Abstract
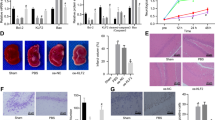
Hypoxia-inducible factor-1α (HIF-1α), a well-identified hypoxia-related protein, is involved in regulating the biological functions of various cell types including neurons. The traditional biological function of HIF-1α is promoting the transcription of some pro-survival genes when exposing to low oxygen conditions. Meanwhile, some studies also point out that HIF-1α shows the detrimental role in several central nervous system (CNS) disorders. Up to now, the knowledge of HIF-1α function in CNS is still limited. To investigate whether HIF-1α is involved in CNS impairment and repair, we employed a traumatic brain injury model in adult rats. Upregulation of HIF-1α was observed in the injured brain cortex by western blot analysis and immunohistochemistry staining. Terminal deoxynucleotidyl transferase deoxy-UTP nick-end labeling (TUNEL) and 4′,6-diamidino-2-phenylindole (DAPI) staining suggested that HIF-1α was relevant to neuronal apoptosis after brain injury. In addition, glutamate excitotoxic model of primary cortex neurons was introduced to further investigate the role of HIF-1α in neuronal apoptosis; the result implied HIF-1α was associated with the regulation of p53 and BNIP3 in the apoptotic neurons. Based on our data, we suggested that HIF-1α might play an important role in neuronal apoptosis after traumatic brain injury in rat, which might also provide a basis for the further study on its role in regulating the transcription of target genes in apoptotic neurons, and might gain a novel strategy for the clinical therapy for traumatic brain injury.







Similar content being viewed by others
References
Agostinho P, Oliveira CR (2003) Involvement of calcineurin in the neurotoxic effects induced by amyloid-beta and prion peptides. Eur J Neurosci 17:1189–96
Althaus J, Bernaudin M, Petit E, Toutain J, Touzani O, Rami A (2006) Expression of the gene encoding the pro-apoptotic BNIP3 protein and stimulation of hypoxia-inducible factor-1alpha (HIF-1alpha) protein following focal cerebral ischemia in rats. Neurochem Int 48:687–95
Badawi Y, Ramamoorthy P, Shi H (2012) Hypoxia-inducible factor 1 protects hypoxic astrocytes against glutamate toxicity. ASN Neuro 4:231–41
Bruick RK (2000) Expression of the gene encoding the proapoptotic Nip3 protein is induced by hypoxia. Proc Natl Acad Sci U S A 97:9082–7
Chen S, Pickard JD, Harris NG (2003) Time course of cellular pathology after controlled cortical impact injury. Exp Neurol 182:87–102
Cregan SP, MacLaurin JG, Craig CG, Robertson GS, Nicholson DW, Park DS, Slack RS (1999) Bax-dependent caspase-3 activation is a key determinant in p53-induced apoptosis in neurons. J Neurosci 19:7860–9
Di Giovanni S, Knights CD, Rao M, Yakovlev A, Beers J, Catania J, Avantaggiati ML, Faden AI (2006) The tumor suppressor protein p53 is required for neurite outgrowth and axon regeneration. EMBO J 25:4084–96
Ding JY, Kreipke CW, Speirs SL, Schafer P, Schafer S, Rafols JA (2009) Hypoxia-inducible factor-1alpha signaling in aquaporin upregulation after traumatic brain injury. Neurosci Lett 453:68–72
Endo H, Kamada H, Nito C, Nishi T, Chan PH (2006) Mitochondrial translocation of p53 mediates release of cytochrome c and hippocampal CA1 neuronal death after transient global cerebral ischemia in rats. J Neurosci 26:7974–83
Feng JF, Gurkoff GG, Van KC, Song M, Lowe DA, Zhou J, Lyeth BG (2012) NAAG peptidase inhibitor reduces cellular damage in a model of TBI with secondary hypoxia. Brain Res 1469:144–52
Harris NG, Carmichael ST, Hovda DA, Sutton RL (2009) Traumatic brain injury results in disparate regions of chondroitin sulfate proteoglycan expression that are temporally limited. J Neurosci Res 87:2937–50
Ha YM, Kim MY, Park MK, Lee YS, Kim YM, Kim HJ, Lee JH, Chang KC (2012) Higenamine reduces HMGB1 during hypoxia-induced brain injury by induction of heme oxygenase-1 through PI3K/Akt/Nrf-2 signal pathways. Apoptosis 17:463–74
Hota KB, Hota SK, Srivastava RB, Singh SB (2012) Neuroglobin regulates hypoxic response of neuronal cells through Hif-1alpha- and Nrf2-mediated mechanism. J Cereb Blood Flow Metab 32:1046–60
Jeremitsky E, Omert L, Dunham CM, Protetch J, Rodriguez A (2003) Harbingers of poor outcome the day after severe brain injury: hypothermia, hypoxia, and hypoperfusion. J Trauma 54:312–9
Jones PA, Andrews PJ, Midgley S, Anderson SI, Piper IR, Tocher JL, Housley AM, Corrie JA, Slattery J, Dearden NM et al (1994) Measuring the burden of secondary insults in head-injured patients during intensive care. J Neurosurg Anesthesiol 6:4–14
Kan EM, Ling EA, Lu J (2012) Microenvironment changes in mild traumatic brain injury. Brain Res Bull 87:359–72
Kaya SS, Mahmood A, Li Y, Yavuz E, Goksel M, Chopp M (1999) Apoptosis and expression of p53 response proteins and cyclin D1 after cortical impact in rat brain. Brain Res 818:23–33
Keane RW, Kraydieh S, Lotocki G, Alonso OF, Aldana P, Dietrich WD (2001) Apoptotic and antiapoptotic mechanisms after traumatic brain injury. J Cereb Blood Flow Metab 21:1189–98
Kim TH, Hur EG, Kang SJ, Kim JA, Thapa D, Lee YM, Ku SK, Jung Y, Kwak MK (2011) NRF2 blockade suppresses colon tumor angiogenesis by inhibiting hypoxia-induced activation of HIF-1alpha. Cancer Res 71:2260–75
Maas AI, Stocchetti N, Bullock R (2008) Moderate and severe traumatic brain injury in adults. Lancet Neurol 7:728–41
Mabjeesh NJ, Escuin D, LaVallee TM, Pribluda VS, Swartz GM, Johnson MS, Willard MT, Zhong H, Simons JW, Giannakakou P (2003) 2ME2 inhibits tumor growth and angiogenesis by disrupting microtubules and dysregulating HIF. Cancer Cell 3:363–75
Mangat HS (2012) Severe traumatic brain injury. Continuum (Minneap Minn) 18:532–46
Mannix RC, Whalen MJ (2012) Traumatic brain injury, microglia, and Beta amyloid. Int J Alzheimers Dis 2012:608732
McHugh GS, Engel DC, Butcher I, Steyerberg EW, Lu J, Mushkudiani N, Hernandez AV, Marmarou A, Maas AI, Murray GD (2007) Prognostic value of secondary insults in traumatic brain injury: results from the IMPACT study. J Neurotrauma 24:287–93
Mellergard P, Sjogren F, Hillman J (2012) The cerebral extracellular release of glycerol, glutamate, and FGF2 is increased in older patients following severe traumatic brain injury. J Neurotrauma 29:112–8
Pearson WS, Sugerman DE, McGuire LC, Coronado VG (2012) Emergency department visits for traumatic brain injury in older adults in the United States: 2006–08. West J Emerg Med 13:289–93
Piret JP, Mottet D, Raes M, Michiels C (2002) Is HIF-1alpha a pro- or an anti-apoptotic protein? Biochem Pharmacol 64:889–92
Prabhakaran K, Li L, Zhang L, Borowitz JL, Isom GE (2007) Upregulation of BNIP3 and translocation to mitochondria mediates cyanide-induced apoptosis in cortical cells. Neuroscience 150:159–67
Risdall JE, Menon DK (2011) Traumatic brain injury. Philos Trans R Soc Lond B Biol Sci 366:241–50
Sermeus A, Michiels C (2011) Reciprocal influence of the p53 and the hypoxic pathways. Cell Death Dis 2:e164
Shenaq M, Kassem H, Peng C, Schafer S, Ding JY, Fredrickson V, Guthikonda M, Kreipke CW, Rafols JA, Ding Y (2012) Neuronal damage and functional deficits are ameliorated by inhibition of aquaporin and HIF1alpha after traumatic brain injury (TBI). J Neurol Sci 323:134–40
Shi H (2009) Hypoxia inducible factor 1 as a therapeutic target in ischemic stroke. Curr Med Chem 16:4593–600
Stoica BA, Faden AI (2010) Cell death mechanisms and modulation in traumatic brain injury. Neurotherapeutics 7:3–12
Tedeschi A, Nguyen T, Puttagunta R, Gaub P, Di Giovanni S (2009) A p53-CBP/p300 transcription module is required for GAP-43 expression, axon outgrowth, and regeneration. Cell Death Differ 16:543–54
Trollmann R, Gassmann M (2009) The role of hypoxia-inducible transcription factors in the hypoxic neonatal brain. Brain Dev 31:503–9
Vazquez-Valls E, Flores-Soto ME, Chaparro-Huerta V, Torres-Mendoza BM, Gudino-Cabrera G, Rivera-Cervantes MC, Pallas M, Camins A, Armendariz-Borunda J, Beas-Zarate C (2011) HIF-1alpha expression in the hippocampus and peripheral macrophages after glutamate-induced excitotoxicity. J Neuroimmunol 238:12–8
Yan EB, Hellewell SC, Bellander BM, Agyapomaa DA, Morganti-Kossmann MC (2011) Post-traumatic hypoxia exacerbates neurological deficit, neuroinflammation and cerebral metabolism in rats with diffuse traumatic brain injury. J Neuroinflammation 8:147
Zhang HM, Cheung P, Yanagawa B, McManus BM, Yang DC (2003) BNips: a group of pro-apoptotic proteins in the Bcl-2 family. Apoptosis 8:229–36
Zhang Z, Shi R, Weng J, Xu X, Li XM, Gao TM, Kong J (2011) The proapoptotic member of the Bcl-2 family Bcl-2 / E1B-19K-interacting protein 3 is a mediator of caspase-independent neuronal death in excitotoxicity. FEBS J 278:134–42
Acknowledgements
The project was funded by the Open Science Foundation of the Jiangsu Province (KF200953).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, A., Sun, X., Ni, Y. et al. HIF-1α Involves in Neuronal Apoptosis after Traumatic Brain Injury in Adult Rats. J Mol Neurosci 51, 1052–1062 (2013). https://doi.org/10.1007/s12031-013-0084-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-013-0084-7