Abstract
Purpose
Despite decades of research, consensus on optimal treatment for pancreatic cancer has not been reached and survival remains bleak. This study aimed to evaluate predictors of treatment and survival among patients treated in community settings.
Methods
A sample of pancreatic cancer patients who were diagnosed in 2009 and reported to the National Cancer Institute’s Surveillance Epidemiology and End Results (SEER) program (n = 977) was included. Logistic regression was used to assess factors associated with therapy modality. Survival was examined using the Kaplan-Meier method and Cox proportional hazards regression. All analyses were conducted stratified by tumor stage.
Results
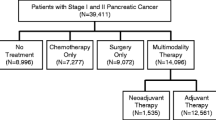
Among stages I–II patients, 27 % received no treatment and only 47 % received surgery. Among these patients, no treatment was associated with older age, being a woman, not being married, lower income, and larger or unknown tumor size. Additionally, the type of adjuvant therapy received varied by tumor characteristics, race/ethnicity, and socioeconomic status. The most common therapies for stage III tumors were chemoradiation (40.8 %) and chemotherapy (21.9 %) alone. Half of stage IV patients received chemotherapy; chemotherapy was less common in patients who were older, in a minority race/ethnicity, and not married. Although treatment was associated with better prognosis, even among stages I–II patients who underwent surgery, a quarter succumbed to their disease within 17 months.
Conclusion
A significant proportion of pancreatic cancer patients remain undertreated primarily due to nonclinical factors, including marital status. Further investigating what aspects of marriage are contributing to this association will provide a better understanding of pancreatic cancer treatment barriers.

Similar content being viewed by others
Abbreviations
- API:
-
Asian-Pacific Islanders
- CI:
-
Confidence interval
- HR:
-
Hazard ratio
- ICD-O3:
-
International Classification of Diseases for Oncology 3rd edition
- NCI:
-
National Cancer Institute
- OR:
-
Odds ratio
- POC:
-
Patterns of care
- SEER:
-
Surveillance Epidemiology and End Results
References
American Cancer Society. Cancer facts & figures: American Cancer Society, Atlanta, 2013.
Abraham A, Al-Refaie WB, Parsons HM, Dudeja V, Vickers SM, Habermann EB. Disparities in pancreas cancer care. Ann Surg Oncol. 2013;20(6):2078–87.
Bilimoria KY, Bentrem DJ, Ko CY, Stewart AK, Winchester DP, Talamonti MS. National failure to operate on early stage pancreatic cancer. Ann Surg. 2007;246(2):173–80.
Bilimoria KY, Bentrem DJ, Ko CY, et al. Multimodality therapy for pancreatic cancer in the U.S. : utilization, outcomes, and the effect of hospital volume. Cancer. 2007;110(6):1227–34.
Murphy MM, Simons JP, Hill JS, et al. Pancreatic resection: a key component to reducing racial disparities in pancreatic adenocarcinoma. Cancer. 2009;115(17):3979–90.
Riall TS, Townsend Jr CM, Kuo YF, Freeman JL, Goodwin JS. Dissecting racial disparities in the treatment of patients with locoregional pancreatic cancer: a 2-step process. Cancer. 2010;116(4):930–9.
Shavers VL, Harlan LC, Jackson M, Robinson J. Racial/ethnic patterns of care for pancreatic cancer. J Palliat Med. 2009;12(7):623–30.
Simons JP, Ng SC, McDade TP, Zhou Z, Earle CC, Tseng JF. Progress for resectable pancreatic cancer?: a population-based assessment of US practices. Cancer. 2010;116(7):1681–90.
National Comprehensive Cancer Network, Inc. Practice Guidelines in Oncology—v1.2013. Pancreatic adenocarcinoma. http://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf Accessed: 5/2013.
Neoptolemos JP, Stocken DD, Friess H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med. 2004;350(12):1200–10.
Oettle H, Post S, Neuhaus P, et al. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAMA. 2007;297(3):267–77.
Regine WF, Winter KA, Abrams RA, et al. Fluorouracil vs gemcitabine chemotherapy before and after fluorouracil-based chemoradiation following resection of pancreatic adenocarcinoma: a randomized controlled trial. JAMA. 2008;299(9):1019–26.
American Joint Committee on Cancer. American Joint Committee on Cancer 7th edition: pancreatic cancer staging. 2009. http://www.cancerstaging.org/staging/posters/pancreas12x15.pdf Accessed:5/2013.
The Gastrointestinal Tumor Study Group. A multi-institutional comparative trial of radiation therapy alone and in combination with 5-fluorouracil for locally unresectable pancreatic carcinoma. Ann Surg. 1979;189(2):205–8.
Chauffert B, Mornex F, Bonnetain F, et al. Phase III trial comparing intensive induction chemoradiotherapy (60 Gy, infusional 5-FU and intermittent cisplatin) followed by maintenance gemcitabine with gemcitabine alone for locally advanced unresectable pancreatic cancer. Definitive results of the 2000–01 FFCD/SFRO study. Ann Oncol. 2008;19(9):1592–9.
Cohen SJ, Dobelbower Jr R, Lipsitz S, et al. A randomized phase III study of radiotherapy alone or with 5-fluorouracil and mitomycin-C in patients with locally advanced adenocarcinoma of the pancreas: Eastern Cooperative Oncology Group study E8282. Int J Radiat Oncol Biol Phys. 2005;62(5):1345–50.
Loehrer Sr PJ, Feng Y, Cardenes H, et al. Gemcitabine alone versus gemcitabine plus radiotherapy in patients with locally advanced pancreatic cancer: an Eastern Cooperative Oncology Group trial. J Clin Oncol. 2011;29(31):4105–12.
Iacobuzio-Donahue CA, Fu B, Yachida S, et al. DPC4 gene status of the primary carcinoma correlates with patterns of failure in patients with pancreatic cancer. J Clin Oncol. 2009;27(11):1806–13.
Burris 3rd HA, Moore MJ, Andersen J, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol. 1997;15(6):2403–13.
Moore MJ, Goldstein D, Hamm J, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2007;25(15):1960–6.
National Cancer Institute. SEER registry groupings for analyses. http://seer.cancer.gov/registries/terms.html Accessed: 05/2013.
American Hospital Association: American Hospital Association annual survey database. http://www.aha.org/research/rc/stat-studies/data-and-directories.shtml Accessed 5/2013.
. National Cancer Institute. SEER training modules: coding CS tumor size. http://training.seer.cancer.gov/collaborative/system/tnm/t/size/ Accessed on 8/2013.
World Health Organization. International Classification of Diseases for Oncology, 3rd Edition (ICD-O-3). http://www.who.int/classifications/icd/adaptations/oncology/en/ Accessed: 5/2013.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987;40 (373–383)
Aizer AA, Chen MH, McCarthy EP, et al. Marital status and survival in patients with cancer. J Clin Oncol. 2013;31(31):3869–76.
Al-Sharaf K, Andren-Sandberg A, Ihse I. Subtotal pancreatectomy for cancer can be safe in the elderly. Eur J Surg. 1999;165(3):230–5.
Casadei R, Zanini N, Morselli-Labate AM, et al. Prognostic factors in periampullary and pancreatic tumor resection in elderly patients. World J Surg. 2006;30(11):1992–2001. discussion 02–3.
Richter A, Niedergethmann M, Lorenz D, Sturm JW, Trede M, Post S. Resection for cancers of the pancreatic head in patients aged 70 years or over. Eur J Surg. 2002;168(6):339–44.
Balcom JH, Rattner DW, Warshaw AL, Chang Y, Fernandez-del Castillo C. Ten-year experience with 733 pancreatic resections: changing indications, older patients, and decreasing length of hospitalization. Arch Surg. 2001;136(4):391–8.
Ballarin R, Spaggiari M, Di Benedetto F, et al. Do not deny pancreatic resection to elderly patients. J Gastrointest Surg. 2009;13(2):341–8.
Hatzaras I, Schmidt C, Klemanski D, et al. Pancreatic resection in the octogenarian: a safe option for pancreatic malignancy. J Am Coll Surg. 2011;212(3):373–7.
Wagner M, Redaelli C, Lietz M, Seiler CA, Friess H, Buchler MW. Curative resection is the single most important factor determining outcome in patients with pancreatic adenocarcinoma. Br J Surg. 2004;91(5):586–94.
Barugola G, Falconi M, Bettini R, et al. The determinant factors of recurrence following resection for ductal pancreatic cancer. JOP. 2007;8(1 Suppl):132–40.
Yokoyama Y, Nimura Y, Nagino M. Advances in the treatment of pancreatic cancer: limitations of surgery and evaluation of new therapeutic strategies. Surg Today. 2009;39(6):466–75.
National Cancer Institute. Pancreatic cancer: a summary of NCI’s FY2010 and FY2011 portfolio and selected research advances. 2012. http://www.cancer.gov/researchandfunding/reports/pancreatic-research-progress-2012.pdf. Accessed 11/2013
Conflict of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Enewold, L., Harlan, L.C., Tucker, T. et al. Pancreatic Cancer in the USA: Persistence of Undertreatment and Poor Outcome. J Gastrointest Canc 46, 9–20 (2015). https://doi.org/10.1007/s12029-014-9668-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-014-9668-x