Abstract
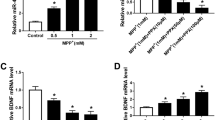
Parkinson’s disease (PD), the second most prevalent neurodegenerative disorder with only symptomatic treatment available, is characterized by a progressive loss of dopaminergic neurons in the midbrain. Ample evidence indicated that microRNAs (miRs) could regulate post-transcriptional gene expression and neuronal disease. In the present study, we have evaluated the effects and mechanism of miR-22 in PC12 pheochromocytoma cells treated with 6-hydroxydopamine (6-OHDA) to mimic PD. RT-PCR results showed that the expression of miR-22 is downregulated in 6-OHDA-treated PC12 cells, and the overexpression of miR-22 significantly promoted the survival and proliferation of 6-OHDA-induced PC12 cells, whereas miR-22 inhibitor reversed these effects. In addition, PC12 cells were treated with miR-22 mimics or inhibitor following 6-OHDA administration, which medicated ROS production and upregulation or downregulation of caspase-3 activity, respectively. A luciferase reporter assay revealed that transient receptor potential melastatin 7 (TRPM7) is a direct target gene of miR-22, and miR-22 overexpression markedly downregulated the level of TRPM7. Strikingly, further analysis showed that miR-22 mediated 6-OHDA-induced PC12 cell survival and proliferation by targeting TRPM7. Taken together, the present study showed that miR-22 overexpression exhibited neuroprotective and reversal effects on the 6-OHDA-induced PC12 cell growth and apoptosis by targeting TRPM7.




Similar content being viewed by others
References
Aarts M, Iihara K, Wei W-L, Xiong Z-G, Arundine M, Cerwinski W, MacDonald JF, Tymianski M (2003) A key role for TRPM7 channels in anoxic neuronal death. Cell 115:863–877
Alvarez-Erviti L, Seow Y, Schapira AH, Rodriguez-Oroz MC, Obeso JA, Cooper J (2013) Influence of microRNA deregulation on chaperone-mediated autophagy and α-synuclein pathology in Parkinson’s disease. Cell Death Dis 4:e545
Asrar S, Aarts M (2013) TRPM7, the cytoskeleton and neuronal death. Channels 7:6–16
Berenguer J, Herrera A, Vuolo L, Torroba B, Llorens F, Sumoy L, Pons S (2013) MicroRNA 22 regulates cell cycle length in cerebellar granular neuron precursors. Mol Cell Biol 33:2706–2717
Brichta L, Greengard P, Flajolet M (2013) Advances in the pharmacological treatment of Parkinson’s disease: targeting neurotransmitter systems. Trends Neurosci 36:543–554
Cheng B, Guo Y, Li C, Ji B, Pan Y, Chen J, Bai B (2014) Edaravone protected PC12 cells against MPP (+)-cytoxicity via inhibiting oxidative stress and up-regulating heme oxygenase-1 expression. J Neurol Sci 343:115–119
Cho HJ, Liu G, Jin SM, Parisiadou L, Xie C, Yu J, Sun L, Ma B, Ding J, Vancraenenbroeck R (2013) MicroRNA-205 regulates the expression of Parkinson’s disease-related leucine-rich repeat kinase 2 protein. Hum Mol Genet 22:608–620
Choi PS, Zakhary L, Choi W-Y, Caron S, Alvarez-Saavedra E, Miska EA, McManus M, Harfe B, Giraldez AJ, Horvitz RH (2008) Members of the miRNA-200 family regulate olfactory neurogenesis. Neuron 57:41–55
Chong C-M, Zhou Z-Y, Razmovski-Naumovski V, Cui G-Z, Zhang L-Q, Sa F, Hoi P-M, Chan K, Lee SM-Y (2013a) Danshensu protects against 6-hydroxydopamine-induced damage of PC12 cells in vitro and dopaminergic neurons in zebrafish. Neurosci Lett 543:121–125
Chong CM, Zhou ZY, Razmovski-Naumovski V, Cui GZ, Zhang LQ, Sa F, Hoi PM, Chan K, Lee SM (2013b) Danshensu protects against 6-hydroxydopamine-induced damage of PC12 cells in vitro and dopaminergic neurons in zebrafish. Neurosci Lett 543:121–125
Cohen G, Heikkila RE (1974) The generation of hydrogen peroxide, superoxide radical, and hydroxyl radical by 6-hydroxydopamine, dialuric acid, and related cytotoxic agents. J Biol Chem 249:2447–2452
de Mena L, Coto E, Cardo LF, Díaz M, Blázquez M, Ribacoba R, Salvador C, Pastor P, Samaranch L, Moris G (2010) Analysis of the micro-RNA-133 and PITX3 genes in Parkinson’s disease. Am J Med Genet B Neuropsychiatr Genet 153:1234–1239
Eacker SM, Dawson TM, Dawson VL (2009) Understanding microRNAs in neurodegeneration. Nat Rev Neurosci 10:837–841
Filipowicz W, Bhattacharyya SN, Sonenberg N (2008) Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 9:102–114
Harraz MM, Dawson TM, Dawson VL (2011) MicroRNAs in Parkinson’s disease. J Chem Neuroanat 42:127–130
Hensley K, Maidt ML, Yu Z, Sang H, Markesbery WR, Floyd RA (1998) Electrochemical analysis of protein nitrotyrosine and dityrosine in the Alzheimer brain indicates region-specific accumulation. J Neurosci 18:8126–8132
Jeon BS, Jackson-Lewis V, Burke RE (1995) 6-Hydroxydopamine lesion of the rat substantia nigra: time course and morphology of cell death. Neurodegeneration 4:131–137
Jiang H, Tian S-L, Zeng Y, Li L-L, Shi J (2008) TrkA pathway (s) is involved in regulation of TRPM7 expression in hippocampal neurons subjected to ischemic-reperfusion and oxygen–glucose deprivation. Brain Res Bull 76:124–130
Jovicic A, Zaldivar Jolissaint JF, Moser R, Silva Santos Mde F, Luthi-Carter R (2013a) MicroRNA-22 (miR-22) overexpression is neuroprotective via general anti-apoptotic effects and may also target specific Huntington’s disease-related mechanisms. PLoS One 8:e54222
Jovicic A, Zaldivar Jolissaint JF, Moser R, Silva Santos Mde F, Luthi-Carter R (2013b) MicroRNA-22 (miR-22) overexpression is neuroprotective via general anti-apoptotic effects and may also target specific Huntington’s disease-related mechanisms. PLoS One 8:17
Kim J, Inoue K, Ishii J, Vanti WB, Voronov SV, Murchison E, Hannon G, Abeliovich A (2007) A MicroRNA feedback circuit in midbrain dopamine neurons. Science 317:1220–1224
Liu L, Jiang Y, Zhang H, Greenlee AR, Yu R, Yang Q (2010) miR-22 functions as a micro-oncogene in transformed human bronchial epithelial cells induced by anti-benzo [a] pyrene-7, 8-diol-9, 10-epoxide. Toxicol in Vitro 24:1168–1175
Margis R, Rieder CR (2011) Identification of blood microRNAs associated to Parkinsonis disease. J Biotechnol 152:96–101
Margis R, Margis R, Rieder CR (2011) Identification of blood microRNAs associated to Parkinsońs disease. J Biotechnol 152:96–101
Miñones-Moyano E, Porta S, Escaramís G, Rabionet R, Iraola S, Kagerbauer B, Espinosa-Parrilla Y, Ferrer I, Estivill X, Martí E (2011) MicroRNA profiling of Parkinson’s disease brains identifies early downregulation of miR-34b/c which modulate mitochondrial function. Hum Mol Genet 20:3067–3078
Monteilh-Zoller MK, Hermosura MC, Nadler MJ, Scharenberg AM, Penner R, Fleig A (2003) TRPM7 provides an ion channel mechanism for cellular entry of trace metal ions. J Gen Physiol 121:49–60
Montell C, Birnbaumer L, Flockerzi V (2002) The TRP channels, a remarkably functional family. Cell 108:595–598
Mouradian MM (2012) MicroRNAs in Parkinson’s disease. Neurobiol Dis 46:279–284
Shi L, Chen J, Yang J, Pan T, Zhang S, Wang Z (2010) MiR-21 protected human glioblastoma U87MG cells from chemotherapeutic drug temozolomide induced apoptosis by decreasing Bax/Bcl-2 ratio and caspase-3 activity. Brain Res 1352:255–264
Shinde SMS, Mohsen G, et al. (2015) Biofluid-based microRNA biomarkers for Parkinson’s disease: an overview and update. AIMS. Med Sci 2:15–25
Sun H-S, Jackson MF, Martin LJ, Jansen K, Teves L, Cui H, Kiyonaka S, Mori Y, Jones M, Forder JP (2009) Suppression of hippocampal TRPM7 protein prevents delayed neuronal death in brain ischemia. Nat Neurosci 12:1300–1307
Tolleson CM, Fang JY (2013) Advances in the mechanisms of Parkinson’s disease. Discov Med 15:61–66
Tonelli DDP, Pulvers JN, Haffner C, Murchison EP, Hannon GJ, Huttner WB (2008) miRNAs are essential for survival and differentiation of newborn neurons but not for expansion of neural progenitors during early neurogenesis in the mouse embryonic neocortex. Development 135:3911–3921
Wang G, van der Walt JM, Mayhew G, Li Y-J, Züchner S, Scott WK, Martin ER, Vance JM (2008) Variation in the miRNA-433 binding site of FGF20 confers risk for Parkinson disease by overexpression of α-synuclein. Am J Hum Genet 82:283–289
Wei W-L, Sun H-S, Olah ME, Sun X, Czerwinska E, Czerwinski W, Mori Y, Orser BA, Xiong Z-G, Jackson MF (2007) TRPM7 channels in hippocampal neurons detect levels of extracellular divalent cations. Proc Natl Acad Sci 104:16323–16328
Woodgate A, MacGibbon G, Walton M, Dragunow M (1999) The toxicity of 6-hydroxydopamine on PC12 and P19 cells. Brain Res Mol Brain Res 69:84–92
Yang J, Chen L, Yang J, Ding J, Li S, Wu H, Zhang J, Fan Z, Dong W, Li X (2014) MicroRNA-22 targeting CBP protects against myocardial ischemia–reperfusion injury through anti-apoptosis in rats. Mol Biol Rep 41:555–561
Ye Y, Perez-Polo JR, Qian J, Birnbaum Y (2011) The role of microRNA in modulating myocardial ischemia-reperfusion injury. Physiol Genomics 43:534–542
Zhang Z, Li G, Szeto SS, Chong CM, Quan Q, Huang C, Cui W, Guo B, Wang Y, Han Y, et al. (2015) Examining the neuroprotective effects of protocatechuic acid and chrysin on in vitro and in vivo models of Parkinson disease. Free Radic Biol Med 84:331–343
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Supplementary fig 1
miR-22 overexpression reversed the effects of 6-OHDA on SH-SY5Y cell survival and proliferation. (A) miR-22 significantly increased 6-OHDA (100 μM) induced SH-SY5Y cell death. Cell viability was measured by MTT assay; (B) PC12 cell proliferation was measured by CCK-8 assay. Experiments were repeated three times. Data are presented as mean ± SD, N = 3. * P < 0.05 vs control, # P < 0.05 vs 6-OHDA group. (GIF 70 kb)
Rights and permissions
About this article
Cite this article
Yang, C.P., Zhang, Z.H., Zhang, L.H. et al. Neuroprotective Role of MicroRNA-22 in a 6-Hydroxydopamine-Induced Cell Model of Parkinson’s Disease via Regulation of Its Target Gene TRPM7. J Mol Neurosci 60, 445–452 (2016). https://doi.org/10.1007/s12031-016-0828-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-016-0828-2