Abstract
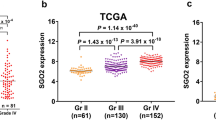
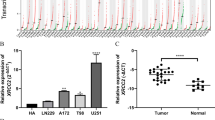
Cut-like homeobox-1 (CUX1) is expressed in the upper layer of the cortex and participates in DNA replication, cell cycle control, and DNA repair. It has been shown to be involved in the proliferation of various types of solid tumors. The aims of this study were to explore the relationship between CUX1 expression and the prognosis of glioma by performing a series of functional experiments and bioinformatic analyses. Firstly, we found that CUX1 expression levels differed among patients with different grades of gliomas, and they were significantly correlated with the prognosis of glioma patients according to an analysis of data from a public database. qRT-PCR, western blotting, and immunohistochemical analysis of CUX1 were performed to demonstrate that the expression of CUX1 was positively correlated with the glioma WHO grade (P < 0.05) and several malignant clinical pathological parameters, including Ki67 and P53mut. In addition, the multivariate Cox regression and Kaplan–Meier curves showed that CUX1 expression exerted predictive value for overall survival. Finally, to further investigate the functions of CUX1, we identified CUX1-associated genes and, though GO/KEGG analysis, their associated biological functions and signaling pathways; the results suggested that the activity of CUX1 might be exerted via the JAK-STAT pathway or other key regulators of the cell cycle to promote proliferation, inflammation, and chemotherapy resistance in glioma. Taken together, these results indicate that CUX1 is a potential biomarker of malignancy and prognosis and may serve as a potential therapeutic target for glioma patients.






Similar content being viewed by others
References
Cancer Genome Atlas Research, N (2008) Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455:1061–1068. https://doi.org/10.1038/nature07385
Chandrashekar DS, Bashel B, Balasubramanya SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi BVSK, Varambally S (2017) UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia 19:649–658. https://doi.org/10.1016/j.neo.2017.05.002
Cipriani S, Nardelli J, Verney C, Delezoide AL, Guimiot F, Gressens P, Adle-Biassette H (2016) Dynamic expression patterns of progenitor and pyramidal neuron layer markers in the developing human hippocampus. Cereb Cortex 26:1255–1271. https://doi.org/10.1093/cercor/bhv079
Clark CE, Hingorani SR, Mick R, Combs C, Tuveson DA, Vonderheide RH (2007) Dynamics of the immune reaction to pancreatic cancer from inception to invasion. Cancer Res 67:9518–9527. https://doi.org/10.1158/0008-5472.CAN-07-0175
Darsigny M, St-Jean S, Boudreau F (2010) Cux1 transcription factor is induced in inflammatory bowel disease and protects against experimental colitis. Inflamm Bowel Dis 16:1739–1750. https://doi.org/10.1002/ibd.21274
Diodato A, Ruinart de Brimont M, Yim YS, Derian N, Perrin S, Pouch J, Klatzmann D, Garel S, Choi GB, Fleischmann A (2016) Molecular signatures of neural connectivity in the olfactory cortex. Nat Commun 7:12238. https://doi.org/10.1038/ncomms12238
Fan X, Wang H, Zhou J, Wang S, Zhang X, Li T, Nie Y, Liu B (2014) The transcription factor CUTL1 is associated with proliferation and prognosis in malignant melanoma. Melanoma Res 24:198–206. https://doi.org/10.1097/CMR.0000000000000064
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. https://doi.org/10.1016/j.cell.2011.02.013
Hao Z, Hu S, Liu Z, Song W, Zhao Y, Li M (2018) Circular RNAs: functions and prospects in glioma. J Mol Neurosci. https://doi.org/10.1007/s12031-018-1211-2
Harada R, Vadnais C, Sansregret L, Leduy L, Berube G, Robert F, Nepveu A (2008) Genome-wide location analysis and expression studies reveal a role for p110 CUX1 in the activation of DNA replication genes. Nucleic Acids Res 36:189–202. https://doi.org/10.1093/nar/gkm970
Hilmi C, Larribere L, Giuliano S, Bille K, Ortonne JP, Ballotti R, Bertolotto C (2008) IGF1 promotes resistance to apoptosis in melanoma cells through an increased expression of BCL2, BCL-X(L), and survivin. J Invest Dermatol 128:1499–1505. https://doi.org/10.1038/sj.jid.5701185
Huang T, Kim CK, Alvarez AA, Pangeni RP, Wan X, Song X, Shi T, Yang Y, Sastry N, Horbinski CM, Lu S, Stupp R, Kessler JA, Nishikawa R, Nakano I, Sulman EP, Lu X, James CD, Yin XM, Hu B, Cheng SY (2017) MST4 phosphorylation of ATG4B regulates autophagic activity, tumorigenicity, and radioresistance in glioblastoma. Cancer Cell 32:840–855 e8. https://doi.org/10.1016/j.ccell.2017.11.005
Hulea L, Nepveu A (2012) CUX1 transcription factors: from biochemical activities and cell-based assays to mouse models and human diseases. Gene 497:18–26. https://doi.org/10.1016/j.gene.2012.01.039
Kaur S, Ramdzan ZM, Guiot MC, Li L, Leduy L, Ramotar D, Sabri S, Abdulkarim B, Nepveu A (2018) CUX1 stimulates APE1 enzymatic activity and increases the resistance of glioblastoma cells to the mono-alkylating agent temozolomide. Neuro-Oncology 20:484–493. https://doi.org/10.1093/neuonc/nox178
Kedinger V, Sansregret L, Harada R, Vadnais C, Cadieux C, Fathers K, Park M, Nepveu A (2009) p110 CUX1 homeodomain protein stimulates cell migration and invasion in part through a regulatory cascade culminating in the repression of E-cadherin and occludin. J Biol Chem 284:27701–27711. https://doi.org/10.1074/jbc.M109.031849
Kojima K, Takata A, Vadnais C, Otsuka M, Yoshikawa T, Akanuma M, Kondo Y, Kang YJ, Kishikawa T, Kato N, Xie Z, Zhang WJ, Yoshida H, Omata M, Nepveu A, Koike K (2011) MicroRNA122 is a key regulator of alpha-fetoprotein expression and influences the aggressiveness of hepatocellular carcinoma. Nat Commun 2:338. https://doi.org/10.1038/ncomms1345
Kuhnemuth B, Muhlberg L, Schipper M, Griesmann H, Neesse A, Milosevic N, Wissniowski T, Buchholz M, Gress TM, Michl P (2015) CUX1 modulates polarization of tumor-associated macrophages by antagonizing NF-kappaB signaling. Oncogene 34:177–187. https://doi.org/10.1038/onc.2013.530
Liu H, Xie X, Yang X, Li Y, Wang Y, Xu D (2017) Enhanced inflammatory damage by microRNA-136 targeting Klotho expression in HK-2 cells by modulating JAK/STAT pathway. Pharmazie 72:265–271. https://doi.org/10.1691/ph.2017.6990
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820. https://doi.org/10.1007/s00401-016-1545-1
Michl P, Ramjaun AR, Pardo OE, Warne PH, Wagner M, Poulsom R, D'Arrigo C, Ryder K, Menke A, Gress T, Downward J (2005) CUTL1 is a target of TGF(beta) signaling that enhances cancer cell motility and invasiveness. Cancer Cell 7:521–532. https://doi.org/10.1016/j.ccr.2005.05.018
Nduom EK, Wei J, Yaghi NK, Huang N, Kong LY, Gabrusiewicz K, Ling X, Zhou S, Ivan C, Chen JQ, Burks JK, Fuller GN, Calin GA, Conrad CA, Creasy C, Ritthipichai K, Radvanyi L, Heimberger AB (2016) PD-L1 expression and prognostic impact in glioblastoma. Neuro-Oncology 18:195–205. https://doi.org/10.1093/neuonc/nov172
Ostrom QT, Gittleman H, Xu J, Kromer C, Wolinsky Y, Kruchko C, Barnholtz-Sloan JS (2016) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2009–2013. Neuro-Oncology 18:v1–v75. https://doi.org/10.1093/neuonc/now207
Ramdzan ZM, Nepveu A (2014) CUX1, a haploinsufficient tumour suppressor gene overexpressed in advanced cancers. Nat Rev Cancer 14:673–682. https://doi.org/10.1038/nrc3805
Ramdzan ZM, Vadnais C, Pal R, Vandal G, Cadieux C, Leduy L, Davoudi S, Hulea L, Yao L, Karnezis AN, Paquet M, Dankort D, Nepveu A (2014) RAS transformation requires CUX1-dependent repair of oxidative DNA damage. PLoS Biol 12:e1001807. https://doi.org/10.1371/journal.pbio.1001807
Ripka S, Neesse A, Riedel J, Bug E, Aigner A, Poulsom R, Fulda S, Neoptolemos J, Greenhalf W, Barth P, Gress TM, Michl P (2010) CUX1: target of Akt signalling and mediator of resistance to apoptosis in pancreatic cancer. Gut 59:1101–1110. https://doi.org/10.1136/gut.2009.189720
Sansregret L, Goulet B, Harada R, Wilson B, Leduy L, Bertoglio J, Nepveu A (2006) The p110 isoform of the CDP/Cux transcription factor accelerates entry into S phase. Mol Cell Biol 26:2441–2455. https://doi.org/10.1128/MCB.26.6.2441-2455.2006
Sansregret L, Gallo D, Santaguida M, Leduy L, Harada R, Nepveu A (2010) Hyperphosphorylation by cyclin B/CDK1 in mitosis resets CUX1 DNA binding clock at each cell cycle. J Biol Chem 285:32834–32843. https://doi.org/10.1074/jbc.M110.156406
Sansregret L, Vadnais C, Livingstone J, Kwiatkowski N, Awan A, Cadieux C, Leduy L, Hallett MT, Nepveu A (2011) Cut homeobox 1 causes chromosomal instability by promoting bipolar division after cytokinesis failure. Proc Natl Acad Sci U S A 108:1949–1954. https://doi.org/10.1073/pnas.1008403108
Shi J, Hou S, Huang J, Wang S, Huan W, Huang C, Liu X, Jiang R, Qian W, Lu J, Wang X, Shi W, Huang R, Chen J (2017) An MSN-PEG-IP drug delivery system and IL13Ralpha2 as targeted therapy for glioma. Nanoscale 9:8970–8981. https://doi.org/10.1039/c6nr08786h
Szklarczyk D, Morris JH, Cook H, Kuhn M, Wyder S, Simonovic M, Santos A, Doncheva NT, Roth A, Bork P, Jensen LJ, von Mering C (2017) The string database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res 45:D362–D368. https://doi.org/10.1093/nar/gkw937
Thoennissen NH, Lasho T, Thoennissen GB, Ogawa S, Tefferi A, Koeffler HP (2011) Novel CUX1 missense mutation in association with 7q- at leukemic transformation of MPN. Am J Hematol 86:703–705. https://doi.org/10.1002/ajh.22069
Truscott M, Denault JB, Goulet B, Leduy L, Salvesen GS, Nepveu A (2007) Carboxyl-terminal proteolytic processing of CUX1 by a caspase enables transcriptional activation in proliferating cells. J Biol Chem 282:30216–30226. https://doi.org/10.1074/jbc.M702328200
Tu Y, Zhong Y, Fu J, Cao Y, Fu G, Tian X, Wang B (2011) Activation of JAK/STAT signal pathway predicts poor prognosis of patients with gliomas. Med Oncol 28:15–23. https://doi.org/10.1007/s12032-010-9435-1
Vadnais C, Shooshtarizadeh P, Rajadurai CV, Lesurf R, Hulea L, Davoudi S, Cadieux C, Hallett M, Park M, Nepveu A (2014) Autocrine activation of the Wnt/beta-catenin pathway by CUX1 and GLIS1 in breast cancers. Biol Open 3:937–946. https://doi.org/10.1242/bio.20148193
Vallee A, Lecarpentier Y, Guillevin R, Vallee JN (2018) Opposite interplay between the canonical WNT/beta-catenin pathway and PPAR gamma: a potential therapeutic target in gliomas. Neurosci Bull 34:573–588. https://doi.org/10.1007/s12264-018-0219-5
Van Meir EG, Hadjipanayis CG, Norden AD, Shu HK, Wen PY, Olson JJ (2010) Exciting new advances in neuro-oncology: the avenue to a cure for malignant glioma. CA Cancer J Clin 60:166–193. https://doi.org/10.3322/caac.20069
Xie RL, Gupta S, Miele A, Shiffman D, Stein JL, Stein GS, van Wijnen AJ (2003) The tumor suppressor interferon regulatory factor 1 interferes with SP1 activation to repress the human CDK2 promoter. J Biol Chem 278:26589–26596. https://doi.org/10.1074/jbc.M301491200
Xu J, Li P (2016) Expression of EAP1 and CUX1 in the hypothalamus of female rats and relationship with KISS1 and GnRH. Endocr J 63:681–690. https://doi.org/10.1507/endocrj.EJ16-0123
Xu H, He JH, Xiao ZD, Zhang QQ, Chen YQ, Zhou H, Qu LH (2010) Liver-enriched transcription factors regulate microRNA-122 that targets CUTL1 during liver development. Hepatology 52:1431–1442. https://doi.org/10.1002/hep.23818
Zhang L, Zhang W, Li Y, Alvarez A, Li Z, Wang Y, Song L, Lv D, Nakano I, Hu B, Cheng SY, Fen H (2016) SHP-2-upregulated ZEB1 is important for PDGFRalpha-driven glioma epithelial-mesenchymal transition and invasion in mice and humans. Oncogene 35:5641–5652. https://doi.org/10.1038/onc.2016.100
Zhang Y, Xie P, Wang X, Pan P, Wang Y, Zhang H, Dong Y, Shi Y, Jiang Y, Yu R, Zhou X (2018) YAP promotes migration and invasion of human glioma cells. J Mol Neurosci 64:262–272. https://doi.org/10.1007/s12031-017-1018-6
Funding
This work was supported by the Natural Science Foundation of Shandong Province (No. ZR2015HL044 and No. ZR2014HL069).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Written informed consent was obtained from all patients, and the study was approved by the Ethics Committee of Linyi People’s Hospital.
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, X., Feng, F., Yang, C. et al. Upregulated Expression of CUX1 Correlates with Poor Prognosis in Glioma Patients: a Bioinformatic Analysis. J Mol Neurosci 69, 527–537 (2019). https://doi.org/10.1007/s12031-019-01355-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-019-01355-3