Abstract
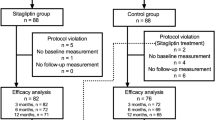
The objectives of this study is to evaluate the efficacy and safety of alogliptin versus very low fat/calorie traditional Japanese diet (non-inferiority trial) as an initial therapy for newly diagnosed, drug naïve subjects with type 2 diabetes (T2DM). Study design was prospective, randomized, non-double-blind, controlled trial. The study was conducted at outpatient units of municipal hospital. Patients were newly diagnosed, drug naïve patients who visited the outpatient units. The patients randomly received 12.5–25 mg/day alogliptin (n = 25) or severe low calorie traditional Japanese diet (n = 26). The procedure of this trial was assessed by the consolidated standards of reporting trials statement. The primary end point was the change of HbA1c at 3 months. Secondary end points included the changes of fasting blood glucose, insulin, homeostasis model assessment-R (HOMA-R), HOMA-B, body mass index (BMI), and lipid parameters. Similar, significant reductions of HbA1c levels were observed in both groups (from 10.51 to 8.74% for alogliptin and from 10.01 to 8.39% for traditional Japanese diet) without any clinically significant adverse events. In the alogliptin group, some subjects (16%) had mild hypoglycemic evens which could be managed by taking glucose drinks by themselves. HOMA-B significantly increased in both groups with varying degrees, whereas HOMA-R significantly decreased only in the Japanese diet group. Atherogenic lipids, such as, total cholesterol, non-high density lipoprotein cholesterol, and low density lipoprotein cholesterol levels significantly decreased in both groups. BMI had no change in the alogliptin group, whereas it significantly decreased in the Japanese diet group. (1) Concerning its glycemic efficacy, alogliptin is effective and non-inferior to traditional Japanese diet as an initial therapeutic option for newly diagnosed T2DM. However, regarding the reductions of body weight and insulin resistance, traditional Japanese diet is superior. (2) Both alogliptin and traditional Japanese diet have favorable effects on atherogenic lipid profiles.

Similar content being viewed by others
References
J.J. Neumiller, Clinical pharmacology of incretin therapies for type 2 diabetes mellitus: implications for treatment. Clin. Ther. 33(5), 528–576 (2011)
J.A. Lovshin, D.J. Drucker, Incretin-based therapies for type 2 diabetes mellitus. Nat. Rev. Endocrinol. 5(5), 262–269 (2009)
E. Kutoh, Sitagliptin is effective and safe as add-on to insulin in patients with absolute insulin deficiency: a case series. J. Med. Case Rep. 5, 117 (2011)
J.R. White, Alogliptin for the treatment of type 2 diabetes. Drugs Today 47(2), 99–107 (2011)
K. Takeuchi, T. Fujita, S. Hiroi, Pharmacological and clinical profile of alogliptin benzoate (NESINA®). Nihon Yakurigaku Zasshi 137(1), 43–50 (2011)
L.J. Scott, Alogliptin: a review of its use in the management of type 2 diabetes mellitus. Drugs 70(15), 2051–2072 (2010)
R.A. DeFronzo, P.R. Fleck, C.A. Wilson, Q. Mekkim, Efficacy and safety of the dipeptidyl peptidase-4 inhibitor alogliptin in patients with type 2 diabetes and inadequate glycemic control. Diabetes Care 31(12), 2315–2317 (2008)
R. Andukuri, A. Drincic, M. Rendell, Alogliptin: a new addition to the class of DPP-4 inhibitors. Diabetes Metab. Syndr. Obes. 2, 117–126 (2009)
D.M. Nathan, J.B. Buse, M.B. Davidson, E. Ferrannini, R.R. Holman, R. Sherwin, B. Zinman, American Diabetes Association; European Association for Study of Diabetes, Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 32(1), 193–203 (2009)
D.M. Nathan, J.B. Buse, M.B. Davidson, E. Ferrannini, R.R. Holman, R. Sherwin, B. Zinman, American Diabetes Association; European Association for Study of Diabetes, Medical management of hyperglycaemia in type 2 diabetes mellitus: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 52(1), 17–30 (2009)
M. Tominaga, Diagnostic criteria for diabetes mellitus. Rinsho Byori 47(10), 901–908 (1999)
K.F. Schulz, D.G. Altman, D. Moher, CONSORT Group, CONSORT 2010 statement: updated guidelines for reporting parallel group randomized trials. Ann. Intern. Med. 152(11), 726–732 (2010)
K. Cwiertka, A note on the making of culinary tradition—an example of modern Japan. Appetite 30(2), 117–128 (1998)
K. Shiwaku, M. Hashimoto, A. Nogi, K. Kitajima, M. Yamasaki, Traditional Japanese dietary basics: a solution for modern health issues? Lancet 363(9422), 1737–1738 (2004)
Y. Ma, B.C. Olendzki, P.A. Merriam, D.E. Chiriboga, A.L. Culver, W. Li, J.R. Hébert, I.S. Ockene, J.A. Griffith, S.L. Pagoto, A randomized clinical trial comparing low-glycemic index versus ADA dietary education among individuals with type 2 diabetes. Nutrition 24(1), 45–56 (2008)
D.R. Matthews, J.P. Hosker, A.S. Rudenski, B.A. Naylor, D.F. Treacher, R.C. Turner, Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7), 412–419 (1985)
J.A. Davidson, Advances in therapy for type 2 diabetes: GLP-1 receptor agonists and DPP-4 inhibitors. Cleve. Clin. J. Med. 76(Suppl 5), S28–S38 (2009)
J. White, Efficacy and safety of incretin based therapies: clinical trial data. J. Am. Pharm. Assoc. 49(Suppl 1), S30–S40 (2009)
F. Giorgino, A. Leonardini, A. Natalicchio, L. Laviola, Multifactorial intervention in type 2 diabetes: the promise of incretin-based therapies. J. Endocrinol. Invest. 34(1), 69–77 (2011)
Z. Shah, T. Kampfrath, J.A. Deiuliis, J Zhong, C. Pineda, Z. Ying, X. Xu, B. Lu, S. Moffatt-Bruce, R. Durairaj, Q. Sun, G. Mihai, A. Maiseyeu, S. Rajagopalan, Long-term dipeptidyl-peptidase 4 inhibition reduces atherosclerosis and inflammation via effects on monocyte recruitment and chemotaxis. Circulation (2011). doi:10.1161/CIRCULATIONAHA.111.041418
Acknowledgments
The author thanks Drs. Jan Wajs, Gaku Miyakawa, Susumu Touchika, Sachiko Yokoyama, and Hiroshi Kawashima for discussions and Naoki Takeda for the advice of statistical analysis.
Conflict of interest
The authors have nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Clinical Trail Registration: UMIN 000006860.
Rights and permissions
About this article
Cite this article
Kutoh, E., Ukai, Y. Alogliptin as an initial therapy in patients with newly diagnosed, drug naïve type 2 diabetes: a randomized, control trial. Endocrine 41, 435–441 (2012). https://doi.org/10.1007/s12020-012-9596-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-012-9596-0