Abstract
Purpose
Pancreatic cancer is one of the most fatal malignancies and the fourth leading cause of cancer-related mortality in the USA. Most clinical trials involving pancreatic adenocarcinoma (PAC) patients exclude subjects with a prior malignancy because of the possible effect of prior malignancies on survival. However, no data in the medical literature support this assumption. In this paper, we aim to study the impact of having a prior malignancy on the survival outcomes of stage IV PAC.
Methods
We used the surveillance, epidemiology, and end results database to review patients with stage IV PAC diagnosed between 1973 and 2014. We calculated overall and pancreatic cancer-specific survival of these patients using unadjusted Kaplan-Meier test and multivariable covariate-adjusted Cox models.
Results
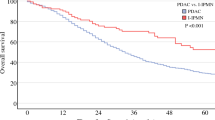
We reviewed 66,874 stage IV PAC patients, of which 4942 had a prior malignancy. Kaplan-Meier and Cox models showed that a history of prior malignancy did not cause significant difference in overall survival (HR = 0.938, 95%CI = 0.880–1.000, p = .052). However, a prior malignancy was associated with a better pancreatic cancer-specific survival (HR = 0.855, 95% CI = 0.796–0.918, p < .001).
Conclusion
A prior malignancy before stage IV PAC was not associated with worse survival outcomes. Researchers should take these results into consideration when including/excluding patients to improve the generalizability and accuracy of their results.

Similar content being viewed by others
References
American Cancer Society: Cancer Facts and Figures 2017. Atlanta, Ga: American Cancer Society, 2017. Available online at: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Last accessed July 22, 2017.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66(1):7–30.
National Cancer Institute. Estimated new cancer cases and deaths for 2013. SEER (Surveillance Epidemiology and End Results) cancer statistics review. Available from: http://www.seer.cancer.gov.
Lowenfels AB, Maisonneuve P. Epidemiology and prevention of pancreatic cancer. Jpn J Clin Oncol. 2004;34(5):238–44.
Mohammed S, Van Buren GI, Fisher WE. Pancreatic cancer: advances in treatment. World Journal of Gastroenterology: WJG. 2014;20(28):9354.
Laccetti AL, Pruitt SL, Xuan L, Halm EA, Gerber DE. Effect of prior cancer on outcomes in advanced lung cancer: implications for clinical trial eligibility and accrual. J Natl Cancer Inst. 2015;107
Tournoux C, Katsahian S, Chevret S, Levy V. Factors influencing inclusion of patients with malignancies in clinical trials. Cancer. 2006;106(2):258–70.
Van Spall HG, Toren A, Kiss A, Fowler RA. Eligibility criteria of randomized controlled trials published in high-impact general medical journals: a systematic sampling review. JAMA. 2007;297(11):1233–40.
Ries LA, Harkins D, Krapcho M, Mariotto A, Miller BA, Feuer EJ, Clegg LX, Eisner MP, Horner MJ, Howlader N, Hayat M. SEER cancer statistics review. 1975-2003.
Surveillance Research Program. National Cancer Institute SEER*Stat software (www.seer.cancer.gov/seerstat) version 8.3.4
Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence - SEER 18 Regs Custom Data (with additional treatment fields), November 2016 Sub (1973–2014 varying) - Linked To County Attributes - Total U.S., 1969–2015 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2017, based on the November 2016 submission.
Concato J, Shah N, Horwitz RI. Randomized, controlled trials, observational studies, and the hierarchy of research designs. N Engl J Med. 2000;342(25):1887–92.
Gross CP, Mallory R, Heiat A, Krumholz HM. Reporting the recruitment process in clinical trials: who are these patients and how did they get there? Ann Intern Med. 2002;137(1):10–6.
Lara PN Jr, Higdon R, Lim N, Kwan K, Tanaka M, Lau DH, et al. Prospective evaluation of cancer clinical trial accrual patterns: identifying potential barriers to enrollment. J Clin Oncol. 2001;19(6):1728–33.
Rothwell PM. External validity of randomised controlled trials:“to whom do the results of this trial apply?”. Lancet. 2005;365(9453):82–93.
Smyth EC, Tarazona N, Peckitt C, Armstrong E, Mansukhani S, Cunningham D, et al. Exclusion of gastrointestinal cancer patients with prior cancer from clinical trials: is this justified? Clin Colorectal Cancer. 2016;15(2):e53–e9.
Gerber DE, Laccetti AL, Xuan L, Halm EA, Pruitt SL. Impact of prior cancer on eligibility for lung cancer clinical trials. J Natl Cancer Inst. 2014;106(11):dju302.
Varty PP, Delrio P, Boulos PB. Survival in colorectal carcinoma associated with previous extracolonic cancer. Ann R Coll Surg Engl. 1994;76(3):180–4.
Pulte D, Gondos A, Brenner H. Long-term survival of patients diagnosed with non-Hodgkin lymphoma after a previous malignancy. Leuk Lymphoma. 2009;50(2):179–86.
Saad AM, Al-Husseini MJ, Elgebaly A, et al. Impact of prior malignancy on outcomes of stage IV esophageal carcinoma; SEER based study. Expert Review of Gastroenterology & Hepatology (IERH). 2018. https://doi.org/10.1080/17474124.2018.1426458.
Noone A-M, Lund JL, Mariotto A, Cronin K, McNeel T, Deapen D, et al. Comparison of SEER treatment data with Medicare claims. Med Care. 2016;54(9):e55–64.
James BYM, Gross CP, Wilson LD, Smith BD. NCI SEER public-use data: applications and limitations in oncology research. Oncology. 2009;23(3):288.
Talarico L, Chen G, Pazdur R. Enrollment of elderly patients in clinical trials for cancer drug registration: a 7-year experience by the US Food and Drug Administration. J Clin Oncol. 2004;22(22):4626–31.
Acknowledgments
We would like to thank Saad L. Janoudi for his help in data preparation and statistical analysis.
Role of Authors
All authors participated in designing the concept of the paper. AS and MA conducted the analysis of the data and had the access to the database. All authors have contributed to data interpretation and writing the paper. All authors have revised and agreed to the content of the paper. OA supervised the whole project scientifically and had final responsibility for the decision to submit for publication. MA managed and coordinated the research activity planning and execution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Al-Husseini, M.J., Saad, A.M., Turk, T. et al. Impact of Prior Malignancy on Survival Outcomes of Stage IV Pancreatic Adenocarcinoma: SEER-Based Cohort. J Gastrointest Canc 50, 794–800 (2019). https://doi.org/10.1007/s12029-018-0158-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-018-0158-4