Abstract
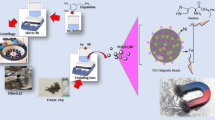
Thermoresponsive magnetic nanoparticles with an upper critical solution temperature (UCST) in aqueous solution were synthesized for the first time. Named Therma-Max, the material was synthesized by redox copolymerization of N-acryloyl glycinamide with a monomer form of biotin using methacrylated dextran-magnetite. While the resulting Therma-Max was completely dispersed at temperatures above the UCST (18°C) and could not be separated by a permanent magnet, it was rapidly flocculated when the temperature fell below the UCST and was easily separated by a permanent magnet. The flocculated particles dispersed completely when the temperature was raised to above the UCST. Because biotin was immobilized on the Therma-Max, avidin and antibodies were subsequently immobilized with good efficiency. Furthermore, transiently transfected Arabidopsis protoplasts, which have surface display of CD4 antigen, were efficiently captured and enriched by using a biotinylated anti-CD4 antibody in combination with avidin-conjugated Therma-Max. Also, the silkworm storage protein (SP2) was efficiently separated from the silkworm hemolymph by using biotinylated anti-IgG antibody and anti-SP2 antibody in combination with avidin-conjugated Therma-Max. In both cases, it was confirmed that specificity and adsorption capacity were markedly improved by converting the conventional micro-size fine magnetic particles to nano-size particles. These results show the potential of Therma-Max with a UCST in bioaffinity separation of cells and biomolecules.
Similar content being viewed by others
References
Dunnill, P. and Lilly, M. D. (1974), Biotechnol. 16, 987–990.
Sada, E., Katoh, S., and Terashima, M. (1981), Biotechnol. Bioeng. 23, 1037–1044.
Setchell, C. H. (1985), J. Chem. Technol. Biotechnol. 35B, 175–182.
Uhlen, M. (1989), Nature 340, 733, 734.
Nakamura, N., Hashimoto, K., and Matsunaga, T. (1991), Anal. Chem. 63, 268–272.
Shinkai, M., Honda, H., and Kobayashi, T. (1991), Biocatalysis 5, 61–69.
Khng, H. P. et al. (1998), Biotechnol. Bioeng. 60, 419–424.
Seta, K. et al. (1998), Chromatography 19, 112, 113.
Welter, B. H. and Price, T. M. (1999), Biotechniques 27, 282–286.
Honda, H., Kawabe, A., Shinkai, M., and Kobayashi, T. (1999), Biochem. Eng. J. 3, 157–160.
Kondo, A., Kamura, H., and Higashitani, K. (1994), Appl. Microbiol. Biotechnol. 41, 99–105.
Kondo, A. and Fukuda, H. (1997), J. Ferment. Bioeng. 84, 337–341.
Heskins, M. and Guillet, J. E. (1968), J. Macromol. Sci. Chem. A2, 1441–1455.
Hoshino, F., Fujimoto, T., Kawaguchi, H., and Ohtsuka, Y. (1987), Polym. J. 19, 241–247.
Katono, H. et al. (1991), J.Controlled Release 16, 215–228.
Miltenyi, S., Muller, W., Weichel, W., & Radbruch, A. (1990), Cytometry 11, 231–238.
Dorr, I., Miltenyi, S., Salamini, F., and Uhrig, H. (1994), Biotechnology 12, 511–515.
Barth, S., Goerlich, R., and Schnabl, H. (1994), J. Biochem. Biophys. Methods 29, 83–86.
Kausch, A. P., Owen, Jr., T. P., Narayanswami, S., and Bruce, B. D. (1999), Biotechniques 26, 336–343.
Telfer, W., Keim, P., and Law, J. (1983), Insect Biochem. 13, 601–613.
Tojo, S., Nagata, M., and Kobayashi, M. (1980), Insect Biochem. 10, 289–303.
Haas, H. C. and Schuler, N. W. (1964), Polymer Letters 2, 1095–1096.
Axelos, M., Curie, C., Mazzolini, L., Bardet, C., and Lescure, B. (1992), Biochemistry 30, 123–128.
An, C. I., Sawada, A., Fukusaki, E., and Kobayashi, A. (2003), Biosci. Biotechnol. Biochem. 67, 2674–2677.
Kang, B. H., Busse, J. S., Dickey, C., Rancour, D. M., and Bednarek, S. Y. (2001), Plant Physiol. 126, 47–68.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ohnishi, N., Furukawa, H., Hideyuki, H. et al. High-efficiency bioaffinity separation of cells and proteins using novel thermoresponsive biotinylated magnetic nanoparticles. Nanobiotechnol 2, 43–49 (2006). https://doi.org/10.1007/s12030-006-0006-7
Issue Date:
DOI: https://doi.org/10.1007/s12030-006-0006-7