12):1209–1210. https://doi.org/10.1038/nmeth.2689
Guaiquil VH, Pan Z, Karagianni N, Fukuoka S, Alegre G, Rosenblatt MI (2014) Vegf-B selectively regenerates injured peripheral neurons and restores sensory and trophic functions. Proc Natl Acad Sci U S A 111(48):17272–17277. https://doi.org/10.1073/pnas.1407227111
Guérit S, Allain AE, Léon C, Cazenave W, Ferrara N, Branchereau P, Bikfalvi A (2014) VEGF modulates synaptic activity in the developing spinal cord. Dev Neurobiol 74(11):1110–1122. https://doi.org/10.1002/dneu.22187
Hao T, Rockwell P (2013) Signaling through the vascular endothelial growth factor receptor VEGFR-2 protects hippocampal neurons from mitochondrial dysfunction and oxidative stress. Free Radic Biol Med 63:421–431. https://doi.org/10.1016/j.freeradbiomed.2013.05.036
Henshall DC, Bonislawski DP, Skradski SL, Araki T, Lan JQ, Schindler CK, Meller R, Simon RP (2001) Formation of the Apaf-1/cytochrome c complex precedes activation of caspase-9 during seizure-induced neuronal death. Cell Death Differ 8(12):1169–1181. https://doi.org/10.1038/sj.cdd.4400921
Houser CR, Esclapez M (1996) Vulnerability and plasticity of the GABA system in the pilocarpine model of spontaneous recurrent seizures. Epilepsy Res 26(1):207–218. https://doi.org/10.1016/S0920-1211(96)00054-X
Kortenbruck G, Berger E, Speckmann EJ, Musshoff U (2001) RNA editing at the Q/R site for the glutamate receptor subunits GluR2, GluR5, and GluR6 in hippocampus and temporal cortex from epileptic patients. Neurobiol Dis 8(3):459–468. https://doi.org/10.1006/nbdi.2001.0394
Lee TS, Mane S, Eid T, Zhao H, Lin A, Guan Z, Kim JH, Schweitzer J, King-Stevens D, Weber P, Spencer SS, Spencer DD, de Lanerolle NC (2007) Gene expression in temporal lobe epilepsy is consistent with increased release of glutamate by astrocytes. Mol Med 13(1–2):1–13. https://doi.org/10.2119/2006-00079.Lee
Lewerenz J, Baxter P, Kassubek R, Albrecht P, Van Liefferinge J, Westhoff MA et al (2014) Phosphoinositide 3-kinases upregulate system xc- via eukaryotic initiation factor 2α and activating transcription factor 4-a pathway active in glioblastomas and epilepsy. Antioxid Redox Signal 20(18):2907–2922. https://doi.org/10.1089/ars.2013.5455
Li T, Fan Y, Luo Y, Xiao B, Lu C (2006) In vivo delivery of a XIAP (BIR3-RING) fusion protein containing the protein transduction domain protects against neuronal death induced by seizures. Exp Neurol 197(2):301–308. https://doi.org/10.1016/j.expneurol.2005.08.021
Li X, Lee C, Tang Z, Zhang F, Arjunan P, Li Y, Hou X, Kumar A, Dong L (2009) VEGF-B: a survival, or an angiogenic factor? Cell Adhes Migr 3(4):322–327. https://doi.org/10.4161/cam.3.4.9459
Lin YX, Lin K, Liu XX, Kang DZ, Ye ZX, Wang XF et al (2015) PI3K-AKT pathway polymerase chain reaction (PCR) array analysis of epilepsy induced by type II focal cortical dysplasia. Genet Mol Res 14(3):9994–10000. https://doi.org/10.4238/2015.August.21.5
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408
Marchi N, Lerner-Natoli M (2013) Cerebrovascular remodeling and epilepsy. Neuroscientist 19(3):304–312. https://doi.org/10.1177/1073858412462747
Mathern GW, Pretorius JK, Kornblum HI, Mendoza D, Lozada A, Leite JP, Chimelli L, Born DE, Fried I, Sakamoto AC, Assirati JA, Peacock WJ, Ojemann GA, Adelson PD (1998) Altered hippocampal kainate-receptor mRNA levels in temporal lobe epilepsy patients. Neurobiol Dis 5(3):151–176. https://doi.org/10.1006/nbdi.1998.0200
McCloskey DP, Croll SD, Scharfman HE (2005) Depression of synaptic transmission by vascular endothelial growth factor in adult rat hippocampus and evidence for increased efficacy after chronic seizures. J Neurosci 25(39):8889–8897. https://doi.org/10.1523/JNEUROSCI.2577-05.2005
Minelli A, Zanardini R, Abate M, Bortolomasi M, Gennarelli M, & Bocchio-Chiavetto L (2011) Vascular endothelial growth factor (VEGF) serum concentration during electroconvulsive therapy (ECT) in treatment resistant depressed patients Progress in neuropsychopharmacology & biological psychiatry, 35(5), 1322–1325. https://doi.org/10.1016/j.pnpbp.2011.04.013
Morales Chacón L, Estupiñan B, Lorigados Pedre L, Trápaga Quincoses O, García Maeso I, Sanchez A et al (2009) Microscopic mild focal cortical dysplasia in temporal lobe dual pathology: an electrocorticography study. Seizure 18(8):593–600. https://doi.org/10.1016/j.seizure.2009.06.008
Morin-Brureau M, Lebrun A, Rousset MC, Fagni L, Bockaert J, de Bock F, Lerner-Natoli M (2011) Epileptiform activity induces vascular remodeling and zonula occludens 1 downregulation in organotypic hippocampal cultures: role of VEGF signaling pathways. J Neurosci 31(29):10677–10688. https://doi.org/10.1523/JNEUROSCI.5692-10.2011
Overwater IE, Rietman AB, van Eeghen AM, de Wit MCY (2019) Everolimus for the treatment of refractory seizures associated with tuberous sclerosis complex (TSC): current perspectives. Ther Clin Risk Manag 15:951–955. https://doi.org/10.2147/TCRM.S145630
Özbas-Gerçeker F, Redeker S, Boer K, Özgüç M, Saygi S, Dalkara T et al (2006) Serial analysis of gene expression in the hippocampus of patients with mesial temporal lobe epilepsy. Neuroscience 138(2):457–474. https://doi.org/10.1016/j.neuroscience.2005.11.043
Paternain AV, Herrera MT, Nieto MA, Lerma J (2000) GluR5 and GluR6 kainate receptor subunits coexist in hippocampal neurons and coassemble to form functional receptors. J Neurosci 20(1):196–205
Peret A, Christie LA, Ouedraogo DW, Gorlewicz A, Epsztein JÔ, Mulle C, Crépel V (2014) Contribution of aberrant GluK2-containing kainate receptors to chronic seizures in temporal lobe epilepsy. Cell Rep 8(2):347–354. https://doi.org/10.1016/j.celrep.2014.06.032
Rigau V, Morin M, Rousset MC, de Bock F, Lebrun A, Coubes P, Picot MC, Baldy-Moulinier M, Bockaert J, Crespel A, Lerner-Natoli M (2007) Angiogenesis is associated with blood-brain barrier permeability in temporal lobe epilepsy. Brain 130(Pt 7):1942–1956. https://doi.org/10.1093/brain/awm118
Rosenstein JM, Krum JM, Ruhrberg C (2010) VEGF in the nervous system. Organogenesis 6(2):107–114. https://doi.org/10.4161/org.6.2.11687
Ryu HJ, Kim JE, Yeo SI, Kim DS, Kwon OS, Choi SY, Kang TC (2010) Potential roles of D-serine and serine racemase in experimental temporal lobe epilepsy. J Neurosci Res 88(11):2469–2482. https://doi.org/10.1002/jnr.22415
Schindler CK, Pearson EG, Bonner HP, So NK, Simon RP, Prehn JHM, Henshall DC (2006) Caspase-3 cleavage and nuclear localization of caspase-activated DNase in human temporal lobe epilepsy. J Cereb Blood Flow Metab 26(4):583–589. https://doi.org/10.1038/sj.jcbfm.9600219
Shirley MD, Tang H, Gallione CJ, Baugher JD, Frelin LP, Cohen B, North PE, Marchuk DA, Comi AM, Pevsner J (2013) Sturge-Weber syndrome and port-wine stains caused by somatic mutation in GNAQ. N Engl J Med 368(21):1971–1979. https://doi.org/10.1056/NEJMoa1213507
Sigel E, Steinmann ME (2012) Structure, function, and modulation of GABAA receptors. J Biol Chem 287:40224–40231. https://doi.org/10.1074/jbc.R112.386664
Sperk G, Schwarzer C, Tsunashima K, Kandlhofer S (1998) Expression of GABA(A) receptor subunits in the hippocampus of the rat after kainic acid-induced seizures. Epilepsy Res 32(1–2):129–139. https://doi.org/10.1016/S0920-1211(98)00046-1
Tessler S, Danbolt NC, Faull RLM, Storm-Mathisen J, Emson PC (1999) Expression of the glutamate transporters in human temporal lobe epilepsy. Neuroscience 88(4):1083–1091. https://doi.org/10.1016/S0306-4522(98)00301-7
Thom M, Eriksson S, Martinian L, Caboclo LO, McEvoy AW, Duncan JS, Sisodiya SM (2009) Temporal lobe sclerosis associated with hippocampal sclerosis in temporal lobe epilepsy: neuropathological features. J Neuropathol Exp Neurol 68(8):928–938. https://doi.org/10.1097/NEN.0b013e3181b05d67
Tica J, Bradbury EJ, & Didangelos A (2018) Combined transcriptomics, proteomics and bioinformatics identify drug targets in spinal cord injury. Int J Mol Sci, 19(5), pii: E1461. https://doi.org/10.3390/ijms19051461
Ulu MO, Tanriverdi T, Oz B, Biceroglu H, Isler C, Eraslan BS, Ozkara C, Ozyurt E, Uzan M (2010) The expression of astroglial glutamate transporters in patients with focal cortical dysplasia: an immunohistochemical study. Acta Neurochir 152(5):845–853. https://doi.org/10.1007/s00701-009-0548-2
Van Liefferinge J, Jensen CJ, Albertini G, Bentea E, Demuyser T, Merckx E et al (2015) Altered vesicular glutamate transporter expression in human temporal lobe epilepsy with hippocampal sclerosis. Neurosci Lett 590:184–188. https://doi.org/10.1016/j.neulet.2015.01.080
Venugopal AK, Sameer Kumar GS, Mahadevan A, Selvan LDN, Marimuthu A, Dikshit JB et al (2012) Transcriptomic profiling of medial temporal lobe epilepsy. J Proteomics Bioinform 5(2):31–39. https://doi.org/10.4172/jpb.1000210
Vollmar W, Gloger J, Berger E, Kortenbruck G, Köhling R, Speckmann EJ, Musshoff U (2004) RNA editing (R/G site) and flip-flop splicing of the AMPA receptor subunit GluR2 in nervous tissue of epilepsy patients. Neurobiol Dis 15(2):371–379. https://doi.org/10.1016/j.nbd.2003.11.006
Wiebe S, Blume WT, Girvin JP, Eliasziw M (2001) A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med 345:311–318. https://doi.org/10.1056/NEJM200108023450501
Yu W, Blas ALD (2008) Gephyrin expression and clustering affects the size of glutamatergic synaptic contacts. J Neurochem 104(3):830–845. https://doi.org/10.1111/j.1471-4159.2007.05014.x
Acknowledgments
The authors thank their institutions that provided the economic and technical support to carry out this study.
Funding
This work is supported by the National Council of Science and Technology of Mexico (CONACYT; Grant no. 177594).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by RDCT, MEUG, LLMC, BED, LR, SOS, and MAV. The first draft of the manuscript was written by RDCT and MEUG and all authors commented on previous versions of the manuscript. Funding acquisition: CBZ and MEUG. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
The Ethics Committees of each institution involved in this research approved the present research in line with the Declaration of Helsinki. We obtained informed consent from the patients to perform this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• GABA, glutamate, and VEGF signaling genes were upregulated in the temporal cortex of patients with pharmacoresistant TLE.
• G protein q polypeptide, serine racemase, and gephyrin appeared as novel genes to be studied in pharmacoresistant TLE.
• Similarly, glutamate/cystine antiporter system xCT and VEGF signaling genes should be considered for future studies in pharmacoresistant TLE.
• The VEGF signaling is confirmed as a relevant mediator in pharmacoresistant TLE.
Electronic supplementary material
ESM 1
(PDF 1508 kb)
Rights and permissions
About this article
Cite this article
Castro-Torres, R.D., Ureña-Guerrero, M.E., Morales-Chacón, L.M. et al. New Aspects of VEGF, GABA, and Glutamate Signaling in the Neocortex of Human Temporal Lobe Pharmacoresistant Epilepsy Revealed by RT-qPCR Arrays. J Mol Neurosci 70, 916–929 (2020). https://doi.org/10.1007/s12031-020-01519-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01519-6