Abstract
Background
Delayed deterioration associated with vasospasm (DDAV) after aneurismal subarachnoid hemorrhage (SAH) is a major cause of morbidity. We have previously shown that myeloid cell depletion before experimental SAH in a murine model ameliorates DDAV. In this study, we address whether systemic administration of lipopolysaccharide (LPS) worsens DDAV in a myeloid cell-dependent fashion.
Methods
We challenged mice in our experimental SAH model with LPS before hemorrhage and evaluated the degree of vasospasm on day 6 with India ink angiography; behavioral deficits by rotorod, Y-maze, and Barnes maze testing; microglial activation early after SAH by immunohistochemistry; and the brain levels of the chemokines CCL5 and KC at the time of vasospasm. Another group of animals were given the myeloid cell-depleting antibody against the neutrophil antigen Ly6G/C prior to LPS administration and SAH.
Results
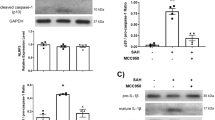
LPS followed by SAH significantly worsens angiographic vasospasm as well as performance on the Barnes maze but not the Y-maze or rotorod tests. There was an increased activation of microglia in animals with LPS before SAH compared to SAH alone. Depletion of myeloid cells before LPS administration inhibited the development of vasospasm, improved the performance on behavioral tests, and reduced microglial activation. The chemokines CCL5 and KC were incrementally elevated in SAH and LPS SAH, but suppressed in animals with myeloid cell depletion.
Conclusions
LPS administration before SAH worsens DDAV through a myeloid cell-dependent mechanism supporting studies in humans which show that systemic inflammation increases the likelihood of developing DDAV.



Similar content being viewed by others
References
van Gijn J, Rinkel GJ. Subarachnoid haemorrhage: diagnosis, causes and management. Brain. 2001;124:249–78.
Etminan N, Vergouwen MD, Ilodigwe D, Macdonald RL. Effect of pharmaceutical treatment on vasospasm, delayed cerebral ischemia, and clinical outcome in patients with aneurysmal subarachnoid hemorrhage: a systematic review and meta-analysis. J Cereb Blood Flow Metab. 2011;31(6):1443–51.
Stein SC, Levine JM, Nagpal S, LeRoux PD. Vasospasm as the sole cause of cerebral ischemia: how strong is the evidence? Neurosurg Focus. 2006;21:E2.
Vergouwen MD, Ilodigwe D, Macdonald RL. Cerebral infarction after subarachnoid hemorrhage contributes to poor outcome by vasospasm-dependent and -independent effects. Stroke. 2011;42:924–9.
Provencio JJ, Vora N. Subarachnoid hemorrhage and inflammation: bench to bedside and back. Semin Neurol. 2005;25:435–44.
Provencio JJ, Altay T, Smithason S, Moore SK, Ransohoff RM. Depletion of Ly6G/C(+) cells ameliorates delayed cerebral vasospasm in subarachnoid hemorrhage. J Neuroimmunol. 2010;232(1–2):94–100.
Clatterbuck RE, Gailloud P, Ogata L, et al. Prevention of cerebral vasospasm by a humanized anti-CD11/CD18 monoclonal antibody administered after experimental subarachnoid hemorrhage in nonhuman primates. J Neurosurg. 2003;99:376–82.
Zubkov AY, Tibbs RE, Aoki K, Zhang JH. Prevention of vasospasm in penetrating arteries with MAPK inhibitors in dog double-hemorrhage model. Surg Neurol. 2000;54:221–7.
Provencio JJ, Fu X, Siu A, Rasmussen PA, Hazen SL, Ransohoff RM. CSF neutrophils are implicated in the development of vasospasm in subarachnoid hemorrhage. Neurocrit Care. 2010;12:244–51.
Pradilla G, Wang PP, Legnani FG, Ogata L, Dietsch GN, Tamargo RJ. Prevention of vasospasm by anti-CD11/CD18 monoclonal antibody therapy following subarachnoid hemorrhage in rabbits. J Neurosurg. 2004;101:88–92.
Recinos PF, Pradilla G, Thai Q-A, Perez M, Hdeib AM, Tamargo RJ. Controlled release of lipopolysaccharide in the subarachnoid space of rabbits induces chronic vasospasm in the absence of blood. Surg Neurol. 2006;66:463–9.
Zhou C, Yamaguchi M, Kusaka G, Schonholz C, Nanda A, Zhang JH. Caspase inhibitors prevent endothelial apoptosis and cerebral vasospasm in dog model of experimental subarachnoid hemorrhage. J Cereb Blood Flow Metab. 2004;24:419–31.
Dhar R, Diringer MN. The burden of the systemic inflammatory response predicts vasospasm and outcome after subarachnoid hemorrhage. Neurocrit Care. 2008;8:404–12.
Tam AK, Ilodigwe D, Mocco J, et al. Impact of systemic inflammatory response syndrome on vasospasm, cerebral infarction, and outcome after subarachnoid hemorrhage: exploratory analysis of CONSCIOUS-1 database. Neurocrit Care. 2010;13:182–9.
Yoshimoto Y, Tanaka Y, Hoya K. Acute systemic inflammatory response syndrome in subarachnoid hemorrhage. Stroke. 2001;32:1989–93.
Altay T, Smithason S, Volokh N, Rasmussen PA, Ransohoff RM, Provencio JJ. A novel method for subarachnoid hemorrhage to induce vasospasm in mice. J Neurosci Methods. 2009;183:136–40.
Goergen SK, Barrie D, Sacharias N, Waugh JR. Perimesencephalic subarachnoid haemorrhage: negative angiography and favourable prognosis. Australas Radiol. 1993;37:156–60.
Hang CH, Shi JX, Tian J, Li JS, Wu W, Yin HX. Effect of systemic LPS injection on cortical NF-kappaB activity and inflammatory response following traumatic brain injury in rats. Brain Res. 2004;1026:23–32.
Powell J, Kitchen N, Heslin J, Greenwood R. Psychosocial outcomes at 18 months after good neurological recovery from aneurysmal subarachnoid haemorrhage. J Neurol Neurosurg Psychiatry. 2004;75:1119–24.
Noble AJ, Baisch S, Mendelow AD, Allen L, Kane P, Schenk T. Posttraumatic stress disorder explains reduced quality of life in subarachnoid hemorrhage patients in both the short and long term. Neurosurgery. 2008;63:1095–104.
Clatterbuck RE, Oshiro EM, Hoffman PA, Dietsch GN, Pardoll DM, Tamargo RJ. Inhibition of vasospasm with lymphocyte function-associated antigen-1 monoclonal antibody in a femoral artery model in rats. J Neurosurg. 2002;97:676–82.
Aihara Y, Kasuya H, Onda H, Hori T, Takeda J. Quantitative analysis of gene expressions related to inflammation in canine spastic artery after subarachnoid hemorrhage. Stroke. 2001;32:212–7.
Pyne-Geithman GJ, Morgan CJ, Wagner K, et al. Bilirubin production and oxidation in CSF of patients with cerebral vasospasm after subarachnoid hemorrhage. J Cereb Blood Flow Metab. 2005;25:1070–7.
Chaichana KL, Levy AP, Miller-Lotan R, Shakur S, Tamargo RJ. Haptoglobin 2–2 genotype determines chronic vasospasm after experimental subarachnoid hemorrhage. Stroke. 2007;38:3266–71.
Ellis TN, Beaman BL. Murine polymorphonuclear neutrophils produce interferon-gamma in response to pulmonary infection with Nocardia asteroides. J Leukoc Biol. 2002;72:373–81.
Acknowledgments
The authors would like to thank James Bena for the statistical assistance. This work was supported by a grant from the NIH (K08-NS051350 JJP) and the Cleveland Clinic Cerebrovascular Center (Institutional Support JJP).
Conflict of interest
The authors declared that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smithason, S., Moore, S.K. & Provencio, J.J. Systemic Administration of LPS Worsens Delayed Deterioration Associated with Vasospasm After Subarachnoid Hemorrhage Through a Myeloid Cell-Dependent Mechanism. Neurocrit Care 16, 327–334 (2012). https://doi.org/10.1007/s12028-011-9651-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-011-9651-3