Abstract
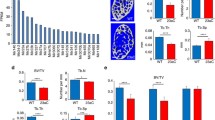
MicroRNAs (miRNAs) have been identified to play important functions during osteoblast proliferation, differentiation, and apoptosis. The miR-17~92 cluster is highly conserved in all vertebrates. Loss-of-function of the miR-17-92 cluster results in smaller embryos and immediate postnatal death of all animals. Germline hemizygous deletions of MIR17HG are accounted for microcephaly, short stature, and digital abnormalities in a few cases of Feingold syndrome. These reports indicate that miR-17~92 may play important function in skeletal development and mature. To determine the functional roles of miR-17~92 in bone metabolism as well as osteoblast proliferation and differentiation. Murine embryonic stem cells D3 and osteoprogenitor cell line MC3T3-E1 were induced to differentiate into osteoblasts; the expression of miR-17-92 was assayed by quantitative real-time RT-PCR. The skeletal phenotypes were assayed in mice heterozygous for miR-17~92 (miR-17~92 +/Δ). To determine the possibly direct function of miR-17~92 in bone cells, osteoblasts from miR-17~92 +/Δ mice were investigated by ex vivo cell culture. miR-17, miR-92a, and miR-20a within miR-17-92 cluster were expressed at high level in bone tissue and osteoblasts. The expression of miR-17-92 was down-regulated along with osteoblast differentiation, the lowest level was found in mature osteoblasts. Compared to wildtype controls, miR-17-92 +/Δ mice showed significantly lower trabecular and cortical bone mineral density, bone volume and trabecular number at 10 weeks old. mRNA expression of Runx2 and type I collagen was significantly lower in bone from miR-17-92 +/Δ mice. Osteoblasts from miR-17-92 +/Δ mice showed lower proliferation rate, ALP activity and less calcification. Our research suggests that the miR-17-92 cluster critically regulates bone metabolism, and this regulation is mostly through its function in osteoblasts.





Similar content being viewed by others
References
L.P. Lim, N.C. Lau, P. Garrett-Engele, A. Grimson, J.M. Schelter, J. Castle, D.P. Bartel, P.S. Linsley, J.M. Johnson, Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature 433(7027), 769–773 (2005). doi:10.1038/nature03315
A. Ota, H. Tagawa, S. Karnan, S. Tsuzuki, A. Karpas, S. Kira, Y. Yoshida, M. Seto, Identification and characterization of a novel gene, C13orf25, as a target for 13q31-q32 amplification in malignant lymphoma. Cancer Res. 64(9), 3087–3095 (2004)
S. Volinia, G.A. Calin, C.G. Liu, S. Ambs, A. Cimmino, F. Petrocca, R. Visone, M. Iorio, C. Roldo, M. Ferracin, R.L. Prueitt, N. Yanaihara, G. Lanza, A. Scarpa, A. Vecchione, M. Negrini, C.C. Harris, C.M. Croce, A microRNA expression signature of human solid tumors defines cancer gene targets. Proc. Natl. Acad. Sci. USA 103(7), 2257–2261 (2006). doi:0510565103
F. Petrocca, A. Vecchione, C.M. Croce, Emerging role of miR-106b-25/miR-17-92 clusters in the control of transforming growth factor beta signaling. Cancer Res. 68(20), 8191–8194 (2008). doi:10.1158/0008-5472.CAN-08-1768
L. He, J.M. Thomson, M.T. Hemann, E. Hernando-Monge, D. Mu, S. Goodson, S. Powers, C. Cordon-Cardo, S.W. Lowe, G.J. Hannon, S.M. Hammond, A microRNA polycistron as a potential human oncogene. Nature 435(7043), 828–833 (2005). doi:nature03552
K.A. O’Donnell, E.A. Wentzel, K.I. Zeller, C.V. Dang, J.T. Mendell, c-Myc-regulated microRNAs modulate E2F1 expression. Nature 435(7043), 839–843 (2005). doi:10.1038/nature03677
J. Xiang, J. Wu, Feud or friend? The role of the miR-17-92 cluster in tumorigenesis. Curr. Genomics 11(2), 129–135 (2010). doi:10.2174/138920210790886853
B.D. Aguda, Y. Kim, M.G. Piper-Hunter, A. Friedman, C.B. Marsh, MicroRNA regulation of a cancer network: consequences of the feedback loops involving miR-17-92, E2F, and Myc. Proc. Natl. Acad. Sci. USA 105(50), 19678–19683 (2008). doi:10.1073/pnas.0811166106
M. Inomata, H. Tagawa, Y.M. Guo, Y. Kameoka, N. Takahashi, K. Sawada, MicroRNA-17-92 down-regulates expression of distinct targets in different B-cell lymphoma subtypes. Blood 113(2), 396–402 (2009). doi:10.1182/blood-2008-07-163907
F. Kuhnert, C.J. Kuo, miR-17-92 angiogenesis micromanagement. Blood 115(23), 4631–4633 (2010). doi:10.1182/blood-2010-03-276428
A. Ventura, A.G. Young, M.M. Winslow, L. Lintault, A. Meissner, S.J. Erkeland, J. Newman, R.T. Bronson, D. Crowley, J.R. Stone, R. Jaenisch, P.A. Sharp, T. Jacks, Targeted deletion reveals essential and overlapping functions of the miR-17 through 92 family of miRNA clusters. Cell 132(5), 875–886 (2008). doi:10.1016/j.cell.2008.02.019
Y. Lu, J.M. Thomson, H.Y. Wong, S.M. Hammond, B.L. Hogan, Transgenic over-expression of the microRNA miR-17-92 cluster promotes proliferation and inhibits differentiation of lung epithelial progenitor cells. Dev. Biol. 310(2), 442–453 (2007). doi:S0012-1606(07)01252-3
L. de Pontual, E. Yao, P. Callier, L. Faivre, V. Drouin, S. Cariou, A. Van Haeringen, D. Genevieve, A. Goldenberg, M. Oufadem, S. Manouvrier, A. Munnich, J.A. Vidigal, M. Vekemans, S. Lyonnet, A. Henrion-Caude, A. Ventura, J. Amiel, Germline deletion of the miR-17 approximately 92 cluster causes skeletal and growth defects in humans. Nat. Genet. 43(10), 1026–1030 (2011). doi:10.1038/ng.915
Y. Liu, W. Liu, C. Hu, Z. Xue, G. Wang, B. Ding, H. Luo, L. Tang, X. Kong, X. Chen, N. Liu, Y. Ding, Y. Jin, MiR-17 modulates osteogenic differentiation through a coherent feed-forward loop in mesenchymal stem cells isolated from periodontal ligaments of patients with periodontitis. Stem Cells 29(11), 1804–1816 (2011)
J.E. Onyia, B. Miller, J. Hulman, J. Liang, R. Galvin, C. Frolik, S. Chandrasekhar, A.K. Harvey, J. Bidwell, J. Herring, J.M. Hock, Proliferating cells in the primary spongiosa express phenotype in vitro. Bone 20(2), 93–100 (1997). doi:S875632829600350X
K.J. Livak, T.D. Schmittgen, Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4), 402–408 (2001). doi:10.1006/meth.2001.1262
T.C. Doetschman, H. Eistetter, M. Katz, W. Schmidt, R. Kemler, The in vitro development of blastocyst-derived embryonic stem cell lines: formation of visceral yolk sac, blood islands and myocardium. J. Embryol. Exp. Morphol. 87, 27–45 (1985)
L. Duplomb, M. Dagouassat, P. Jourdon, D. Heymann, Concise review: embryonic stem cells: a new tool to study osteoblast and osteoclast differentiation. Stem Cells 25(3), 544–552 (2007). doi:2006-0395
L. Duplomb, M. Dagouassat, P. Jourdon, D. Heymann, Differentiation of osteoblasts from mouse embryonic stem cells without generation of embryoid body. In Vitro Cell. Dev. Biol. Anim. 43(1), 21–24 (2007). doi:10.1007/s11626-006-9010-4
T. Xia, Y. Liang, J. Ma, M. Li, M. Gong, X. Yu, Loss-of-function of SHARPIN causes an osteopenic phenotype in mice. Endocrine 39(2), 104–112 (2011)
M. Suzuki, H. Kobayashi, N. Kanayama, Y. Saga, M. Suzuki, C.Y. Lin, R.B. Dickson, T. Terao, Inhibition of tumor invasion by genomic down-regulation of matriptase through suppression of activation of receptor-bound pro-urokinase. J. Biol. Chem. 279(15), 14899–14908 (2004)
S.H. Lee, J.S. Kim, H.K. Jun, H.R. Lee, D. Lee, B.K. Choi, The major outer membrane protein of a periodontopathogen induces IFN-beta and IFN-stimulated genes in monocytes via lipid raft and TANK-binding kinase 1/IFN regulatory factor-3. J. Immunol. 182(9), 5823–5835 (2009)
Z. Li, M.Q. Hassan, M. Jafferji, R.I. Aqeilan, R. Garzon, C.M. Croce, A.J. van Wijnen, J.L. Stein, G.S. Stein, J.B. Lian, Biological functions of miR-29b contribute to positive regulation of osteoblast differentiation. J. Biol. Chem. 284(23), 15676–15684 (2009). doi:10.1074/jbc.M809787200M809787200
A. Palmieri, F. Pezzetti, G. Brunelli, M. Martinelli, L. Scapoli, M. Arlotti, E. Masiero, F. Carinci, Medpor regulates osteoblast’s microRNAs. Biomed. Mater. Eng. 18(2), 91–97 (2008)
Y. Mizuno, Y. Tokuzawa, Y. Ninomiya, K. Yagi, Y. Yatsuka-Kanesaki, T. Suda, T. Fukuda, T. Katagiri, Y. Kondoh, T. Amemiya, H. Tashiro, Y. Okazaki, miR-210 promotes osteoblastic differentiation through inhibition of AcvR1b. FEBS Lett. 583(13), 2263–2268 (2009). doi:10.1016/j.febslet.2009.06.006
J. Zhang, Q. Tu, L.F. Bonewald, X. He, G. Stein, J. Lian, J. Chen, Effects of miR-335-5p in modulating osteogenic differentiation by specifically downregulating Wnt antagonist DKK1. J. Bone Miner. Res. 26(8), 1953–1963 (2011). doi:10.1002/jbmr.377
R. Hu, W. Liu, H. Li, L. Yang, C. Chen, Z.Y. Xia, L.J. Guo, H. Xie, H.D. Zhou, X.P. Wu, X.H. Luo, A Runx2/miR-3960/miR-2861 regulatory feedback loop during mouse osteoblast differentiation. J. Biol. Chem. 286(14), 12328–12339 (2011). doi:10.1074/jbc.M110.176099
Z. Li, M.Q. Hassan, S. Volinia, A.J. van Wijnen, J.L. Stein, C.M. Croce, J.B. Lian, G.S. Stein, A microRNA signature for a BMP2-induced osteoblast lineage commitment program. Proc. Natl. Acad. Sci. USA 105(37), 13906–13911 (2008). doi:10.1073/pnas.0804438105
J. Huang, L. Zhao, L. Xing, D. Chen, MicroRNA-204 regulates Runx2 protein expression and mesenchymal progenitor cell differentiation. Stem Cells 28(2), 357–364 (2010). doi:10.1002/stem.288
Y. Zhang, R.L. Xie, C.M. Croce, J.L. Stein, J.B. Lian, A.J. van Wijnen, G.S. Stein, A program of microRNAs controls osteogenic lineage progression by targeting transcription factor Runx2. Proc. Natl. Acad. Sci. USA 108(24), 9863–9868 (2011). doi:10.1073/pnas.1018493108
L. Guo, J. Xu, J. Qi, L. Zhang, J. Wang, J. Liang, N. Qian, H. Zhou, L. Wei, L. Deng, MicroRNA-17~92a upregulation by estrogen leads to Bim targeting and inhibition of osteoblasts apoptosis. J. Cell Sci. (2012). doi:10.1242/jcs.117515
R.M. Boggs, J.A. Moody, C.R. Long, K.L. Tsai, K.E. Murphy, Identification, amplification and characterization of miR-17-92 from canine tissue. Gene 404(1–2), 25–30 (2007). doi:10.1016/j.gene.2007.08.015
J.K. Van Camp, S. Beckers, D. Zegers, E. Boudin, T.L. Nielsen, M. Andersen, G. Roef, Y. Taes, K. Brixen, W. Van Hul, Genetic association study of WNT10B polymorphisms with BMD and adiposity parameters in Danish and Bmaleselgian. Endocrine (2013). doi:10.1007/s12020-012-9869-7
Y.H. Deng, L. Zhao, M.J. Zhang, C.M. Pan, S.X. Zhao, H.Y. Zhao, L.H. Sun, B. Tao, H.D. Song, W.Q. Wang, G. Ning, J.M. Liu, The influence of the genetic and non-genetic factors on bone mineral density and osteoporotic fractures in Chinese women. Endocrine 43(1), 127–135 (2013). doi:10.1007/s12020-012-9726-8
F. Rivadeneira, Hunting osteoporosis susceptibility genes: bigger is better but diverse is also welcome. Endocrine 43(1), 6–7 (2013). doi:10.1007/s12020-012-9804-y
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No. 81072190 to Yu X, No. 81101920 to Ma J), the Science &Technology Department of Sichuan Province (2010SZ0168 to Yu X), and the Ministry of Education of the People Republic of China ([2011]1139 to Yu X).
Author information
Authors and Affiliations
Corresponding author
Additional information
Mingliang Zhou and Junrong Ma have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhou, M., Ma, J., Chen, S. et al. MicroRNA-17-92 cluster regulates osteoblast proliferation and differentiation. Endocrine 45, 302–310 (2014). https://doi.org/10.1007/s12020-013-9986-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-013-9986-y