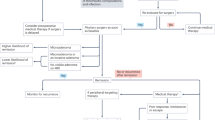
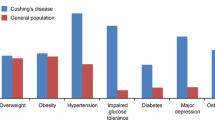
Abstract
Cushing’s disease is a rare chronic disease caused by a pituitary adenoma, which leads to excess secretion of adrenocorticotropic hormone (ACTH). The over-production of ACTH leads to hyperstimulation of the adrenal glands and a chronic excess of cortisol, resulting in the signs and symptoms of a severe clinical state (Cushing’s syndrome) that leads to significant morbidity, negative impacts on the patient’s quality of life, and, if untreated, increased mortality. The management of patients with Cushing’s disease is complicated by the heterogeneity of the condition, with signs and symptoms that overlap with those of other diseases, and high subclinical incidence rates. Controversies surrounding the tests used for screening and identifying patients with Cushing’s disease add to the challenge of patient management. Surgical intervention to remove the adenoma is the first-line treatment for patients with Cushing’s disease, but medical therapies are useful in patients who relapse or are unsuitable for surgery. The recent introduction of pasireotide, the first pituitary-directed medical therapy, expands the number of treatment options available for patients with Cushing’s disease. This state-of-the-art review aims to provide an overview of the most recent scientific research and clinical information regarding Cushing’s disease. Continuing research into improving the diagnosis and treatment of Cushing’s disease will help to optimize patient management.


Similar content being viewed by others
References
S.S. Praw, A.P. Heaney, Medical treatment of Cushing’s disease: overview and recent findings. Int. J. Gen. Med. 29, 209–217 (2009)
J. Newell-Price, X. Bertagna, A.B. Grossman et al., Cushing’s syndrome. Lancet 367, 1605–1617 (2006)
J. Lindholm, S. Juul, J.O. Jørgensen et al., Incidence and late prognosis of Cushing’s syndrome: a population-based study. J. Clin. Endocrinol. Metab. 86, 117–123 (2001)
J. Etxabe, J.A. Vazquez, Morbidity and mortality in Cushing’s disease: an epidemiological approach. Clin. Endocrinol. (Oxf) 40, 479–484 (1994)
R. Pivonello, M.C. De Martino, M. De Leo et al., Cushing’s syndrome. Endocrinol. Metab. Clin. North Am. 37, 135–149 (2008)
M. Boscaro, G. Arnaldi, Approach to the patient with possible Cushing’s syndrome. J. Clin. Endocrinol. Metab. 94, 3121–3131 (2009)
C. Steffensen, A.M. Bak, K.Z. Rubeck et al., Epidemiology of Cushing’s syndrome. Neuroendocrinology 92, 1–5 (2010)
L. Guignat, J. Bertherat, The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline: commentary from a European perspective. Eur. J. Endocrinol. 163, 9–13 (2010)
J. Kreutzer, R. Fahlbusch, Diagnosis and treatment of pituitary tumors. Curr. Opin. Neurol. 17, 693–703 (2004)
A.S. Kanter, A.O. Diallo, J.A. Jane Jr et al., Single-center experience with pediatric Cushing’s disease. J. Neurosurg. 103, 413–420 (2005)
D.F. Kelly, Transsphenoidal surgery for Cushing’s disease: a review of success rates, remission predictors, management of failed surgery, and Nelson’s Syndrome. Neurosurg. Focus 23, E5 (2007)
J. Newell-Price, A.B. Grossman, Differential diagnosis of Cushing’s syndrome. Arq. Bras. Endocrinol. Metabol. 51, 1199–1206 (2007)
B.M. Biller, A.B. Grossman, P.M. Stewart et al., Treatment of adrenocorticotropin-dependent Cushing’s syndrome: a consensus statement. J. Clin. Endocrinol. Metab. 93, 2454–2462 (2008)
D.M. Prevedello, N. Pouratian, J. Sherman et al., Management of Cushing’s disease: outcome in patients with microadenoma detected on pituitary magnetic resonance imaging. J. Neurosurg. 109, 751–759 (2008)
G. Arnaldi, M. Boscaro, New treatment guidelines on Cushing’s disease. F1000 Med. Rep. 1, 64 (2009)
L.S. Blevins Jr, N. Sanai, S. Kunwar et al., An approach to the management of patients with residual Cushing’s disease. J. Neurooncol. 94, 313–319 (2009)
B.M. Biller, A. Colao, S. Petersenn et al., Prolactinomas, Cushing’s disease and acromegaly: debating the role of medical therapy for secretory pituitary adenomas. BMC Endocr. Disord. 10, 10 (2010)
N. Sonino, F. Fallo, G.A. Fava, Psychosomatic aspects of Cushing’s syndrome. Rev. Endocr. Metab. Disord. 11, 95–104 (2010)
A. Rizk, J. Honegger, M. Milian et al., Treatment options in Cushing’s disease. Clin. Med. Insights. Oncol. 6, 75–84 (2012)
F. Castinetti, M. Nagai, H. Dufour et al., Gamma knife radiosurgery is a successful adjunctive treatment in Cushing’s disease. Eur. J. Endocrinol. 156, 91–98 (2007)
J.K. Devin, G.S. Allen, A.J. Cmelak et al., The efficacy of linear accelerator radiosurgery in the management of patients with Cushing’s disease. Stereotact. Funct. Neurosurg. 82, 254–262 (2004)
M. Boschetti, M. De Lucchi, M. Giusti et al., Partial visual recovery from radiation-induced optic neuropathy after hyperbaric oxygen therapy in a patient with Cushing disease. Eur. J. Endocrinol. 154, 813–818 (2006)
C.N. Dang, P. Trainer, Pharmacological management of Cushing’s syndrome: an update. Arq. Bras. Endocrinol. Metabol. 51, 1339–1348 (2007)
A. Colao, S. Petersenn, J. Newell-Price et al., A 12-month phase 3 study of pasireotide in Cushing’s disease. N. Engl. J. Med. 366, 914–924 (2012)
J. Bertherat, W. Ludlam, R. Pivonello et al., Long-term use of pasireotide in Cushing’s disease: 24-month safety results from a randomized Phase III study. Endocr. Abstr. 27, P1405 (2012)
M. Yaneva, S. Vandeva, S. Zacharieva et al., Genetics of Cushing’s syndrome. Neuroendocrinology 92, 6–10 (2010)
Genetics Home Reference. http://ghr.nlm.nih.gov/ (2013). Accessed 5 July 2013
A. Lacroix, ACTH-independent macronodular adrenal hiperplasia. Best practice and research. Clin. Endocrinol. Metab. 23, 245–259 (2009)
S.M. Webb, X. Badia, M.J. Barahona et al., Evaluation of health-related quality of life in patients with Cushing’s syndrome with a new questionnaire. Eur. J. Endocrinol. 158, 623–630 (2008)
A. Santos, E. Resmini, M.A. Martínez et al., Quality of life in patients with pituitary tumors. Curr. Opin. Endocrinol. Diabetes Obes. 16, 299–303 (2009)
A. Colao, A. Cozzolino, R. Pivonello, Quality of life in patients with Cushing’s disease: a modern approach. Clin. Endocrinol. 76, 776–777 (2012)
L.K. Nieman, B.M. Biller, J.W. Findling et al., The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 93, 1526–1540 (2008)
L. Katznelson, J.S. Bogan, J.R. Trob et al., Biochemical assessment of Cushing’s disease in patients with corticotroph macroadenomas. J. Clin. Endocrinol. Metab. 83, 1619–1623 (1998)
R.C. Bakker, P.R. Gallas, J.A. Romijn et al., Cushing’s syndrome complicated by multiple opportunistic infections. J. Endocrinol. Invest. 21, 329–333 (1998)
A. Faggiano, R. Pivonello, D. Melis et al., Nephrolithiasis in Cushing’s disease: prevalence, etiopathogenesis, and modification after disease cure. J. Clin. Endocrinol. Metab. 88, 2076–2080 (2003)
L. Tauchmanovà, R. Pivonello, M.C. De Martino et al., Effects of sex steroids on bone in women with subclinical or overt endogenous hypercortisolism. Eur. J. Endocrinol. 157, 359–366 (2007)
S.G. Kosseifi, D.N. Nassour, M.A. Shaikh et al., Nodular pulmonary histoplasmosis in Cushing’s disease: a case report and literature review. Tenn. Med. 100, 44–46 (2007)
A.W. van der Eerden, M. den Heijer, W.J. Oyen et al., Cushing’s syndrome and bone mineral density: lowest Z scores in young patients. Neth. J. Med. 65, 137–141 (2007)
G. Kaltsas, P. Makras, Skeletal diseases in Cushing’s syndrome: osteoporosis versus arthropathy. Neuroendocrinology 92, 60–64 (2010)
R.A. Feelders, S.J. Pulgar, A. Kempel, A.M. Pereira, The burden of Cushing’s disease: clinical and health-related quality of life aspects. Eur. J. Endocrinol. 167, 311–2620 (2012)
M.D. Bronstein, L.R. Salgado, N.R. de Castro Musolino, Medical management of pituitary adenomas: the special case of management of the pregnant woman. Pituitary 5, 99–107 (2002)
L. Tauchmanovà, R. Rossi, B. Biondi et al., Patients with subclinical Cushing’s syndrome due to adrenal adenoma have increased cardiovascular risk. J. Clin. Endocrinol. Metab. 87, 4872–4878 (2002)
R. Pivonello, A. Faggiano, G. Lombardi et al., The metabolic syndrome and cardiovascular risk in Cushing’s syndrome. Endocrinol. Metab. Clin. North Am. 34, 327–339 (2005)
M. De Leo, R. Pivonello, R.S. Auriemma et al., Cardiovascular disease in Cushing’s syndrome: heart versus vasculature. Neuroendocrinology 92, 50–54 (2010)
F. Fallo, G. Famoso, D. Capizzi et al., Coronary microvascular function in patients with Cushing’s syndrome. Endocrine 43, 206–213 (2013)
S. Savastano, R. Pivonello, A. Colao, Bariatric surgery for obesity and hidden Cushing syndrome. Surg. Obes. Relat. Dis. 5, 121–122 (2009)
P. Chanson, S. Salenave, Metabolic syndrome in Cushing’s syndrome. Neuroendocrinology 92, 96–101 (2010)
E.B. Geer, W. Shen, D. Gallagher et al., MRI assessment of lean and adipose tissue distribution in female patients with Cushing’s disease. Clin. Endocrinol. 73, 469–475 (2010)
O.M. Dekkers, E. Horváth-Puhó, J.O. Jørgensen et al., Multisystem morbidity and mortality in Cushing’s syndrome: a cohort study. J. Clin. Endocrinol. Metab. 98, 2277–2284 (2013)
G. Arnaldi, A. Angeli, A.B. Atkinson et al., Diagnosis and complications of Cushing’s syndrome: a consensus statement. J. Clin. Endocrinol. Metab. 88, 5593–5602 (2003)
D. Graversen, P. Vestergaard, K. Stochholm et al., Mortality in Cushing’s syndrome: a systematic review and meta-analysis. Eur. J. Intern. Med. 23, 278–282 (2012)
R.N. Clayton, D. Raskauskiene, R.C. Reulen et al., Mortality and morbidity in Cushing’s disease over 50 years in Stoke-on-Trent, UK: audit and meta-analysis of literature. J. Clin. Endocrinol. Metab. 96, 632–642 (2011)
O.M. Dekkers, N.R. Biermasz, A.M. Pereira et al., Mortality in patients treated for Cushing’s disease is increased, compared with patients treated for nonfunctioning pituitary macroadenoma. J. Clin. Endocrinol. Metab. 92, 976–981 (2007)
E. Valassi, I. Crespo, A. Santos, S.M. Webb, Clinical consequences of Cushing’s syndrome. Pituitary 15, 319–329 (2012)
R. Pivonello, M.C. De Martino, M. De Leo et al., Cushing’s syndrome: aftermath of the cure. Arq. Bras. Endocrinol. Metabol. 51, 1381–1391 (2007)
A. Faggiano, R. Pivonello, S. Spiezia et al., Cardiovascular risk factors and common carotid artery caliber and stiffness in patients with Cushing’s disease during active disease and 1 year after disease remission. J. Clin. Endocrinol. Metab. 88, 2527–2533 (2003)
A. Colao, R. Pivonello, S. Spiezia et al., Persistence of increased cardiovascular risk in patients with Cushing’s disease after five years of successful cure. J. Clin. Endocrinol. Metab. 84, 2664–2672 (1999)
C. Di Somma, R. Pivonello, S. Loche et al., Effect of 2 years of cortisol normalization on the impaired bone mass and turnover in adolescent and adult patients with Cushing’s disease: a prospective study. Clin Endocrinol (Oxf). 58, 302–308 (2003)
A. Faggiano, R. Pivonello, M. Filippella et al., Spine abnormalities and damage in patients cured from Cushing’s disease. Pituitary 4, 153–161 (2001)
O. Ragnarsson, G. Johannsson, Management of endocrine disease: Cushing’s syndrome: a structured short- and long-term management plan for patients in remission. Eur. J. Endocrinol. 169, R139–R152 (2013)
C. Garcia, B.M. Biller, A. Klibanski, The role of the clinical laboratory in the diagnosis of Cushing syndrome. Am. J. Clin. Pathol. 120, S38–S45 (2003)
J. Newell-Price, P. Trainer, M. Besser et al., The diagnosis and differential diagnosis of Cushing’s syndrome and pseudo-Cushing’s states. Endocr. Rev. 19, 647–672 (1998)
L.F. Chan, H.L. Storr, A.B. Grossman et al., Pediatric Cushing’s syndrome: clinical features, diagnosis, and treatment. Arq. Bras. Endocrinol. Metabol. 51, 1261–1271 (2007)
F. Lumachi, P. Marchesi, D. Miotto et al., CT and MR imaging of the adrenal glands in cortisol-secreting tumors. Anticancer Res. 31, 2923–2926 (2011)
D.C. Aron, Cushing’s syndrome: why is diagnosis so difficult? Rev. Endocr. Metab. Disord. 11, 105–116 (2010)
S. Hasinski, Assessment of adrenal glucocorticoid function. Which tests are appropriate for screening? Postgrad. Med. 104(61–64), 69–72 (1998)
D. Vassiliadi, S. Tsagarakis, Unusual causes of Cushing’s syndrome. Arq. Bras. Endocrinol. Metabol. 51, 1245–1252 (2007)
T.B. Carroll, J.W. Findling, Cushing’s syndrome of nonpituitary causes. Curr. Opin. Endocrinol. Diabetes Obes. 16, 308–315 (2009)
M.M. Foisy, E.M. Yakiwchuk, I. Chiu et al., Adrenal suppression and Cushing’s syndrome secondary to an interaction between ritonavir and fluticasone: a review of the literature. HIV Med. 9, 389–396 (2008)
J. Lo, S.K. Grinspoon, Adrenal function in HIV infection. Curr. Opin. Endocrinol. Diabetes Obes. 17, 205–209 (2010)
C. Bernecker, T.B. West, G. Mansmann et al., Hypercortisolism caused by ritonavir associated inhibition of CYP 3A4 under inhalative glucocorticoid therapy. 2 case reports and a review of the literature. Exp. Clin. Endocrinol. Diabetes 120, 125–127 (2012)
C. Levin, H.I. Maibach, Topical corticosteroid-induced adrenocortical insufficiency: clinical implications. Am. J. Clin. Dermatol. 3, 141–147 (2002)
B. Coureau, J.F. Bussières, S. Tremblay, Cushing’s syndrome induced by misuse of moderate- to high-potency topical corticosteroids. Ann. Pharmacother. 42, 1903–1907 (2008)
T. Tempark, V. Phatarakijnirund, S. Chatproedprai et al., Exogenous Cushing’s syndrome due to topical corticosteroid application: case report and review literature. Endocrine 38, 328–334 (2010)
E. Castela, E. Archier, S. Devaux et al., Topical corticosteroids in plaque psoriasis: a systematic review of risk of adrenal axis suppression and skin atrophy. J. Eur. Acad. Dermatol. Venereol. 26, 47–51 (2012)
L. Vilar, Mda C. Freitas, M. Faria et al., Pitfalls in the diagnosis of Cushing’s syndrome. Arq. Bras. Endocrinol. Metabol. 51, 1207–1216 (2007)
F. Pecori Giraldi, Recent challenges in the diagnosis of Cushing’s syndrome. Horm. Res. 71, 123–127 (2009)
D.G. Morris, A.B. Grossman, Dynamic tests in the diagnosis and differential diagnosis of Cushing’s syndrome. J. Endocrinol. Invest. 26, 64–73 (2003)
M.B. Elamin, M.H. Murad, R. Mullan et al., Accuracy of diagnostic tests for Cushing’s syndrome: a systematic review and metaanalyses. J. Clin. Endocrinol. Metab. 93, 1553–1562 (2008)
H. Raff, Update on late-night salivary cortisol for the diagnosis of Cushing’s syndrome: methodological considerations. Endocrine. Epub ahead of print (2013)
A. Viardot, P. Huber, J.J. Puder et al., Reproducibility of nighttime salivary cortisol and its use in the diagnosis of hypercortisolism compared with urinary free cortisol and overnight dexamethasone suppression test. J. Clin. Endocrinol. Metab. 90, 5730–5736 (2005)
S.A. Doi, J. Clark, A.W. Russell, Concordance of the late night salivary cortisol in patients with Cushing’s syndrome and elevated urine-free cortisol. Endocrine 43, 327–333 (2013)
C.A. Carrasco, M. García, M. Goycoolea et al., Reproducibility and performance of one or two samples of salivary cortisol in the diagnosis of Cushing’s syndrome using an automated immunoassay system. Endocrine 41, 487–493 (2012)
T. Psaras, M. Milian, V. Hattermann et al., Demographic factors and the presence of comorbidities do not promote early detection of Cushing’s disease and acromegaly. Exp. Clin. Endocrinol. Diabetes 119, 21–25 (2011)
A. Meyer, M. Behrend, Cushing’s syndrome: adrenalectomy and long-term results. Dig. Surg. 21, 363–370 (2004)
L. Manenschijn, J.W. Koper, E.L. van den Akker et al., A novel tool in the diagnosis and follow-up of (cyclic) Cushing’s syndrome: measurement of long-term cortisol in scalp hair. J. Clin. Endocrinol. Metab. 97, E1836–E1843 (2012)
P. Leach, A.H. Abou-Zeid, T. Kearney et al., Endoscopic transsphenoidal pituitary surgery: evidence of an operative learning curve. Neurosurgery. 67, 1205–1212 (2010)
X. Bertagna, L. Guignat, Approach to the Cushing’s disease patient with persistent/recurrent hypercortisolism after pituitary surgery. J. Clin. Endocrinol. Metab. 98, 1307–1318 (2013)
D.A. Rees, F.W. Hanna, J.S. Davies et al., Long-term follow-up results of transsphenoidal surgery for Cushing’s disease in a single centre using strict criteria for remission. Clin. Endocrinol. (Oxf) 56, 541–551 (2002)
I. Shimon, Z. Ram, Z.R. Cohen et al., Transsphenoidal surgery for Cushing’s disease: endocrinological follow-up monitoring of 82 patients. Neurosurgery. 51, 57–61 (2002)
G.D. Hammer, J.B. Tyrrell, K.R. Lamborn et al., Transsphenoidal microsurgery for Cushing’s disease: initial outcome and long-term results. J. Clin. Endocrinol. Metab. 89, 6348–6357 (2004)
A.B. Atkinson, A. Kennedy, M.I. Wiggam et al., Long-term remission rates after pituitary surgery for Cushing’s disease: the need for long-term surveillance. Clin. Endocrinol. (Oxf) 63, 549–559 (2005)
G. Rollin, N.P. Ferreira, M.A. Czepielewski, Prospective evaluation of transsphenoidal pituitary surgery in 108 patients with Cushing’s disease. Arq. Bras. Endocrinol. Metabol. 51, 1355–1361 (2007)
B.M. Hofmann, M. Hlavac, R. Martinez et al., Long-term results after microsurgery for Cushing disease: experience with 426 primary operations over 35 years. J. Neurosurg. 108, 9–18 (2008)
A.S. Mahmoud-Ahmed, J.H. Suh, Radiation therapy for Cushing’s disease: a review. Pituitary. 5, 175–180 (2002)
R.B. Friedman, E.H. Oldfield, L.K. Nieman et al., Repeat transsphenoidal surgery for Cushing’s disease. J. Neurosurg. 71, 520–527 (1989)
R.J. Benveniste, W.A. King, J. Walsh et al., Repeated transsphenoidal surgery to treat recurrent or residual pituitary adenoma. J. Neurosurg. 102, 1004–1012 (2005)
L.S. Blevins Jr, J.H. Christy, M. Khajavi et al., Outcomes of therapy for Cushing’s disease due to adrenocorticotropin-secreting pituitary macroadenomas. J. Clin. Endocrinol. Metab. 83, 63–67 (1998)
N. Sonino, M. Zielezny, G.A. Fava et al., Risk factors and long-term outcome in pituitary-dependent Cushing’s disease. J. Clin. Endocrinol. Metab. 81, 2647–2652 (1996)
G. Assié, H. Bahurel, J. Coste et al., Corticotroph tumor progression after adrenalectomy in Cushing’s Disease: a reappraisal of Nelson’s Syndrome. J. Clin. Endocrinol. Metab. 92, 172–179 (2007)
M. Boscaro, L. Barzon, F. Fallo et al., Cushing’s syndrome. Lancet 357, 783–791 (2001)
R.A. Feelders, L.J. Hofland, Medical treatment of Cushing’s disease. J. Clin. Endocrinol. Metab. 98, 425–438 (2013)
Pasireotide Summary of Product Characteristics: http://www.medicines.org.uk/emc/medicine/26746/SPC#INDICATIONS (2013). Accessed in 5 July 2013
H.A. Schmid, Pasireotide (SOM230): development, mechanism of action and potential applications. Mol. Cell. Endocrinol. 286, 69–74 (2008)
N.K. Djedovic, S.J. Rainbow, Detection of synthetic glucocorticoids by liquid chromatography–tandem mass spectrometry in patients being investigated for Cushing’s syndrome. Ann. Clin. Biochem. 48, 542–549 (2011)
A.E. Kulle, M. Welzel, P.M. Holterhus, F.G. Riepe, Principles and clinical applications of liquid chromatography–tandem mass spectrometry for the determination of adrenal and gonadal steroid hormones. J. Endocrinol. Invest. 34, 702–708 (2011)
B.M. Fong, S. Tam, K.S. Leung, Improved liquid chromatography-tandem mass spectrometry method in clinical utility for the diagnosis of Cushing’s syndrome. Anal. Bioanal. Chem. 396, 783–790 (2010)
T. Deutschbein, M. Broecker-Preuss, J. Flitsch et al., Salivary cortisol as a diagnostic tool for Cushing’s syndrome and adrenal insufficiency: improved screening by an automatic immunoassay. Eur. J. Endocrinol. 166, 613–618 (2012)
H. Raff, Cushing’s syndrome: diagnosis and surveillance using salivary cortisol. Pituitary. 15, 64–70 (2012)
R. Pivonello, D. Ferone, W.W. de Herder et al., Dopamine receptor expression and function in corticotroph pituitary tumors. J. Clin. Endocrinol. Metab. 89, 2452–2462 (2004)
C. de Bruin, R.A. Feelders, A.M. Waaijers et al., Differential regulation of human dopamine D2 and somatostatin receptor subtype expression by glucocorticoids in vitro. J. Mol. Endocrinol. 42, 47–56 (2009)
R. Pivonello, M.C. De Martino, P. Cappabianca et al., The medical treatment of Cushing’s disease: effectiveness of chronic treatment with the dopamine agonist cabergoline in patients unsuccessfully treated by surgery. J. Clin. Endocrinol. Metab. 94, 223–230 (2009)
E.F. Adams, M.J. Ashby, S.M. Brown et al., Bromocriptine suppresses ACTH secretion from human pituitary tumour cells in culture by a dopaminergic mechanism. Clin. Endocrinol. (Oxf) 15, 479–484 (1981)
C. Invitti, M. De Martin, L. Danesi et al., Effect of injectable bromocriptine in patients with Cushing’s disease. Exp. Clin. Endocrinol. Diabetes 103, 266–271 (1995)
C. de Bruin, R.A. Feelders, S.W. Lamberts et al., Somatostatin and dopamine receptors as targets for medical treatment of Cushing’s Syndrome. Rev. Endocr. Metab. Disord. 10, 91–102 (2009)
M. Rocheville, D.C. Lange, U. Kumar et al., Receptors for dopamine and somatostatin: formation of hetero-oligomers with enhanced functional activity. Science 288, 154–157 (2000)
A.P. Heaney, M. Fernando, W.H. Yong et al., Functional PPAR-gamma receptor is a novel therapeutic target for ACTH-secreting pituitary adenomas. Nat. Med. 8, 1281–1287 (2002)
X. Bertagna, R. Pivonello, M. Fleseriu, et al., Normal urinary cortisol with LCI699 in patients with Cushing’s disease: preliminary results from a proof-of-concept study. 15th Congress of the European NeuroEndocrine Association (ENEA 2012), Vienna, Austria; Klinische Endokrinologie und Stoffwechsel, 5, 22: abstract OC04 (2012)
A. Godbout, M. Manavela, K. Danilowicz et al., Cabergoline monotherapy in the long-term treatment of Cushing’s disease. Eur. J. Endocrinol. 163, 709–716 (2010)
M. Boschetti, F. Gatto, M. Arvigo et al., Role of dopamine receptors in normal and tumoral pituitary corticotropic cells and adrenal cells. Neuroendocrinology 92, 17–22 (2010)
M.D. Culler, Somatostatin-dopamine chimeras: a novel approach to treatment of neuroendocrine tumors. Horm. Metab. Res. 43, 854–857 (2011)
F. Pecori Giraldi, A.G. Ambrogio, M. Andrioli et al., Potential role for retinoic acid in patients with Cushing’s disease. J. Clin. Endocrinol. Metab. 97, 3577–3583 (2012)
M. Fleseriu, B.M. Biller, J.W. Findling et al., Mifepristone, a glucocorticoid receptor antagonist, produces clinical and metabolic benefits in patients with Cushing’s syndrome. J. Clin. Endocrinol. Metab. 97, 2039–2049 (2012)
F. Castinetti, B. Conte-Devolx, T. Brue, Medical treatment of Cushing’s syndrome: glucocorticoid receptor antagonists and mifepristone. Neuroendocrinology 92, 125–130 (2010)
F. Castinetti, T. Brue, B. Conte-Devolx, The use of the glucocorticoid receptor antagonist mifepristone in Cushing’s syndrome. Curr. Opin. Endocrinol. Diabetes. Obesity. 19, 295–299 (2012)
J.D. Carmichael, M. Fleseriu, Mifepristone: is there a place in the treatment of Cushing’s disease? Endocrine 44, 20–32 (2013)
Acknowledgments
The authors received writing/editorial support in the preparation of this manuscript provided by Excerpta Medica, funded by Novartis Oncology Region Europe.
Conflict of interest
Dr. Colao has received unrestricted grants from Novartis for research in neuroendocrine and pituitary tumors and is a member of Novartis global and European boards; Dr. Boscaro has nothing to disclose; Dr. Ferone has received unrestricted grants from Novartis for research purposes and is a member of the Novartis advisory boards; Dr. Casanueva has received advisory board fees from Novartis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Colao, A., Boscaro, M., Ferone, D. et al. Managing Cushing’s disease: the state of the art. Endocrine 47, 9–20 (2014). https://doi.org/10.1007/s12020-013-0129-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-013-0129-2