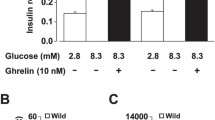
Abstract
Ghrelin and its synthetic analog hexarelin are specific ligands of growth hormone secretagogue (GHS) receptor. GHS have strong growth hormone-releasing effect and other neuroendocrine activities such as stimulatory effects on prolactin and adrenocorticotropic hormone secretion. Recently, several studies have reported other beneficial functions of GHS that are independent of GH. Ghrelin and hexarelin, for examples, have been shown to exert GH-independent cardiovascular activity. Hexarelin has been reported to regulate peroxisome proliferator-activated receptor gamma (PPAR-γ) in macrophages and adipocytes. PPAR-γ is an important regulator of adipogenesis, lipid metabolism, and insulin sensitization. Ghrelin also shows protective effects on beta cells against lipotoxicity through activation of phosphatidylinositol-3 kinase/protein kinase B, c-Jun N-terminal kinase (JNK) inhibition, and nuclear exclusion of forkhead box protein O1. Acylated ghrelin (AG) and unacylated ghrelin (UAG) administration reduces glucose levels and increases insulin-producing beta cell number, and insulin secretion in pancreatectomized rats and in newborn rats treated with streptozotocin, suggesting a possible role of GHS in pancreatic regeneration. Therefore, the discovery of GHS has opened many new perspectives in endocrine, metabolic, and cardiovascular research areas, suggesting the possible therapeutic application in diabetes and diabetic complications especially diabetic cardiomyopathy. Here, we review the physiological roles of ghrelin and hexarelin in the protection and regeneration of beta cells and their roles in the regulation of insulin release, glucose, and fat metabolism and present their potential therapeutic effects in the treatment of diabetes and diabetic-associated heart diseases.


Similar content being viewed by others
References
L. Guariguata, D.R. Whiting, I. Hambleton, J. Beagley, U. Linnenkamp, J.E. Shaw, Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res. Clin. Pract. 103(2), 137–149 (2014). doi:10.1016/j.diabres.2013.11.002
D.A. Stoffers, The development of beta-cell mass: recent progress and potential role of GLP-1. Hormone Metab. Res. 36(11–12), 811–821 (2004). doi:10.1055/s-2004-826168
R. Colagiuri, R. Short, A. Buckley, The status of national diabetes programmes: a global survey of IDF member associations. Diabetes Res. Clin. Pract. 87(2), 137–142 (2010). doi:10.1016/j.diabres.2009.10.005
J.A. Wali, H.E. Thomas, A.P. Sutherland, Linking obesity with type 2 diabetes: the role of T-bet. Diabetes Metabol. Syndr. Obes. 7, 331–340 (2014). doi:10.2147/DMSO.S51432
H. Sakuraba, H. Mizukami, N. Yagihashi, R. Wada, C. Hanyu, S. Yagihashi, Reduced beta-cell mass and expression of oxidative stress-related DNA damage in the islet of Japanese Type II diabetic patients. Diabetologia 45(1), 85–96 (2002). doi:10.1007/s001250200009
P. Marchetti, S. Del Guerra, L. Marselli, R. Lupi, M. Masini, M. Pollera, M. Bugliani, U. Boggi, F. Vistoli, F. Mosca, S. Del Prato, Pancreatic islets from type 2 diabetic patients have functional defects and increased apoptosis that are ameliorated by metformin. J. Clin. Endocrinol. Metab. 89(11), 5535–5541 (2004). doi:10.1210/jc.2004-0150
I. Cozar-Castellano, N. Fiaschi-Taesch, T.A. Bigatel, K.K. Takane, A. Garcia-Ocana, R. Vasavada, A.F. Stewart, Molecular control of cell cycle progression in the pancreatic beta-cell. Endocr. Rev. 27(4), 356–370 (2006). doi:10.1210/er.2006-0004
D.R. Matthews, The natural history of diabetes-related complications: the UKPDS experience. United Kingdom Prospective Diabetes Study. Diabetes Obes. Metab. 1(Suppl 2), S7–S13 (1999)
R.R. Holman, Assessing the potential for alpha-glucosidase inhibitors in prediabetic states. Diabetes Res. Clin. Pract. 40(Suppl), S21–S25 (1998)
M. Prentki, C.J. Nolan, Islet beta cell failure in type 2 diabetes. J. Clin. Investig. 116(7), 1802–1812 (2006). doi:10.1172/JCI29103
B.L. Wajchenberg, Beta-cell failure in diabetes and preservation by clinical treatment. Endocr. Rev. 28(2), 187–218 (2007). doi:10.1210/10.1210/er.2006-0038
P.A. Halban, K.S. Polonsky, D.W. Bowden, M.A. Hawkins, C. Ling, K.J. Mather, A.C. Powers, C.J. Rhodes, L. Sussel, G.C. Weir, Beta-cell failure in type 2 diabetes: postulated mechanisms and prospects for prevention and treatment. Diabetes Care 37(6), 1751–1758 (2014). doi:10.2337/dc14-0396
W. Quan, E.K. Jo, M.S. Lee, Role of pancreatic beta-cell death and inflammation in diabetes. Diabetes Obes. Metab. 15(Suppl 3), 141–151 (2013). doi:10.1111/dom.12153
M.S. Akash, K. Rehman, S. Chen, Role of inflammatory mechanisms in pathogenesis of type 2 diabetes mellitus. J. Cell. Biochem. 114(3), 525–531 (2013). doi:10.1002/jcb.24402
R.P. Robertson, J. Harmon, P.O. Tran, Y. Tanaka, H. Takahashi, Glucose toxicity in beta-cells: type 2 diabetes, good radicals gone bad, and the glutathione connection. Diabetes 52(3), 581–587 (2003)
A. Klip, M.R. Paquet, Glucose transport and glucose transporters in muscle and their metabolic regulation. Diabetes Care 13(3), 228–243 (1990)
I. Shimomura, R.E. Hammer, S. Ikemoto, M.S. Brown, J.L. Goldstein, Leptin reverses insulin resistance and diabetes mellitus in mice with congenital lipodystrophy. Nature 401(6748), 73–76 (1999). doi:10.1038/43448
K. Ebihara, Y. Ogawa, H. Masuzaki, M. Shintani, F. Miyanaga, M. Aizawa-Abe, T. Hayashi, K. Hosoda, G. Inoue, Y. Yoshimasa, O. Gavrilova, M.L. Reitman, K. Nakao, Transgenic overexpression of leptin rescues insulin resistance and diabetes in a mouse model of lipoatrophic diabetes. Diabetes 50(6), 1440–1448 (2001)
K. Hotta, T. Funahashi, Y. Arita, M. Takahashi, M. Matsuda, Y. Okamoto, H. Iwahashi, H. Kuriyama, N. Ouchi, K. Maeda, M. Nishida, S. Kihara, N. Sakai, T. Nakajima, K. Hasegawa, M. Muraguchi, Y. Ohmoto, T. Nakamura, S. Yamashita, T. Hanafusa, Y. Matsuzawa, Plasma concentrations of a novel, adipose-specific protein, adiponectin, in type 2 diabetic patients. Arterioscler. Thromb. Vasc. Biol. 20(6), 1595–1599 (2000)
M.L. Reitman, O. Gavrilova, A-ZIP/F-1 mice lacking white fat: a model for understanding lipoatrophic diabetes. Int. J. Obes. Relat. Metabol. Disord. 24(Suppl 4), S11–S14 (2000)
O. Gavrilova, B. Marcus-Samuels, D. Graham, J.K. Kim, G.I. Shulman, A.L. Castle, C. Vinson, M. Eckhaus, M.L. Reitman, Surgical implantation of adipose tissue reverses diabetes in lipoatrophic mice. J. Clin. Investig. 105(3), 271–278 (2000). doi:10.1172/JCI7901
J.C. Bruning, M.D. Michael, J.N. Winnay, T. Hayashi, D. Horsch, D. Accili, L.J. Goodyear, C.R. Kahn, A muscle-specific insulin receptor knockout exhibits features of the metabolic syndrome of NIDDM without altering glucose tolerance. Mol. Cell 2(5), 559–569 (1998)
E.D. Abel, O. Peroni, J.K. Kim, Y.B. Kim, O. Boss, E. Hadro, T. Minnemann, G.I. Shulman, B.B. Kahn, Adipose-selective targeting of the GLUT4 gene impairs insulin action in muscle and liver. Nature 409(6821), 729–733 (2001). doi:10.1038/35055575
C.J. Shu, C. Benoist, D. Mathis, The immune system’s involvement in obesity-driven type 2 diabetes. Semin. Immunol. 24(6), 436–442 (2012). doi:10.1016/j.smim.2012.12.001
M. Kojima, K. Kangawa, Ghrelin: structure and function. Physiol. Rev. 85(2), 495–522 (2005). doi:10.1152/physrev.00012.2004
C.Y. Chen, A. Asakawa, M. Fujimiya, S.D. Lee, A. Inui, Ghrelin gene products and the regulation of food intake and gut motility. Pharmacol. Rev. 61(4), 430–481 (2009). doi:10.1124/pr.109.001958
F. Broglio, C. Gottero, F. Prodam, C. Gauna, G. Muccioli, M. Papotti, T. Abribat, A.J. Van Der Lely, E. Ghigo, Non-acylated ghrelin counteracts the metabolic but not the neuroendocrine response to acylated ghrelin in humans. J. Clin. Endocrinol. Metab. 89(6), 3062–3065 (2004). doi:10.1210/jc.2003-031964
C. Gauna, P.J. Delhanty, L.J. Hofland, J.A. Janssen, F. Broglio, R.J. Ross, E. Ghigo, A.J. van der Lely, Ghrelin stimulates, whereas des-octanoyl ghrelin inhibits, glucose output by primary hepatocytes. J. Clin. Endocrinol. Metab. 90(2), 1055–1060 (2005). doi:10.1210/jc.2004-1069
R. Kumar, A. Salehi, J.F. Rehfeld, P. Hoglund, E. Lindstrom, R. Hakanson, Proghrelin peptides: desacyl ghrelin is a powerful inhibitor of acylated ghrelin, likely to impair physiological effects of acyl ghrelin but not of obestatin A study of pancreatic polypeptide secretion from mouse islets. Regul. Pept. 164(2–3), 65–70 (2010). doi:10.1016/j.regpep.2010.06.005
G. Muccioli, N. Pons, C. Ghe, F. Catapano, R. Granata, E. Ghigo, Ghrelin and des-acyl ghrelin both inhibit isoproterenol-induced lipolysis in rat adipocytes via a non-type 1a growth hormone secretagogue receptor. Eur. J. Pharmacol. 498(1–3), 27–35 (2004). doi:10.1016/j.ejphar.2004.07.066
K. Toshinai, H. Yamaguchi, Y. Sun, R.G. Smith, A. Yamanaka, T. Sakurai, Y. Date, M.S. Mondal, T. Shimbara, T. Kawagoe, N. Murakami, M. Miyazato, K. Kangawa, M. Nakazato, Des-acyl ghrelin induces food intake by a mechanism independent of the growth hormone secretagogue receptor. Endocrinology 147(5), 2306–2314 (2006). doi:10.1210/en.2005-1357
H. Chung, S. Seo, M. Moon, S. Park, Phosphatidylinositol-3-kinase/Akt/glycogen synthase kinase-3 beta and ERK1/2 pathways mediate protective effects of acylated and unacylated ghrelin against oxygen-glucose deprivation-induced apoptosis in primary rat cortical neuronal cells. J. Endocrinol. 198(3), 511–521 (2008). doi:10.1677/JOE-08-0160
G. Baldanzi, N. Filigheddu, S. Cutrupi, F. Catapano, S. Bonissoni, A. Fubini, D. Malan, G. Baj, R. Granata, F. Broglio, M. Papotti, N. Surico, F. Bussolino, J. Isgaard, R. Deghenghi, F. Sinigaglia, M. Prat, G. Muccioli, E. Ghigo, A. Graziani, Ghrelin and des-acyl ghrelin inhibit cell death in cardiomyocytes and endothelial cells through ERK1/2 and PI 3-kinase/AKT. J. Cell Biol. 159(6), 1029–1037 (2002). doi:10.1083/jcb.200207165
X.M. Guan, H. Yu, O.C. Palyha, K.K. McKee, S.D. Feighner, D.J. Sirinathsinghji, R.G. Smith, L.H. Van der Ploeg, A.D. Howard, Distribution of mRNA encoding the growth hormone secretagogue receptor in brain and peripheral tissues. Brain Res. Mol. Brain Res. 48(1), 23–29 (1997)
A.D. Howard, S.D. Feighner, D.F. Cully, J.P. Arena, P.A. Liberator, C.I. Rosenblum, M. Hamelin, D.L. Hreniuk, O.C. Palyha, J. Anderson, P.S. Paress, C. Diaz, M. Chou, K.K. Liu, K.K. McKee, S.S. Pong, L.Y. Chaung, A. Elbrecht, M. Dashkevicz, R. Heavens, M. Rigby, D.J. Sirinathsinghji, D.C. Dean, D.G. Melillo, A.A. Patchett, R. Nargund, P.R. Griffin, J.A. DeMartino, S.K. Gupta, J.M. Schaeffer, R.G. Smith, L.H. Van der Ploeg, A receptor in pituitary and hypothalamus that functions in growth hormone release. Science 273(5277), 974–977 (1996)
G. Muccioli, A. Baragli, R. Granata, M. Papotti, E. Ghigo, Heterogeneity of ghrelin/growth hormone secretagogue receptors. Toward the understanding of the molecular identity of novel ghrelin/GHS receptors. Neuroendocrinology 86(3), 147–164 (2007). doi:10.1159/000105141
Y. Shuto, T. Shibasaki, K. Wada, I. Parhar, J. Kamegai, H. Sugihara, S. Oikawa, I. Wakabayashi, Generation of polyclonal antiserum against the growth hormone secretagogue receptor (GHS-R): evidence that the GHS-R exists in the hypothalamus, pituitary and stomach of rats. Life Sci. 68(9), 991–996 (2001)
Y. Date, M. Kojima, H. Hosoda, A. Sawaguchi, M.S. Mondal, T. Suganuma, S. Matsukura, K. Kangawa, M. Nakazato, Ghrelin, a novel growth hormone-releasing acylated peptide, is synthesized in a distinct endocrine cell type in the gastrointestinal tracts of rats and humans. Endocrinology 141(11), 4255–4261 (2000). doi:10.1210/endo.141.11.7757
K. Mori, A. Yoshimoto, K. Takaya, K. Hosoda, H. Ariyasu, K. Yahata, M. Mukoyama, A. Sugawara, H. Hosoda, M. Kojima, K. Kangawa, K. Nakao, Kidney produces a novel acylated peptide, ghrelin. FEBS Lett. 486(3), 213–216 (2000)
N. Nagaya, M. Kojima, M. Uematsu, M. Yamagishi, H. Hosoda, H. Oya, Y. Hayashi, K. Kangawa, Hemodynamic and hormonal effects of human ghrelin in healthy volunteers. Am. J. Physiol. Regul. Integr. Comp. Physiol. 280(5), R1483–R1487 (2001)
M. Korbonits, R.A. Jacobs, S.J. Aylwin, J.M. Burrin, P.L. Dahia, J.P. Monson, J. Honegger, R. Fahlbush, P.J. Trainer, S.L. Chew, G.M. Besser, A.B. Grossman, Expression of the growth hormone secretagogue receptor in pituitary adenomas and other neuroendocrine tumors. J. Clin. Endocrinol. Metab. 83(10), 3624–3630 (1998). doi:10.1210/jcem.83.10.5210
K. Kim, K. Arai, N. Sanno, R.Y. Osamura, A. Teramoto, T. Shibasaki, Ghrelin and growth hormone (GH) secretagogue receptor (GHSR) mRNA expression in human pituitary adenomas. Clin. Endocrinol. 54(6), 759–768 (2001)
M. Papotti, P. Cassoni, M. Volante, R. Deghenghi, G. Muccioli, E. Ghigo, Ghrelin-producing endocrine tumors of the stomach and intestine. J. Clin. Endocrinol. Metab. 86(10), 5052–5059 (2001). doi:10.1210/jcem.86.10.7918
M. Kojima, H. Hosoda, Y. Date, M. Nakazato, H. Matsuo, K. Kangawa, Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402(6762), 656–660 (1999). doi:10.1038/45230
E. Arvat, L. Di Vito, F. Broglio, M. Papotti, G. Muccioli, C. Dieguez, F.F. Casanueva, R. Deghenghi, F. Camanni, E. Ghigo, Preliminary evidence that Ghrelin, the natural GH secretagogue (GHS)-receptor ligand, strongly stimulates GH secretion in humans. J. Endocrinol. Invest. 23(8), 493–495 (2000)
E. Arvat, M. Maccario, L. Di Vito, F. Broglio, A. Benso, C. Gottero, M. Papotti, G. Muccioli, C. Dieguez, F.F. Casanueva, R. Deghenghi, F. Camanni, E. Ghigo, Endocrine activities of ghrelin, a natural growth hormone secretagogue (GHS), in humans: comparison and interactions with hexarelin, a nonnatural peptidyl GHS, and GH-releasing hormone. J. Clin. Endocrinol. Metab. 86(3), 1169–1174 (2001). doi:10.1210/jcem.86.3.7314
F. Broglio, A. Benso, C. Castiglioni, C. Gottero, F. Prodam, S. Destefanis, C. Gauna, A.J. van der Lely, R. Deghenghi, M. Bo, E. Arvat, E. Ghigo, The endocrine response to ghrelin as a function of gender in humans in young and elderly subjects. J. Clin. Endocrinol. Metab. 88(4), 1537–1542 (2003). doi:10.1210/jc.2002-021504
A.M. Wren, L.J. Seal, M.A. Cohen, A.E. Brynes, G.S. Frost, K.G. Murphy, W.S. Dhillo, M.A. Ghatei, S.R. Bloom, Ghrelin enhances appetite and increases food intake in humans. J. Clin. Endocrinol. Metab. 86(12), 5992 (2001). doi:10.1210/jcem.86.12.8111
D.E. Cummings, J. Overduin, Gastrointestinal regulation of food intake. J. Clin. Investig. 117(1), 13–23 (2007). doi:10.1172/JCI30227
J. Kamegai, H. Tamura, T. Shimizu, S. Ishii, H. Sugihara, I. Wakabayashi, Chronic central infusion of ghrelin increases hypothalamic neuropeptide Y and Agouti-related protein mRNA levels and body weight in rats. Diabetes 50(11), 2438–2443 (2001)
M. Shintani, Y. Ogawa, K. Ebihara, M. Aizawa-Abe, F. Miyanaga, K. Takaya, T. Hayashi, G. Inoue, K. Hosoda, M. Kojima, K. Kangawa, K. Nakao, Ghrelin, an endogenous growth hormone secretagogue, is a novel orexigenic peptide that antagonizes leptin action through the activation of hypothalamic neuropeptide Y/Y1 receptor pathway. Diabetes 50(2), 227–232 (2001)
M. Tschop, D.L. Smiley, M.L. Heiman, Ghrelin induces adiposity in rodents. Nature 407(6806), 908–913 (2000). doi:10.1038/35038090
N. Nagaya, M. Uematsu, M. Kojima, Y. Ikeda, F. Yoshihara, W. Shimizu, H. Hosoda, Y. Hirota, H. Ishida, H. Mori, K. Kangawa, Chronic administration of ghrelin improves left ventricular dysfunction and attenuates development of cardiac cachexia in rats with heart failure. Circulation 104(12), 1430–1435 (2001)
N. Nagaya, K. Kangawa, Ghrelin improves left ventricular dysfunction and cardiac cachexia in heart failure. Curr. Opin. Pharmacol. 3(2), 146–151 (2003)
N. Nagaya, K. Kangawa, Ghrelin, a novel growth hormone-releasing peptide, in the treatment of chronic heart failure. Regul. Pept. 114(2–3), 71–77 (2003)
Y. Lin, K. Matsumura, M. Fukuhara, S. Kagiyama, K. Fujii, M. Iida, Ghrelin acts at the nucleus of the solitary tract to decrease arterial pressure in rats. Hypertension 43(5), 977–982 (2004). doi:10.1161/01.HYP.0000122803.91559.55
D. Baatar, K. Patel, D.D. Taub, The effects of ghrelin on inflammation and the immune system. Mol. Cell. Endocrinol. 340(1), 44–58 (2011). doi:10.1016/j.mce.2011.04.019
N. Pandya, R. DeMott-Friberg, C.Y. Bowers, A.L. Barkan, C.A. Jaffe, Growth hormone (GH)-releasing peptide-6 requires endogenous hypothalamic GH-releasing hormone for maximal GH stimulation. J. Clin. Endocrinol. Metab. 83(4), 1186–1189 (1998). doi:10.1210/jcem.83.4.4711
E. Ghigo, G. Aimaretti, E. Arvat, F. Camanni, Growth hormone-releasing hormone combined with arginine or growth hormone secretagogues for the diagnosis of growth hormone deficiency in adults. Endocrine 15(1), 29–38 (2001). doi:10.1385/ENDO:15:1:029
E. Ghigo, E. Arvat, G. Muccioli, F. Camanni, Growth hormone-releasing peptides. Eur. J. Endocrinol. 136(5), 445–460 (1997)
V. Guerlavais, D. Boeglin, D. Mousseaux, C. Oiry, A. Heitz, R. Deghenghi, V. Locatelli, A. Torsello, C. Ghe, F. Catapano, G. Muccioli, J.C. Galleyrand, J.A. Fehrentz, J. Martinez, New active series of growth hormone secretagogues. J. Med. Chem. 46(7), 1191–1203 (2003). doi:10.1021/jm020985q
R. Deghenghi, M.M. Cananzi, A. Torsello, C. Battisti, E.E. Muller, V. Locatelli, GH-releasing activity of Hexarelin, a new growth hormone releasing peptide, in infant and adult rats. Life Sci. 54(18), 1321–1328 (1994)
A.V. Kaisary, W.G. Bowsher, D.A. Gillatt, J.B. Anderson, P.R. Malone, B.P. Imbimbo, Pharmacodynamics of a long acting depot preparation of avorelin in patients with prostate cancer. Avorelin Study Group. J. Urol. 162(6), 2019–2023 (1999)
M. Korbonits, G. Kaltsas, L.A. Perry, A.B. Grossman, J.P. Monson, G.M. Besser, P.J. Trainer, Hexarelin as a test of pituitary reserve in patients with pituitary disease. Clin. Endocrinol. 51(3), 369–375 (1999)
M. Gasperi, G. Aimaretti, G. Scarcello, G. Corneli, C. Cosci, E. Arvat, E. Martino, E. Ghigo, Low dose hexarelin and growth hormone (GH)-releasing hormone as a diagnostic tool for the diagnosis of GH deficiency in adults: comparison with insulin-induced hypoglycemia test. J. Clin. Endocrinol. Metab. 84(8), 2633–2637 (1999). doi:10.1210/jcem.84.8.5904
Z. Laron, J. Frenkel, R. Deghenghi, S. Anin, B. Klinger, A. Silbergeld, Intranasal administration of the GHRP hexarelin accelerates growth in short children. Clin. Endocrinol. 43(5), 631–635 (1995)
V. Locatelli, G. Rossoni, F. Schweiger, A. Torsello, V. De Gennaro Colonna, M. Bernareggi, R. Deghenghi, E.E. Muller, F. Berti, Growth hormone-independent cardioprotective effects of hexarelin in the rat. Endocrinology 140(9), 4024–4031 (1999). doi:10.1210/endo.140.9.6948
G. Rossoni, V. Locatelli, V. De GennaroColonna, A. Torsello, F. Schweiger, M. Boghen, M. Nilsson, M. Bernareggi, E.E. Muller, F. Berti, Growth hormone and hexarelin prevent endothelial vasodilator dysfunction in aortic rings of the hypophysectomized rat. J. Cardiovasc. Pharmacol. 34(3), 454–460 (1999)
V. Bodart, M. Febbraio, A. Demers, N. McNicoll, P. Pohankova, A. Perreault, T. Sejlitz, E. Escher, R.L. Silverstein, D. Lamontagne, H. Ong, CD36 mediates the cardiovascular action of growth hormone-releasing peptides in the heart. Circ. Res. 90(8), 844–849 (2002)
A. Rodrigue-Way, A. Demers, H. Ong, A. Tremblay, A growth hormone-releasing peptide promotes mitochondrial biogenesis and a fat burning-like phenotype through scavenger receptor CD36 in white adipocytes. Endocrinology 148(3), 1009–1018 (2007). doi:10.1210/en.2006-0975
H. Zhao, G. Liu, Q. Wang, L. Ding, H. Cai, H. Jiang, Z. Xin, Effect of ghrelin on human endothelial cells apoptosis induced by high glucose. Biochem. Biophys. Res. Commun. 362(3), 677–681 (2007). doi:10.1016/j.bbrc.2007.08.021
M.S. Kim, C.Y. Yoon, P.G. Jang, Y.J. Park, C.S. Shin, H.S. Park, J.W. Ryu, Y.K. Pak, J.Y. Park, K.U. Lee, S.Y. Kim, H.K. Lee, Y.B. Kim, K.S. Park, The mitogenic and antiapoptotic actions of ghrelin in 3T3-L1 adipocytes. Mol. Endocrinol. 18(9), 2291–2301 (2004). doi:10.1210/me.2003-0459
W.G. Li, D. Gavrila, X. Liu, L. Wang, S. Gunnlaugsson, L.L. Stoll, M.L. McCormick, C.D. Sigmund, C. Tang, N.L. Weintraub, Ghrelin inhibits proinflammatory responses and nuclear factor-kappaB activation in human endothelial cells. Circulation 109(18), 2221–2226 (2004). doi:10.1161/01.CIR.0000127956.43874.F2
I. Johansson, S. Destefanis, N.D. Aberg, M.A. Aberg, K. Blomgren, C. Zhu, C. Ghe, R. Granata, E. Ghigo, G. Muccioli, P.S. Eriksson, J. Isgaard, Proliferative and protective effects of growth hormone secretagogues on adult rat hippocampal progenitor cells. Endocrinology 149(5), 2191–2199 (2008). doi:10.1210/en.2007-0733
W. Wang, D. Zhang, H. Zhao, Y. Chen, Y. Liu, C. Cao, L. Han, G. Liu, Ghrelin inhibits cell apoptosis induced by lipotoxicity in pancreatic beta-cell line. Regul. Pept. 161(1–3), 43–50 (2010). doi:10.1016/j.regpep.2009.12.017
C.E. Wrede, L.M. Dickson, M.K. Lingohr, I. Briaud, C.J. Rhodes, Protein kinase B/Akt prevents fatty acid-induced apoptosis in pancreatic beta-cells (INS-1). J. Biol. Chem. 277(51), 49676–49684 (2002). doi:10.1074/jbc.M208756200
J. Buteau, W. El-Assaad, C.J. Rhodes, L. Rosenberg, E. Joly, M. Prentki, Glucagon-like peptide-1 prevents beta cell glucolipotoxicity. Diabetologia 47(5), 806–815 (2004). doi:10.1007/s00125-004-1379-6
E. Karaskov, C. Scott, L. Zhang, T. Teodoro, M. Ravazzola, A. Volchuk, Chronic palmitate but not oleate exposure induces endoplasmic reticulum stress, which may contribute to INS-1 pancreatic beta-cell apoptosis. Endocrinology 147(7), 3398–3407 (2006). doi:10.1210/en.2005-1494
D. Kawamori, H. Kaneto, Y. Nakatani, T.A. Matsuoka, M. Matsuhisa, M. Hori, Y. Yamasaki, The forkhead transcription factor Foxo1 bridges the JNK pathway and the transcription factor PDX-1 through its intracellular translocation. J. Biol. Chem. 281(2), 1091–1098 (2006). doi:10.1074/jbc.M508510200
G. Solinas, W. Naugler, F. Galimi, M.S. Lee, M. Karin, Saturated fatty acids inhibit induction of insulin gene transcription by JNK-mediated phosphorylation of insulin-receptor substrates. Proc. Natl. Acad. Sci. USA 103(44), 16454–16459 (2006). doi:10.1073/pnas.0607626103
Y. Zhang, B. Ying, L. Shi, H. Fan, D. Yang, D. Xu, Y. Wei, X. Hu, Y. Zhang, X. Zhang, T. Wang, D. Liu, L. Dou, G. Chen, F. Jiang, F. Wen, Ghrelin inhibit cell apoptosis in pancreatic beta cell line HIT-T15 via mitogen-activated protein kinase/phosphoinositide 3-kinase pathways. Toxicology 237(1–3), 194–202 (2007). doi:10.1016/j.tox.2007.05.013
R. Granata, F. Settanni, L. Biancone, L. Trovato, R. Nano, F. Bertuzzi, S. Destefanis, M. Annunziata, M. Martinetti, F. Catapano, C. Ghe, J. Isgaard, M. Papotti, E. Ghigo, G. Muccioli, Acylated and unacylated ghrelin promote proliferation and inhibit apoptosis of pancreatic beta-cells and human islets: involvement of 3′,5′-cyclic adenosine monophosphate/protein kinase A, extracellular signal-regulated kinase 1/2, and phosphatidyl inositol 3-Kinase/Akt signaling. Endocrinology 148(2), 512–529 (2007). doi:10.1210/en.2006-0266
W. Wang, Y. Liu, Y. Chen, C. Cao, Y. Xiang, D. Zhang, L. Han, H. Zhao, G. Liu, Inhibition of Foxo1 mediates protective effects of ghrelin against lipotoxicity in MIN6 pancreatic beta-cells. Peptides 31(2), 307–314 (2010). doi:10.1016/j.peptides.2009.11.011
T. Irako, T. Akamizu, H. Hosoda, H. Iwakura, H. Ariyasu, K. Tojo, N. Tajima, K. Kangawa, Ghrelin prevents development of diabetes at adult age in streptozotocin-treated newborn rats. Diabetologia 49(6), 1264–1273 (2006). doi:10.1007/s00125-006-0226-3
A.M. Ackermann, M. Gannon, Molecular regulation of pancreatic beta-cell mass development, maintenance, and expansion. J. Mol. Endocrinol. 38(1–2), 193–206 (2007). doi:10.1677/JME-06-0053
R. Granata, M. Volante, F. Settanni, C. Gauna, C. Ghe, M. Annunziata, B. Deidda, I. Gesmundo, T. Abribat, A.J. van der Lely, G. Muccioli, E. Ghigo, M. Papotti, Unacylated ghrelin and obestatin increase islet cell mass and prevent diabetes in streptozotocin-treated newborn rats. J. Mol. Endocrinol. 45(1), 9–17 (2010). doi:10.1677/JME-09-0141
R. Granata, F. Settanni, M. Julien, R. Nano, G. Togliatto, A. Trombetta, D. Gallo, L. Piemonti, M.F. Brizzi, T. Abribat, A.J. van Der Lely, E. Ghigo, Des-acyl ghrelin fragments and analogues promote survival of pancreatic beta-cells and human pancreatic islets and prevent diabetes in streptozotocin-treated rats. J. Med. Chem. 55(6), 2585–2596 (2012). doi:10.1021/jm201223m
S. Bonner-Weir, D.F. Trent, G.C. Weir, Partial pancreatectomy in the rat and subsequent defect in glucose-induced insulin release. J. Clin. Investig. 71(6), 1544–1553 (1983)
R. Rafaeloff, G.L. Pittenger, S.W. Barlow, X.F. Qin, B. Yan, L. Rosenberg, W.P. Duguid, A.I. Vinik, Cloning and sequencing of the pancreatic islet neogenesis associated protein (INGAP) gene and its expression in islet neogenesis in hamsters. J. Clin. Investig. 99(9), 2100–2109 (1997). doi:10.1172/JCI119383
D. Gu, N. Sarvetnick, Epithelial cell proliferation and islet neogenesis in IFN-g transgenic mice. Development 118(1), 33–46 (1993)
J.F. List, J.F. Habener, Glucagon-like peptide 1 agonists and the development and growth of pancreatic beta-cells. Am. J. Physiol. Endocrinol. Metab. 286(6), E875–E881 (2004). doi:10.1152/ajpendo.00007.2004
F. Sanvito, P.L. Herrera, J. Huarte, A. Nichols, R. Montesano, L. Orci, J.D. Vassalli, TGF-beta 1 influences the relative development of the exocrine and endocrine pancreas in vitro. Development 120(12), 3451–3462 (1994)
Y.Q. Zhang, H. Zhang, A. Maeshima, H. Kurihara, J. Miyagawa, T. Takeuchi, I. Kojima, Up-regulation of the expression of activins in the pancreatic duct by reduction of the beta-cell mass. Endocrinology 143(9), 3540–3547 (2002). doi:10.1210/en.2002-220089
J.T. Hill, T.L. Mastracci, C. Vinton, M.L. Doyle, K.R. Anderson, Z.L. Loomis, J.M. Schrunk, A.D. Minic, K.R. Prabakar, A. Pugliese, Y. Sun, R.G. Smith, L. Sussel, Ghrelin is dispensable for embryonic pancreatic islet development and differentiation. Regul. Pept. 157(1–3), 51–56 (2009). doi:10.1016/j.regpep.2009.02.013
N. Wierup, S. Yang, R.J. McEvilly, H. Mulder, F. Sundler, Ghrelin is expressed in a novel endocrine cell type in developing rat islets and inhibits insulin secretion from INS-1 (832/13) cells. J. Histochem. Cytochem. 52(3), 301–310 (2004)
M. Kerem, B. Salman, S. Ozsoy, H. Pasaoglu, A. Bedirli, R. Haziroglu, T.U. Yilmaz, Exogenous ghrelin enhances endocrine and exocrine regeneration in pancreatectomized rats. J. Gastrointest. Surg. 13(4), 775–783 (2009). doi:10.1007/s11605-008-0778-2
J.H. Nielsen, E.D. Galsgaard, A. Moldrup, B.N. Friedrichsen, N. Billestrup, J.A. Hansen, Y.C. Lee, C. Carlsson, Regulation of beta-cell mass by hormones and growth factors. Diabetes 50(Suppl 1), S25–S29 (2001)
X. Ma, Y. Lin, L. Lin, G. Qin, F.A. Pereira, M.W. Haymond, N.F. Butte, Y. Sun, Ablation of ghrelin receptor in leptin-deficient ob/ob mice has paradoxical effects on glucose homeostasis when compared with ablation of ghrelin in ob/ob mice. Am. J. Physiol. Endocrinol. Metab. 303(3), E422–E431 (2012). doi:10.1152/ajpendo.00576.2011
A. Salehi, C. de la Dornonville Cour, R. Hakanson, I. Lundquist, Effects of ghrelin on insulin and glucagon secretion: a study of isolated pancreatic islets and intact mice. Regul. Pept. 118(3), 143–150 (2004). doi:10.1016/j.regpep.2003.12.001
H.M. Lee, G. Wang, E.W. Englander, M. Kojima, G.H. Greeley Jr, Ghrelin, a new gastrointestinal endocrine peptide that stimulates insulin secretion: enteric distribution, ontogeny, influence of endocrine, and dietary manipulations. Endocrinology 143(1), 185–190 (2002). doi:10.1210/endo.143.1.8602
K. Spiegel, E. Tasali, R. Leproult, N. Scherberg, E. Van Cauter, Twenty-four-hour profiles of acylated and total ghrelin: relationship with glucose levels and impact of time of day and sleep. J. Clin. Endocrinol. Metab. 96(2), 486–493 (2011). doi:10.1210/jc.2010-1978
A.C. Heijboer, A.M. van den Hoek, E.T. Parlevliet, L.M. Havekes, J.A. Romijn, H. Pijl, E.P. Corssmit, Ghrelin differentially affects hepatic and peripheral insulin sensitivity in mice. Diabetologia 49(4), 732–738 (2006). doi:10.1007/s00125-006-0138-2
K.M. Heppner, J. Tong, H. Kirchner, R. Nass, M.H. Tschop, The ghrelin O-acyltransferase-ghrelin system: a novel regulator of glucose metabolism. Curr. Opin. Endocrinol. Diabetes Obes. 18(1), 50–55 (2011). doi:10.1097/MED.0b013e328341e1d3
H. Iwakura, K. Hosoda, C. Son, J. Fujikura, T. Tomita, M. Noguchi, H. Ariyasu, K. Takaya, H. Masuzaki, Y. Ogawa, T. Hayashi, G. Inoue, T. Akamizu, H. Hosoda, M. Kojima, H. Itoh, S. Toyokuni, K. Kangawa, K. Nakao, Analysis of rat insulin II promoter-ghrelin transgenic mice and rat glucagon promoter-ghrelin transgenic mice. J. Biol. Chem. 280(15), 15247–15256 (2005). doi:10.1074/jbc.M411358200
C. Gauna, F.M. Meyler, J.A. Janssen, P.J. Delhanty, T. Abribat, P. van Koetsveld, L.J. Hofland, F. Broglio, E. Ghigo, A.J. van der Lely, Administration of acylated ghrelin reduces insulin sensitivity, whereas the combination of acylated plus unacylated ghrelin strongly improves insulin sensitivity. J. Clin. Endocrinol. Metab. 89(10), 5035–5042 (2004). doi:10.1210/jc.2004-0363
L. Pacifico, E. Poggiogalle, F. Costantino, C. Anania, F. Ferraro, F. Chiarelli, C. Chiesa, Acylated and nonacylated ghrelin levels and their associations with insulin resistance in obese and normal weight children with metabolic syndrome. Eur. J. Endocrinol. 161(6), 861–870 (2009). doi:10.1530/EJE-09-0375
L. Huang, Y. Tong, F. Zhang, Q. Yang, D. Li, S. Xie, Y. Li, H. Cao, L. Tang, X. Zhang, N. Tong, Increased acyl ghrelin but decreased total ghrelin and unacyl ghrelin in Chinese Han people with impaired fasting glucose combined with impaired glucose tolerance. Peptides 60, 86–94 (2014). doi:10.1016/j.peptides.2014.07.022
P. Lucidi, G. Murdolo, C. Di Loreto, A. De Cicco, N. Parlanti, C. Fanelli, F. Santeusanio, G.B. Bolli, P. De Feo, Ghrelin is not necessary for adequate hormonal counterregulation of insulin-induced hypoglycemia. Diabetes 51(10), 2911–2914 (2002)
M.F. Saad, B. Bernaba, C.M. Hwu, S. Jinagouda, S. Fahmi, E. Kogosov, R. Boyadjian, Insulin regulates plasma ghrelin concentration. J. Clin. Endocrinol. Metab. 87(8), 3997–4000 (2002). doi:10.1210/jcem.87.8.8879
Q. Wang, C. Liu, A. Uchida, J.C. Chuang, A. Walker, T. Liu, S. Osborne-Lawrence, B.L. Mason, C. Mosher, E.D. Berglund, J.K. Elmquist, J.M. Zigman, Arcuate AgRP neurons mediate orexigenic and glucoregulatory actions of ghrelin. Mol. Metab. 3(1), 64–72 (2014). doi:10.1016/j.molmet.2013.10.001
V. De Gennaro-Colonna, G. Rossoni, D. Cocchi, A.E. Rigamonti, F. Berti, E.E. Muller, Endocrine, metabolic and cardioprotective effects of hexarelin in obese Zucker rats. J. Endocrinol. 166(3), 529–536 (2000)
R.M. Frieboes, H. Murck, P. Maier, T. Schier, F. Holsboer, A. Steiger, Growth hormone-releasing peptide-6 stimulates sleep, growth hormone, ACTH and cortisol release in normal man. Neuroendocrinology 61(5), 584–589 (1995)
R.G. Clark, G.B. Thomas, D.L. Mortensen, W.B. Won, Y.H. Ma, E.E. Tomlinson, K.M. Fairhall, I.C. Robinson, Growth hormone secretagogues stimulate the hypothalamic-pituitary-adrenal axis and are diabetogenic in the Zucker diabetic fatty rat. Endocrinology 138(10), 4316–4323 (1997). doi:10.1210/endo.138.10.5424
Y. Date, M. Nakazato, S. Hashiguchi, K. Dezaki, M.S. Mondal, H. Hosoda, M. Kojima, K. Kangawa, T. Arima, H. Matsuo, T. Yada, S. Matsukura, Ghrelin is present in pancreatic alpha-cells of humans and rats and stimulates insulin secretion. Diabetes 51(1), 124–129 (2002)
E. Adeghate, A.S. Ponery, Ghrelin stimulates insulin secretion from the pancreas of normal and diabetic rats. J. Neuroendocrinol. 14(7), 555–560 (2002)
K. Dezaki, H. Hosoda, M. Kakei, S. Hashiguchi, M. Watanabe, K. Kangawa, T. Yada, Endogenous ghrelin in pancreatic islets restricts insulin release by attenuating Ca2 + signaling in beta-cells: implication in the glycemic control in rodents. Diabetes 53(12), 3142–3151 (2004)
K. Dezaki, H. Sone, M. Koizumi, M. Nakata, M. Kakei, H. Nagai, H. Hosoda, K. Kangawa, T. Yada, Blockade of pancreatic islet-derived ghrelin enhances insulin secretion to prevent high-fat diet-induced glucose intolerance. Diabetes 55(12), 3486–3493 (2006). doi:10.2337/db06-0878
Y. Wang, M. Nishi, A. Doi, T. Shono, Y. Furukawa, T. Shimada, H. Furuta, H. Sasaki, K. Nanjo, Ghrelin inhibits insulin secretion through the AMPK-UCP2 pathway in beta cells. FEBS Lett. 584(8), 1503–1508 (2010). doi:10.1016/j.febslet.2010.02.069
E.M. Egido, J. Rodriguez-Gallardo, R.A. Silvestre, J. Marco, Inhibitory effect of ghrelin on insulin and pancreatic somatostatin secretion. Eur. J. Endocrinol. 146(2), 241–244 (2002)
M. Colombo, S. Gregersen, J. Xiao, K. Hermansen, Effects of ghrelin and other neuropeptides (CART, MCH, orexin A and B, and GLP-1) on the release of insulin from isolated rat islets. Pancreas 27(2), 161–166 (2003)
C. Gauna, P.J. Delhanty, M.O. van Aken, J.A. Janssen, A.P. Themmen, L.J. Hofland, M. Culler, F. Broglio, E. Ghigo, A.J. van der Lely, Unacylated ghrelin is active on the INS-1E rat insulinoma cell line independently of the growth hormone secretagogue receptor type 1a and the corticotropin releasing factor 2 receptor. Mol. Cell. Endocrinol. 251(1–2), 103–111 (2006). doi:10.1016/j.mce.2006.03.040
M.K. Reimer, G. Pacini, B. Ahren, Dose-dependent inhibition by ghrelin of insulin secretion in the mouse. Endocrinology 144(3), 916–921 (2003). doi:10.1210/en.2002-220819
S.K. Chacko, M.W. Haymond, Y. Sun, J.C. Marini, P.J. Sauer, X. Ma, A.L. Sunehag, Effect of ghrelin on glucose regulation in mice. Am. J. Physiol. Endocrinol. Metab. 302(9), E1055–E1062 (2012). doi:10.1152/ajpendo.00445.2011
R.M. Kiewiet, M.O. van Aken, K. van der Weerd, P. Uitterlinden, A.P. Themmen, L.J. Hofland, Y.B. de Rijke, P.J. Delhanty, E. Ghigo, T. Abribat, A.J. van der Lely, Effects of acute administration of acylated and unacylated ghrelin on glucose and insulin concentrations in morbidly obese subjects without overt diabetes. Eur. J. Endocrinol. 161(4), 567–573 (2009). doi:10.1530/EJE-09-0339
J. Tong, R.L. Prigeon, H.W. Davis, M. Bidlingmaier, S.E. Kahn, D.E. Cummings, M.H. Tschop, D. D’Alessio, Ghrelin suppresses glucose-stimulated insulin secretion and deteriorates glucose tolerance in healthy humans. Diabetes 59(9), 2145–2151 (2010). doi:10.2337/db10-0504
F. Tassone, F. Broglio, S. Destefanis, S. Rovere, A. Benso, C. Gottero, F. Prodam, R. Rossetto, C. Gauna, A.J. van der Lely, E. Ghigo, M. Maccario, Neuroendocrine and metabolic effects of acute ghrelin administration in human obesity. J. Clin. Endocrinol. Metab. 88(11), 5478–5483 (2003). doi:10.1210/jc.2003-030564
F. Broglio, C. Gottero, A. Benso, F. Prodam, S. Destefanis, C. Gauna, M. Maccario, R. Deghenghi, A.J. van der Lely, E. Ghigo, Effects of ghrelin on the insulin and glycemic responses to glucose, arginine, or free fatty acids load in humans. J. Clin. Endocrinol. Metab. 88(9), 4268–4272 (2003). doi:10.1210/jc.2002-021940
F. Broglio, A. Benso, C. Gottero, F. Prodam, C. Gauna, L. Filtri, E. Arvat, A.J. van der Lely, R. Deghenghi, E. Ghigo, Non-acylated ghrelin does not possess the pituitaric and pancreatic endocrine activity of acylated ghrelin in humans. J. Endocrinol. Invest. 26(3), 192–196 (2003)
E.T. Vestergaard, C.B. Djurhuus, J. Gjedsted, S. Nielsen, N. Moller, J.J. Holst, J.O. Jorgensen, O. Schmitz, Acute effects of ghrelin administration on glucose and lipid metabolism. J. Clin. Endocrinol. Metab. 93(2), 438–444 (2008). doi:10.1210/jc.2007-2018
E.T. Vestergaard, L.C. Gormsen, N. Jessen, S. Lund, T.K. Hansen, N. Moller, J.O. Jorgensen, Ghrelin infusion in humans induces acute insulin resistance and lipolysis independent of growth hormone signaling. Diabetes 57(12), 3205–3210 (2008). doi:10.2337/db08-0025
S.S. Damjanovic, N.M. Lalic, P.M. Pesko, M.S. Petakov, A. Jotic, D. Miljic, K.S. Lalic, L. Lukic, M. Djurovic, V.B. Djukic, Acute effects of ghrelin on insulin secretion and glucose disposal rate in gastrectomized patients. J. Clin. Endocrinol. Metab. 91(7), 2574–2581 (2006). doi:10.1210/jc.2005-1482
E.T. Vestergaard, N. Moller, J.O. Jorgensen, Acute peripheral tissue effects of ghrelin on interstitial levels of glucose, glycerol, and lactate: a microdialysis study in healthy human subjects. Am. J. Physiol. Endocrinol. Metab. 304(12), E1273–E1280 (2013). doi:10.1152/ajpendo.00662.2012
A.D. Patel, S.A. Stanley, K.G. Murphy, G.S. Frost, J.V. Gardiner, A.S. Kent, N.E. White, M.A. Ghatei, S.R. Bloom, Ghrelin stimulates insulin-induced glucose uptake in adipocytes. Regul. Pept. 134(1), 17–22 (2006). doi:10.1016/j.regpep.2005.11.001
V. Ott, M. Fasshauer, A. Dalski, B. Meier, N. Perwitz, H.H. Klein, M. Tschop, J. Klein, Direct peripheral effects of ghrelin include suppression of adiponectin expression. Hormone Metab. Res. 34(11–12), 640–645 (2002). doi:10.1055/s-2002-38261
A. Demers, V. Caron, A. Rodrigue-Way, W. Wahli, H. Ong, A. Tremblay, A concerted kinase interplay identifies PPARgamma as a molecular target of ghrelin signaling in macrophages. PLoS ONE 4(11), e7728 (2009). doi:10.1371/journal.pone.0007728
R. Avallone, A. Demers, A. Rodrigue-Way, K. Bujold, D. Harb, S. Anghel, W. Wahli, S. Marleau, H. Ong, A. Tremblay, A growth hormone-releasing peptide that binds scavenger receptor CD36 and ghrelin receptor up-regulates sterol transporters and cholesterol efflux in macrophages through a peroxisome proliferator-activated receptor gamma-dependent pathway. Mol. Endocrinol. 20(12), 3165–3178 (2006). doi:10.1210/me.2006-0146
S. Marleau, D. Harb, K. Bujold, R. Avallone, K. Iken, Y. Wang, A. Demers, M.G. Sirois, M. Febbraio, R.L. Silverstein, A. Tremblay, H. Ong, EP 80317, a ligand of the CD36 scavenger receptor, protects apolipoprotein E-deficient mice from developing atherosclerotic lesions. FASEB J. 19(13), 1869–1871 (2005). doi:10.1096/fj.04-3253fje
M. Lehrke, M.A. Lazar, The many faces of PPARgamma. Cell 123(6), 993–999 (2005). doi:10.1016/j.cell.2005.11.026
A. Chawla, W.A. Boisvert, C.H. Lee, B.A. Laffitte, Y. Barak, S.B. Joseph, D. Liao, L. Nagy, P.A. Edwards, L.K. Curtiss, R.M. Evans, P. Tontonoz, A PPAR gamma-LXR-ABCA1 pathway in macrophages is involved in cholesterol efflux and atherogenesis. Mol. Cell 7(1), 161–171 (2001)
A. Castrillo, P. Tontonoz, Nuclear receptors in macrophage biology: at the crossroads of lipid metabolism and inflammation. Annu. Rev. Cell Dev. Biol. 20, 455–480 (2004). doi:10.1146/annurev.cellbio.20.012103.134432
A.C. Li, K.K. Brown, M.J. Silvestre, T.M. Willson, W. Palinski, C.K. Glass, Peroxisome proliferator-activated receptor gamma ligands inhibit development of atherosclerosis in LDL receptor-deficient mice. J. Clin. Investig. 106(4), 523–531 (2000). doi:10.1172/JCI10370
A.R. Collins, W.P. Meehan, U. Kintscher, S. Jackson, S. Wakino, G. Noh, W. Palinski, W.A. Hsueh, R.E. Law, Troglitazone inhibits formation of early atherosclerotic lesions in diabetic and nondiabetic low density lipoprotein receptor-deficient mice. Arterioscler. Thromb. Vasc. Biol. 21(3), 365–371 (2001)
A. Rodrigue-Way, V. Caron, S. Bilodeau, S. Keil, M. Hassan, E. Levy, G.A. Mitchell, A. Tremblay, Scavenger receptor CD36 mediates inhibition of cholesterol synthesis via activation of the PPARgamma/PGC-1alpha pathway and Insig1/2 expression in hepatocytes. FASEB J. 28(4), 1910–1923 (2014). doi:10.1096/fj.13-240168
H. Hui, F. Dotta, U. Di Mario, R. Perfetti, Role of caspases in the regulation of apoptotic pancreatic islet beta-cells death. J. Cell. Physiol. 200(2), 177–200 (2004). doi:10.1002/jcp.20021
P. Maechler, C.B. Wollheim, Mitochondrial function in normal and diabetic beta-cells. Nature 414(6865), 807–812 (2001). doi:10.1038/414807a
S. Krauss, C.Y. Zhang, L. Scorrano, L.T. Dalgaard, J. St-Pierre, S.T. Grey, B.B. Lowell, Superoxide-mediated activation of uncoupling protein 2 causes pancreatic beta cell dysfunction. J. Clin. Investig. 112(12), 1831–1842 (2003). doi:10.1172/JCI19774
M. Anello, R. Lupi, D. Spampinato, S. Piro, M. Masini, U. Boggi, S. Del Prato, A.M. Rabuazzo, F. Purrello, P. Marchetti, Functional and morphological alterations of mitochondria in pancreatic beta cells from type 2 diabetic patients. Diabetologia 48(2), 282–289 (2005). doi:10.1007/s00125-004-1627-9
C.Y. Zhang, G. Baffy, P. Perret, S. Krauss, O. Peroni, D. Grujic, T. Hagen, A.J. Vidal-Puig, O. Boss, Y.B. Kim, X.X. Zheng, M.B. Wheeler, G.I. Shulman, C.B. Chan, B.B. Lowell, Uncoupling protein-2 negatively regulates insulin secretion and is a major link between obesity, beta cell dysfunction, and type 2 diabetes. Cell 105(6), 745–755 (2001)
Y. Teshima, M. Akao, S.P. Jones, E. Marban, Uncoupling protein-2 overexpression inhibits mitochondrial death pathway in cardiomyocytes. Circ. Res. 93(3), 192–200 (2003). doi:10.1161/01.RES.0000085581.60197.4D
Q. Zhang, W.D. Huang, X.Y. Lv, Y.M. Yang, Ghrelin protects H9c2 cells from hydrogen peroxide-induced apoptosis through NF-kappaB and mitochondria-mediated signaling. Eur. J. Pharmacol. 654(2), 142–149 (2011). doi:10.1016/j.ejphar.2010.12.011
J. Chmielewska, D. Szczepankiewicz, M. Skrzypski, D. Kregielska, M.Z. Strowski, K.W. Nowak, Ghrelin but not obestatin regulates insulin secretion from INS1 beta cell line via UCP2-dependent mechanism. J. Biol. Regul. Homeost. Agents 24(4), 397–402 (2010)
E. ElEter, A. AlTuwaijiri, H. Hagar, M. Arafa, In vivo and in vitro antioxidant activity of ghrelin: attenuation of gastric ischemic injury in the rat. J. Gastroenterol. Hepatol. 22(11), 1791–1799 (2007). doi:10.1111/j.1440-1746.2006.04696.x
B.D. Obay, E. Tasdemir, C. Tumer, H. Bilgin, M. Atmaca, Dose dependent effects of ghrelin on pentylenetetrazole-induced oxidative stress in a rat seizure model. Peptides 29(3), 448–455 (2008). doi:10.1016/j.peptides.2007.11.020
A. Kawczynska-Drozdz, R. Olszanecki, J. Jawien, T. Brzozowski, W.W. Pawlik, R. Korbut, T.J. Guzik, Ghrelin inhibits vascular superoxide production in spontaneously hypertensive rats. Am. J. Hypertens. 19(7), 764–767 (2006). doi:10.1016/j.amjhyper.2006.01.022
S.O. Iseri, G. Sener, B. Saglam, F. Ercan, N. Gedik, B.C. Yegen, Ghrelin alleviates biliary obstruction-induced chronic hepatic injury in rats. Regul. Pept. 146(1–3), 73–79 (2008). doi:10.1016/j.regpep.2007.08.014
X. Zhang, C. Chen, A new insight of mechanisms, diagnosis and treatment of diabetic cardiomyopathy. Endocrine 41(3), 398–409 (2012). doi:10.1007/s12020-012-9623-1
S.N. Verhagen, A.M. Wassink, Y. van der Graaf, Y. Gorter, P.M. Visseren, F.L. Visseren, S.S. Group, Insulin resistance increases the occurrence of new cardiovascular events in patients with manifest arterial disease without known diabetes. The SMART study. Cardiovasc. Diabetol. 10, 100 (2011). doi:10.1186/1475-2840-10-100
E. Bonora, G. Targher, G. Formentini, F. Calcaterra, S. Lombardi, F. Marini, L. Zenari, F. Saggiani, M. Poli, S. Perbellini, A. Raffaelli, L. Gemma, L. Santi, R.C. Bonadonna, M. Muggeo, The metabolic syndrome is an independent predictor of cardiovascular disease in Type 2 diabetic subjects. Prospective data from the Verona Diabetes Complications Study. Diabetic Med. 21(1), 52–58 (2004)
A. Dei Cas, V. Spigoni, V. Ridolfi, M. Metra, Diabetes and chronic heart failure: from diabetic cardiomyopathy to therapeutic approach. Endocr. Metab. Immune Disord. Drug Targets 13(1), 38–50 (2013)
L. van Heerebeek, N. Hamdani, M.L. Handoko, I. Falcao-Pires, R.J. Musters, K. Kupreishvili, A.J. Ijsselmuiden, C.G. Schalkwijk, J.G. Bronzwaer, M. Diamant, A. Borbely, J. van der Velden, G.J. Stienen, G.J. Laarman, H.W. Niessen, W.J. Paulus, Diastolic stiffness of the failing diabetic heart: importance of fibrosis, advanced glycation end products, and myocyte resting tension. Circulation 117(1), 43–51 (2008). doi:10.1161/CIRCULATIONAHA.107.728550
W.D. Gao, Y. Liu, E. Marban, Selective effects of oxygen free radicals on excitation-contraction coupling in ventricular muscle. Implications for the mechanism of stunned myocardium. Circulation 94(10), 2597–2604 (1996)
M. Zabalgoitia, M.F. Ismaeil, L. Anderson, F.A. Maklady, Prevalence of diastolic dysfunction in normotensive, asymptomatic patients with well-controlled type 2 diabetes mellitus. Am. J. Cardiol. 87(3), 320–323 (2001)
J.K. Boyer, S. Thanigaraj, K.B. Schechtman, J.E. Perez, Prevalence of ventricular diastolic dysfunction in asymptomatic, normotensive patients with diabetes mellitus. Am. J. Cardiol. 93(7), 870–875 (2004). doi:10.1016/j.amjcard.2003.12.026
Y. Ma, L. Zhang, J.N. Edwards, B.S. Launikonis, C. Chen, Growth hormone secretagogues protect mouse cardiomyocytes from in vitro ischemia/reperfusion injury through regulation of intracellular calcium. PLoS ONE 7(4), e35265 (2012). doi:10.1371/journal.pone.0035265
F. Berti, E. Muller, V. De GennaroColonna, G. Rossoni, Hexarelin exhibits protective activity against cardiac ischaemia in hearts from growth hormone-deficient rats. Growth Hormone IGF Res. 8(Suppl B), 149–152 (1998)
S. Frascarelli, S. Ghelardoni, S. Ronca-Testoni, R. Zucchi, Effect of ghrelin and synthetic growth hormone secretagogues in normal and ischemic rat heart. Basic Res. Cardiol. 98(6), 401–405 (2003). doi:10.1007/s00395-003-0434-7
G. Bisi, V. Podio, M.R. Valetto, F. Broglio, G. Bertuccio, G. Del Rio, E. Arvat, M.F. Boghen, R. Deghenghi, G. Muccioli, H. Ong, E. Ghigo, Acute cardiovascular and hormonal effects of GH and hexarelin, a synthetic GH-releasing peptide, in humans. J. Endocrinol. Invest. 22(4), 266–272 (1999)
X. Xu, F. Ding, J. Pang, X. Gao, R.K. Xu, W. Hao, J.M. Cao, C. Chen, Chronic administration of hexarelin attenuates cardiac fibrosis in the spontaneously hypertensive rat. Am. J. Physiol. Heart Circ. Physiol. 303(6), H703–H711 (2012). doi:10.1152/ajpheart.00257.2011
L. Chang, Y. Ren, X. Liu, W.G. Li, J. Yang, B. Geng, N.L. Weintraub, C. Tang, Protective effects of ghrelin on ischemia/reperfusion injury in the isolated rat heart. J. Cardiovasc. Pharmacol. 43(2), 165–170 (2004)
G.G. Zhang, X. Teng, Y. Liu, Y. Cai, Y.B. Zhou, X.H. Duan, J.Q. Song, Y. Shi, C.S. Tang, X.H. Yin, Y.F. Qi, Inhibition of endoplasm reticulum stress by ghrelin protects against ischemia/reperfusion injury in rat heart. Peptides 30(6), 1109–1116 (2009). doi:10.1016/j.peptides.2009.03.024
V. De Gennaro Colonna, G. Rossoni, M. Bernareggi, E.E. Muller, F. Berti, Cardiac ischemia and impairment of vascular endothelium function in hearts from growth hormone-deficient rats: protection by hexarelin. Eur. J. Pharmacol. 334(2–3), 201–207 (1997)
A. Torsello, E. Bresciani, G. Rossoni, R. Avallone, G. Tulipano, D. Cocchi, I. Bulgarelli, R. Deghenghi, F. Berti, V. Locatelli, Ghrelin plays a minor role in the physiological control of cardiac function in the rat. Endocrinology 144(5), 1787–1792 (2003). doi:10.1210/en.2002-221048
Y. Mao, T. Tokudome, I. Kishimoto, K. Otani, M. Miyazato, K. Kangawa, One dose of oral hexarelin protects chronic cardiac function after myocardial infarction. Peptides 56, 156–162 (2014). doi:10.1016/j.peptides.2014.04.004
M. Aragno, R. Mastrocola, C. Ghe, E. Arnoletti, E. Bassino, G. Alloatti, G. Muccioli, Obestatin induced recovery of myocardial dysfunction in type 1 diabetic rats: underlying mechanisms. Cardiovasc. Diabetol. 11, 129 (2012). doi:10.1186/1475-2840-11-129
Y. Mao, T. Tokudome, I. Kishimoto, K. Otani, H. Hosoda, C. Nagai, N. Minamino, M. Miyazato, K. Kangawa, Hexarelin treatment in male ghrelin knockout mice after myocardial infarction. Endocrinology 154(10), 3847–3854 (2013). doi:10.1210/en.2013-1291
Y. Ma, L. Zhang, B.S. Launikonis, C. Chen, Growth hormone secretagogues preserve the electrophysiological properties of mouse cardiomyocytes isolated from in vitro ischemia/reperfusion heart. Endocrinology 153(11), 5480–5490 (2012). doi:10.1210/en.2012-1404
J.J. Pang, R.K. Xu, X.B. Xu, J.M. Cao, C. Ni, W.L. Zhu, K. Asotra, M.C. Chen, C. Chen, Hexarelin protects rat cardiomyocytes from angiotensin II-induced apoptosis in vitro. Am. J. Physiol. Heart Circ. Physiol. 286(3), H1063–H1069 (2004). doi:10.1152/ajpheart.00648.2003
T. Soeki, K. Koshiba, T. Niki, K. Kusunose, K. Yamaguchi, H. Yamada, T. Wakatsuki, M. Shimabukuro, K. Minakuchi, I. Kishimoto, K. Kangawa, M. Sata, Effect of ghrelin on autonomic activity in healthy volunteers. Peptides 62C, 1–5 (2014). doi:10.1016/j.peptides.2014.09.015
J. Isgaard, Ghrelin and the cardiovascular system. Endocrinol. Dev. 25, 83–90 (2013). doi:10.1159/000346056
T. Soeki, I. Kishimoto, D.O. Schwenke, T. Tokudome, T. Horio, M. Yoshida, H. Hosoda, K. Kangawa, Ghrelin suppresses cardiac sympathetic activity and prevents early left ventricular remodeling in rats with myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 294(1), H426–H432 (2008). doi:10.1152/ajpheart.00643.2007
T. Soeki, T. Niki, E. Uematsu, S. Bando, T. Matsuura, K. Kusunose, T. Ise, Y. Ueda, N. Tomita, K. Yamaguchi, K. Koshiba, S. Yagi, D. Fukuda, Y. Taketani, T. Iwase, H. Yamada, T. Wakatsuki, M. Akaike, M. Shimabukuro, I. Kishimoto, K. Kangawa, M. Sata, Ghrelin protects the heart against ischemia-induced arrhythmias by preserving connexin-43 protein. Heart Vessel. 28(6), 795–801 (2013). doi:10.1007/s00380-013-0333-2
D.L. Lerner, K.A. Yamada, R.B. Schuessler, J.E. Saffitz, Accelerated onset and increased incidence of ventricular arrhythmias induced by ischemia in Cx43-deficient mice. Circulation 101(5), 547–552 (2000)
K.E. Wiley, A.P. Davenport, Comparison of vasodilators in human internal mammary artery: ghrelin is a potent physiological antagonist of endothelin-1. Br. J. Pharmacol. 136(8), 1146–1152 (2002). doi:10.1038/sj.bjp.0704815
N. Nagaya, K. Miyatake, M. Uematsu, H. Oya, W. Shimizu, H. Hosoda, M. Kojima, N. Nakanishi, H. Mori, K. Kangawa, Hemodynamic, renal, and hormonal effects of ghrelin infusion in patients with chronic heart failure. J. Clin. Endocrinol. Metab. 86(12), 5854–5859 (2001). doi:10.1210/jcem.86.12.8115
C.X. Huang, M.J. Yuan, H. Huang, G. Wu, Y. Liu, S.B. Yu, H.T. Li, T. Wang, Ghrelin inhibits post-infarct myocardial remodeling and improves cardiac function through anti-inflammation effect. Peptides 30(12), 2286–2291 (2009). doi:10.1016/j.peptides.2009.09.004
G. Bisi, V. Podio, M.R. Valetto, F. Broglio, G. Bertuccio, G. Aimaretti, E. Pelosi, G. Del Rio, G. Muccioli, H. Ong, M.F. Boghen, R. Deghenghi, E. Ghigo, Cardiac effects of hexarelin in hypopituitary adults. Eur. J. Pharmacol. 381(1), 31–38 (1999)
M. Imazio, M. Bobbio, F. Broglio, A. Benso, V. Podio, M.R. Valetto, G. Bisi, E. Ghigo, G.P. Trevi, GH-independent cardiotropic activities of hexarelin in patients with severe left ventricular dysfunction due to dilated and ischemic cardiomyopathy. Eur. J. Heart Fail. 4(2), 185–191 (2002)
M. Enomoto, N. Nagaya, M. Uematsu, H. Okumura, E. Nakagawa, F. Ono, H. Hosoda, H. Oya, M. Kojima, K. Kanmatsuse, K. Kangawa, Cardiovascular and hormonal effects of subcutaneous administration of ghrelin, a novel growth hormone-releasing peptide, in healthy humans. Clin. Sci. 105(4), 431–435 (2003). doi:10.1042/CS20030184
Y. Mao, T. Tokudome, I. Kishimoto, The cardiovascular action of hexarelin. J. Geriatr. Cardiol. 11(3), 253–258 (2014). doi:10.11909/j.issn.1671-5411.2014.03.007
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mosa, R.M.H., Zhang, Z., Shao, R. et al. Implications of ghrelin and hexarelin in diabetes and diabetes-associated heart diseases. Endocrine 49, 307–323 (2015). https://doi.org/10.1007/s12020-015-0531-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-015-0531-z