Abstract
Purpose
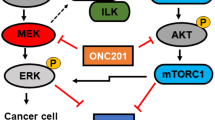
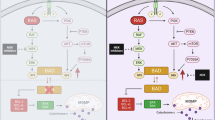
Over the last decades, cytotoxic chemotherapy has been the cornerstone of metastatic pancreatic adenocarcinoma treatment. In late-stage disease, a range of treatment regimens still offers minor benefits. Molecular profiling studies have shown that pancreatic adenocarcinoma (PDAC) is a mutation-driven tumor type, with KRAS mutations found in approximately 90% of cases, which could partially explain the resistance to chemotherapy. Preclinical data on selective targeting of a downstream point of the RAF–MEK–ERK pathway with a MEK inhibitor along with the concurrent use of an autophagy inhibitor such as hydroxychloroquine appears to be one alternative approach to overcome resistance and inhibit cell proliferation.
Methods
We herein aim to investigate the rationale of autophagy inhibitors use and describe the outcomes of patients who received this experimental treatment.
Results
Two patients have received this experimental regimen from January 2020 to the present date, achieving disease stabilization that is clinically meaningful, considering the chemoresistance scenario of the included patients.
Conclusions
Our real-life data regarding KRAS-mutated PDAC patients who received treatment with the MEK inhibitor trametinib combined with hydroxychloroquine after experiencing disease progression are consistent with the preclinical data, pointing to the clinical benefits of this regimen.







Similar content being viewed by others
Data Availability
Not applicable.
References
Saluja A, Dudeja V, Banerjee S. Evolution of novel therapeutic options for pancreatic cancer. Curr Opin Gastroenterol. 2016;32(5):401–7.
Conroy T, Desseigne F, Ychou M, Bouché O, Guimbaud R, Bécouarn Y, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364(19):1817–25.
Hoff V, Daniel D, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369(18):1691–703.
Waters AM, Der CJ. KRAS: the critical driver and therapeutic target for pancreatic cancer. Cold Spring Harbor perspectives in medicine. 2018;8(9):a031435.
Kinsey CG, Camolotto SA, Boespflug AM, Guillen KP, Foth M, Truong A, et al. Protective autophagy elicited by RAF→ MEK→ ERK inhibition suggests a treatment strategy for RAS-driven cancers. Nat Med. 2019;25(4):620–7.
Bryant KL, Stalnecker CA, Zeitouni D, Klomp JE, Peng S, Tikunov AP, et al. Combination of ERK and autophagy inhibition as a treatment approach for pancreatic cancer. Nat Med. 2019;25(4):628–40.
Magliano D, Pasca M, Logsdon CD. Roles for KRAS in pancreatic tumor development and progression. Gastroenterology. 2013;144(6):1220–9.
Spaargaren M, Bischoff JR, McCormick FRANK. Signal transduction by Ras-like GTPases: a potential target for anticancer drugs. Gene Expression The Journal of Liver Research. 1995;4(6):345–56.
Rosenfeldt MT, et al. p53 status determines the role of autophagy in pancreatic tumour development. Nature. 2013;504(7479):296–300.
Robert C, Grob JJ, Stroyakovskiy D, Karaszewska B, Hauschild A, Levchenko E, et al. Five-year outcomes with dabrafenib plus trametinib in metastatic melanoma. N Engl J Med. 2019;381(7):626–36.
Infante JR, Somer BG, Park JO, Li C-P, Scheulen ME, Kasubhai SM, et al. A randomised, double-blind, placebo-controlled trial of trametinib, an oral MEK inhibitor, in combination with gemcitabine for patients with untreated metastatic adenocarcinoma of the pancreas. Eur J Cancer. 2014;50(12):2072–81.
Mans LA, et al. The tumor suppressor LKB1 regulates starvation-induced autophagy under systemic metabolic stress. Sci Rep. 2017;7(1):1–10.
Shaw RJ, Kosmatka M, Bardeesy N, Hurley RL, Witters LA, DePinho RA, et al. The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc Natl Acad Sci. 2004;101(10):3329–35.
Janku F, et al. Autophagy as a target for anticancer therapy. Nature Reviews Clin Oncol. 2011;8(9):528–39.
Mauthe M, Orhon I, Rocchi C, Zhou X, Luhr M, Hijlkema K-J, et al. Chloroquine inhibits autophagic flux by decreasing autophagosome-lysosome fusion. Autophagy. 2018;14(8):1435–55.
Clinicaltrials.gov 2020. Trametinib and hydroxychloroquine in treating patients with pancreatic cancer - full text view - Clinicaltrials.Gov [online] Available at: <https://clinicaltrials.gov/ct2/show/NCT03825289> [Accessed 23 May 2020].
Author information
Authors and Affiliations
Contributions
All authors made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work; drafted the work or revised it critically for important intellectual content; approved the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
This case report was approved by the ethics committee of the Hospital Sírio-Libanês, São Paulo, Brazil.
Consent to Participate
The patients provided consent and gave permission to participate in the study.
Consent for Publication
The patients provided consent and gave permission to have her case, as well as relevant related workup and diagnostic images, presented in the medical literature.
Code Availability
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xavier, C.B., Marchetti, K.R., Castria, T.B. et al. Trametinib and Hydroxychloroquine (HCQ) Combination Treatment in KRAS-Mutated Advanced Pancreatic Adenocarcinoma: Detailed Description of Two Cases. J Gastrointest Canc 52, 374–380 (2021). https://doi.org/10.1007/s12029-020-00556-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-020-00556-z