Abstract
Background
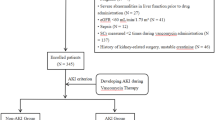
Traumatic brain injury (TBI) is associated with secondary complications, including infection, and patients with TBI often exhibit augmented renal clearance (ARC). This phenomenon has been associated with subtherapeutic levels of renally cleared drugs such as vancomycin, which is dosed based on body weight and creatinine clearance (CrCl). Many clinicians, however, cap CrCl at 120 mL/min/1.73 m2 when calculating vancomycin dosing regimens. We hypothesize that capping patient CrCl, as opposed to utilizing the non-capped CrCl, when determining vancomycin dosing schemes results in subtherapeutic serum trough concentrations in patients with TBI.
Methods
This was a retrospective study of adult patients with TBI admitted between April 2014 and December 2015 who received vancomycin. Population-based pharmacokinetic (PK) parameters using non-capped calculated CrCl and capped CrCl were compared with patient-specific PK parameters based on serum trough concentrations.
Results
Thirty-two patients with TBI were included in the study. ARC was suspected in 24 (75%) patients due to a median estimated CrCl at serum trough concentration of 167.3 (127.7–197.7) mL/min. The mean dosing regimen was 17.1 (13.2–19.2) mg/kg every 8 (8–8) h. There was no difference between the median measured trough concentration and predicted value using non-capped CrCl [10.4 (7.1–15.0) vs. 11.5 (7.8–13.7) mcg/mL; p = 0.7986]. The median measured trough concentration was significantly lower than the predicted trough concentration when calculated based on capping the CrCl at 120 mL/min/1.73 m2 [16.3 (15.3–22.0) vs. 11.5 (7.8–13.7) mcg/mL; p < 0.0001].
Conclusions
Patients with traumatic brain injury appeared to exhibit augmented renal clearance, leading to subtherapeutic vancomycin serum trough concentrations when doses were calculated using the traditional method of capping creatinine clearance at 120 mL/min/1.73 m2. Instead, utilizing patients’ non-capped creatinine clearance when determining a vancomycin dosing regimen is more accurate and provides a better estimation of vancomycin pharmacokinetics and could be applied to other renally excreted medications.
Similar content being viewed by others
References
Frattalone AR, Ling GS. Moderate and severe traumatic brain injury: pathophysiology and management. Neurosurg Clin N Am. 2013;24:309–19.
Brain Trauma Foundation. Guidelines for the management of severe traumatic brain injury. J Neurotrauma. 2007;24:S1–106.
Kinoshita K. Traumatic brain injury: pathophysiology for neurocritical care. J Intensive Care. 2016;4:29.
Helling TS, Evans LL, Fowler DL, et al. Infectious complication in patients with severe head injury. J Trauma. 1988;28(11):1575–7.
Kourbeti IS, Vakis AF, Papadakis JA, et al. Infections in traumatic brain injury patients. Clin Microbiol Infect. 2012;18(4):359–64.
Morbitzer KA, Jordan JD, Sullivan KA, et al. Vancomycin pharmacokinetic parameters in patients with acute brain injury undergoing controlled normothermia, therapeutic hypothermia, or pentobarbital infusion. Neurocrit Care. 2015;22(2):258–64.
Udy AA, Baptista JP, Lim NL, et al. Augmented renal clearance in the ICU: results of a multicenter observational study of renal function in critically ill patients with normal plasma creatinine concentrations. Crit Care Med. 2014;42:520–7.
Carrie C, Betejac M, Cottenceau V, et al. Association between augmented renal clearance and clinical failure of antibiotic treatment in brain-injured patients with ventilator-acquired pneumonia: a preliminary study. Anaesth Crit Care Pain Med. 2018;37:35–41. https://doi.org/10.1016/j.accpm.2017.06.006.
Udy AA, Boots R, Senthuran S, et al. Augmented renal clearance in traumatic brain injury: a single center observational study of atrial natriuretic peptide, cardiac output, and creatinine clearance. J Neurotrauma. 2017;34:137–44.
Cook AM, Arora S, Davis J, Pittman T. Augmented renal clearance of vancomycin and levetiracetam in traumatic brain injury patient. Neurocrit Care. 2013;19:210–4.
Wilhelm SM, Kale-Pradhan PB. Estimating creatinine clearance: a meta-analysis. Pharmacotherapy. 2011;31(7):658–64.
Winter MA, Guhr KN, Berg GM. Impact of various body weights and serum creatinine concentrations on the bias and accuracy of the Cockcroft–Gault equation. Pharmacotherapy. 2012;32(7):604–12.
Dooley MJ, Singh S, Rischin D. Rounding of low serum creatinine levels and consequent impact on accuracy of bedside estimates of renal function in cancer patients. Br J Cancer. 2004;90(5):991–5.
Michaud L. Drug dosing and new measures of serum creatinine: clear as mud. HemOnc Today. 2010. Available at: https://www.healio.com/hematology-oncology/news/print/hemonc-today/%7B4bc077c3-ee73-4329-8187-a658eedcc61b%7D/drug-dosing-and-new-measures-of-serum-creatinine-clear-as-mud. Accessed 13 June 2018.
Llopis-Salvia P, Jimenez-Torres NV. Population pharmacokinetic parameters of vancomycin in critically ill patients. J Clin Pharm Ther. 2006;31(5):447–54.
National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39(2 Suppl 1):S1–266.
Stevens LA, Levey AS. Measured GFR as a confirmatory test for estimated GFR. J Am Soc Nephrol. 2009;20(11):2305–13.
Family Practice Notebook. Creatinine clearance. Available at: https://fpnotebook.com/Renal/Lab/CrtnClrnc.htm. Accessed 24 May 2018.
Stevens LA, Coresh J, Greene T, Levey AS. Assessing kidney function—measured and estimated glomerular filtration rate. N Engl J Med. 2006;354:2473–83.
Matzke GR, McGory RW, Halstenson CE, Keane WF. Pharmacokinetics of vancomycin in patients with various degrees of renal function. Antimicrob Agents Chemother. 1984;25(4):433–7.
Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16:31–41.
Wurtz R, Itokazu G, Rodvold K. Antimicrobial dosing in obese patients. Clin Infect Dis. 1997;25:112–8.
Sawyer WT, Canaday BR, Poe TE, et al. Variables affecting creatinine clearance prediction. Am J Hosp Pharm. 1983;40:2175–80.
Ambrose P, Winter ME. Vancomycin. In: Beringer P, editor. Winter’s basic clinical pharmacokinetics. 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2004. p. 451–476.
Oswalt A, Joseph AC, Sima A, Kurczewski L. Evaluation of intravenous vancomycin pharmacokinetic parameters in patients with acute brain injury. J Pharm Pract. 2017. https://doi.org/10.1177/0897190017743133.
McAuley D. Creatinine clearance calculator. GlobalRPh.com. Last updated 11/13/2017. Available at: www.globalrph.com/crcl.htm. Accessed 3 November 2017.
Evidence-based Medicine Consult. Adult creatinine clearance (CrCl) calculator. www.ebmconsult.com. Available at: https://www.ebmconsult.com/app/medical-calculators/creatinine-clearance-crcl-calculator. Accessed 24 May 2018.
Funding
No sources of support were provided for this research.
Author information
Authors and Affiliations
Contributions
All authors provided substantial contributions to conception and design, acquisition of data, and analysis and interpretation of data; drafting the article and revising it critically for important intellectual content; and final approval of the version to be published.
Corresponding author
Ethics declarations
Funding
No funding was received to carry out this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
This study received ethic approval by the Institutional Review Board at the University of North Carolina at Chapel Hill. This is included within the Methods and Materials section of the manuscript.
Rights and permissions
About this article
Cite this article
Nelson, N.R., Morbitzer, K.A., Jordan, J.D. et al. The Impact of Capping Creatinine Clearance on Achieving Therapeutic Vancomycin Concentrations in Neurocritically Ill Patients with Traumatic Brain Injury. Neurocrit Care 30, 126–131 (2019). https://doi.org/10.1007/s12028-018-0583-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-018-0583-z