Abstract
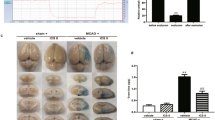
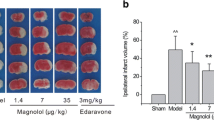
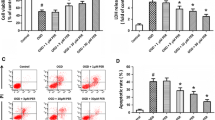
The strategy for the development of effective and safe neuroprotective agents has great potential to reduce cerebral ischemia–reperfusion injury and improve the functional outcome in stroke patients. Recently, doxycycline, a tetracycline antibiotic, has been shown to have neuroprotective efficiency in reduction of a variety of ischemia–reperfusion injuries as well as ischemic brain damage. We used the rat models of middle cerebral artery occlusion (MCAO) and reperfusion to investigate the effects of treatments with doxycycline against the blood-brain barrier (BBB) leakage at 3, 12, 72, and 120 h of reperfusion. Male Sprague–Dawley rats were subjected to MCAO for 2 h followed by reperfusion for 3, 12, 72, and 120 h and received either doxycycline (45 mg/kg) or saline. The results showed that the treatment of doxycycline significantly reduced the BBB leakage and cerebral infarct volume, which were proved by Evans blue assay and TTC staining. Real-time PCR, immunohistochemistry, and western blot assay verified that the administration of doxycycline significantly up-regulated the expression of tight junction claudin-5, occludin, and ZO-1 from 3 to 120 h after reperfusion. The results of real-time PCR, western blot, and gelatin zymography analyses revealed that the gene and protein expression and activities of matrix metalloproteinases (MMPs) MMP-2 and MMP-9 were significantly elevated in a different time-dependent manner after ischemia–reperfusion but significantly inhibited by doxycycline treatment. Moreover, doxycycline could also significantly down-regulate the expression of PKCδ mRNA and protein after ischemia–reperfusion. These results suggested that the protective effects of doxycycline against BBB damage induced by reperfusion might be related to the up-regulation of tight junction proteins and inhibition of MMP-2, MMP-9, and PKCδ.








Similar content being viewed by others
References
Angelow S, Zeni P, Höhn B, Galla HJ (2005) Phorbol ester induced short-and long-term permeabilization of the blood–CSF barrier in vitro. Brain Res 1063:168–179
Ballabh P, Braun A, Nedergaard M (2004) The blood-brain barrier: an overview: structure, regulation, and clinical implications. Neurobiol Dis 6:1–13
Bauer AT, Bürgers HF, Rabie T, Marti HH (2010) Matrix metalloproteinase-9 mediates hypoxia-induced vascular leakage in the brain via tight junction rearrangement. J Cereb Blood Flow Metab 30:837–848
Bazzoni G, Dejana E (2004) Endothelial cell-to-cell junctions: molecular organization and role in vascular homeostasis. Physiol Rev 84:869–901
Biegel D, Spencer DD, Pachter JS (1995) Isolation and culture of human brain microvessel endothelial cells for the study of blood-brain barrier properties in vitro. Brain Res 692:183–189
Bright R, Mochly-Rosen D (2005) The role of protein kinase C in cerebral ischemic and reperfusion Injury. Stroke 36:2781–2790
Bright R, Raval AP, Dembner JM, Pérez-Pinzón MA, Steinberg GK, Yenari MA, Mochly-Rosen D (2004) Protein kinase C delta mediates cerebral reperfusion injury in vivo. J Neurosci 24:6880–6888
Castro MM, Kandasamy AD, Youssef N, Schulz R (2011) Matrix metalloproteinase inhibitor properties of tetracyclines: therapeutic potential in cardiovascular diseases. Pharmacol Res. May 31. [Epub ahead of print]
Chakraborti S, Mandal A, Das S, Chakraborti T (2005) Role of MMP-2 in PKCδ-mediated inhibition of Na+ dependent Ca2+ uptake in microsomes of pulmonary smooth muscle: involvement of a pertussis toxin sensitive protein. Mol Cell Biochem 280:107–117
Chou WH, Messing RO (2008) Hypertensive encephalopathy and the blood-brain barrier: is δPKC a gatekeeper? J Clin Invest 118:17–20
Clark WM, Lessov N, Lauten JD, Hazel K (1997) Doxycycline treatment reduces ischemic brain damage in transient middle cerebral artery occlusion in the rat. J Mol Neurosci 9:103–108
Collino M, Aragno M, Mastrocola R, Gallicchio M, Rosa AC, Dianzani C, Danni O, Thiemermann C, Fantozzi R (2006) Modulation of the oxidative stress and inflammatory response by PPAR-gamma agonists in the hippocampus of rats exposed to cerebral ischemia/reperfusion. Eur J Pharmacol 530:70–80
Elkind MS (2009) Outcomes after stroke: risk of recurrent ischemic stroke and other events. Am J Med 122:S7–S13
Fainaru O, Adini I, Benny O, Bazinet L, Pravda E, D'Amato R, Folkman J (2008) Doxycycline induces membrane expression of VEcadherin on endothelial cells and prevents vascular hyperpermeability. FASEB J 22:3728–3735
Fukuda S, Fini CA, Mabuchi T, Koziol JA, Eggleston LL Jr, del Zoppo GJ (2004) Focal cerebral ischemia induces active proteases that degrade microvascular matrix. Stroke 35:998–1004
Jiao H, Wang Z, Liu Y, Xue Y (2011) Specific role of tight junction protein claudin-5, occludin, and ZO-1 of the blood-brain barrier in a focal cerebral ischemic insult. J Mol Neurosci 44:130–139
Jung JE, Kim GS, Chen H, Maier CM, Narasimhan P, Song YS, Niizuma K, Katsu M, Okami N, Yoshioka H, Sakata H, Goeders CE, Chan PH (2010) Reperfusion and neurovascular dysfunction in stroke: from basic mechanisms to potential strategies for neuroprotection. Mol Neurobiol 41:172–179
Kago T, Takagi N, Date I, Takenaga Y, Takagi K, Takeo S (2006) Cerebral ischemia enhances tyrosine phosphorylation of occludin in brain capillaries. Biochem Biophys Res Commun 339:1197–1203
Koistinaho M, Malm TM, Kettunen MI, Goldsteins G, Starckx S, Kauppinen RA, Opdenakker G, Koistinaho J (2005) Minocycline protects against permanent cerebral ischemia in wild type but not in matrix metalloprotease-9-deficient mice. J Cereb Blood Flow Metab 25:460–467
Latour LL, Kang DW, Ezzeddine MA, Chalela JA, Warach S (2004) Early blood-brain barrier disruption in human focal brain ischemia. Ann Neurol 56:468–477
Lin CW, Hou WC, Shen SC, Juan SH, Ko CH, Wang LM, Chen YC (2008) Quercetin inhibition of tumor invasion via suppressing PKCδ/ERK/AP-1-dependent matrix metalloproteinase-9 activation in breast carcinoma cells. Carcinogenesis 29:1807–1815
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
Miettinen S, Roivainen R, Keinänen R, Hökfelt T, Koistinaho J (1996) Specific induction of protein kinase C delta subspecies after transient middle cerebral artery occlusion in the rat brain: inhibition by MK-801. J Neurosci 16:6236–6245
Porter KE, Thompson MM, Loftus IM, McDermott E, Jones L, Crowther M, Bell PR, London NJ (1999) Production and inhibition of the gelatinolytic matrix metalloproteinases in a human model of vein graft stenosis. Eur J Vasc Endovasc Surg 17:404–412
Roach DM, Fitridge RA, Laws PE, Millard SH, Varelias A, Cowled PA (2002) Up-regulation of MMP-2 and MMP-9 leads to degradation of type IV collagen during skeletal muscle reperfusion injury; protection by the MMP inhibitor, doxycycline. Eur J Vasc Endovasc Surg 23:260–269
Rosenberg GA, Navratil M, Barone F, Feuerstein G (1996) Proteolytic cascade enzymes increase in focal cerebral ischemia in rat. J Cereb Blood Flow Metab 16:360–366
Rosenberg GA, Estrada EY, Dencoff JE (1998) Matrix metalloproteinases and TIMPs are associated with blood-brain barrier opening after reperfusion in rat brain. Stroke 29:2189–2195
Shimakura A, Kamanaka Y, Ikeda Y, Kondo K, Suzuki Y, Umemura K (2000) Neutrophil elastase inhibition reduces cerebral ischemic damage in the middle cerebral artery occlusion. Brain Res 858:55–60
Smith JR, Gabler WL (1995) Protective effects of doxycycline in mesenteric ischemia and reperfusion. Res Commun Mol Pathol Pharmacol 88:303–315
Strbian D, Karjalainen-Lindsberg ML, Kovanen PT, Tatlisumak T, Lindsberg PJ (2007) Mast cell stabilization reduces hemorrhage formation and mortality after administration of thrombolytics in experimental ischemic stroke. Circulation 116:411–418
Tsukita S, Furuse M (1998) Overcoming barriers in the study of tight junction function: from occludin to claudin. Genes Cells 3:569–573
Wang ZH, Xue YX, Liu YH (2010) The modulation of protein kinase A and heat shock protein 70 is involved in the reversible increase of blood-brain tumor barrier permeability induced by papaverine. Brain Res Bull 83:367–373
Yang GY, Betz AL (1994) Reperfusion-induced injury to the blood-brain barrier after middle cerebral artery occlusion in rats. Stroke 25:1658–1665
Yi JH, Park SW, Kapadia R, Vemuganti R (2007) Role of transcription factors in mediating post-ischemic cerebral inflammation and brain damage. Neurochem Int 50:1014–1027
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (Nos. 30872656, 30973079, 81172197, 81171131, and 81072056), the special fund for Scientific Research of Doctor-degree Subjects in Colleges and Universities (Nos. 20092104110015 and 20102104110009), and Shenyang Science and Technology Plan Projects (Nos. F10-205-1-22 and F10-205-1-37).
Conflict of Interest Statement
The authors declare that they have no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhenhua Wang, Yixue Xue, and Haixia Jiao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, Z., Xue, Y., Jiao, H. et al. Doxycycline-Mediated Protective Effect Against Focal Cerebral Ischemia–Reperfusion Injury Through the Modulation of Tight Junctions and PKCδ Signaling in Rats. J Mol Neurosci 47, 89–100 (2012). https://doi.org/10.1007/s12031-011-9689-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-011-9689-x