Abstract
Introduction
Conivaptan is the first arginine vasopressin antagonist to be FDA-approved for the treatment of euvolemic hyponatremia, a common complication in neurointensive care patients. Due to risks for cerebral edema and seizures, sodium levels are generally aggressively maintained within normal levels (135–145 meq/l) in this patient population.
Objective
To assess the safety and efficacy of conivaptan for the treatment of euvolemic hyponatremia in the neurocritical care unit.
Methods
Data were obtained retrospectively on 22 patients treated with conivaptan for euvolemic hyponatremia. End points evaluated included time to [Na] increase of ≥6 meq/l; incidences of rapid overcorrection of [Na] (defined as an increase of >12 meq/l in a 24-h period while on conivaptan), infusion site reactions, or other adverse events; and whether sodium levels decreased after discontinuation of conivaptan.
Results
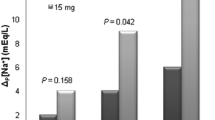
A [Na] increase of ≥6 meq/l was reached in 19/22 (86%) patients, with an average time to goal of 13.1 h. No patients experienced a rapid overcorrection of [Na]. Five patients had an infusion site reaction necessitating an IV change. One patient experienced hypotension and another complained of thirst during infusion. Conivaptan was initiated in 11/22 patients (50%) who were hyponatremic despite already being on conventional therapies.
Conclusion
Conivaptan was safe and effective in this small series of neurointensive care patients, including many patients who were hyponatremic despite traditional treatments to maintain normal sodium levels. Further studies are needed to clarify the role of conivaptan as an adjunctive and/or alternative therapy for hyponatremia in this patient population.



Similar content being viewed by others
References
Androgué HJ, Madias NE. Hyponatremia. N Engl J Med. 2000;342:1581–9. doi:10.1056/NEJM200005253422107.
Sterns RH, Silver SM. Brain volume regulation in response to hypo-osmolality and its correction. Am J Med. 2006;119:S12–6. doi:10.1016/j.amjmed.2006.05.003.
Bhardwaj A, Ulatowski JA. Hypertonic solutions and brain injury. Curr Opin Crit Care. 2004;10:126–31. doi:10.1097/00075198-200404000-00009.
Diringer MN, Zazulia AR. Hyponatremia in neurologic patients: consequences and approaches to treatment. Neurologist. 2006;12(3):117–26. doi:10.1097/01.nrl.0000215741.01699.77.
Nathan BR. Cerebral correlates of hyponatremia. Neurocrit Care. 2007;6:72–8. doi:10.1385/NCC:6:1:72.
Zeltser D, Rosansky S, van Rensburg H, Verbalis JG, Smith N, for the Conivaptan Study Group. Assessment of the efficacy of intravenous conivaptan in euvolemic and hypervolemic hyponatremia. Am J Nephrol. 2007;27:447–57. doi:10.1159/000106456.
Vaprisol package insert. Deerfield: Astellas Pharma, U.S. Inc.; 2007
Verbalis JG. AVP receptor antagonists as aquaretics: review and assessment of clinical data. Cleve Clin J Med. 2006;73(Suppl 3):S24–33.
Palmer BF. Hyponatremia in patients with central nervous system disease: SIADH versus CSW. Trends Endocrinol Metab. 2003;14(4):182–7. doi:10.1016/S1043-2760(03)00048-1.
Gross P, Reimann D, Henschkowski J, Damian M. Treatment of severe hyponatremia: conventional and novel aspects. J Am Soc Nephrol. 2001;12:S10–4.
Verbalis JG. Vasopressin V2 receptor antagonists. J Mol Endocrinol. 2002;29:1–9. doi:10.1677/jme.0.0290001.
Bhardwaj A. Neurological impact of vasopressin dysregulation and hyponatremia. Ann Neurol. 2006;59:229–36. doi:10.1002/ana.20788.
Hasan D, Vermeulen M, Wijdicks EF, Hijdra A, van Gijn J. Effect of fluid intake and antihypertensive treatment on cerebral ischemia after subarachnoid hemorrhage. Stroke. 1989;20(11):1511–5.
Murphy T, Dhar R, Axelrod A, Corry J, Russel E, Diringer M. Conivaptan for the correction of hyponatremia in the neurocritical care unit. Neurocrit Care. 2008;8:196.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wright, W.L., Asbury, W.H., Gilmore, J.L. et al. Conivaptan for Hyponatremia in the Neurocritical Care Unit. Neurocrit Care 11, 6–13 (2009). https://doi.org/10.1007/s12028-008-9152-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-008-9152-1