Abstract
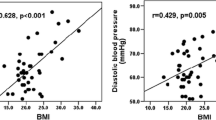
Classical congenital adrenal hyperplasia (CAH) is characterized by the defects in cortisol and aldosterone secretion, and accompanied with adrenal hyperandrogenism. It is likely that the impaired adrenocortical function and intermittent treatment-related hypercortisolism may predispose patients to the development of metabolic syndrome in adulthood. Our aim was to assess the impact of hyperandrogenism on metabolic profiles in CAH women without glucocorticosteroid treatment. We evaluated the clinical characteristics and metabolic profiles in 30 untreated Chinese female adults with simple virilizing congenital adrenal hyperplasia (SV–CAH). Mutation analysis was performed by sequencing the entire 21-hydroxylase gene (CYP21A2). As compared with the controls, CAH patients had higher BMI (BMI, 21.5 ± 2.1 vs. 20.0 ± 1.8 kg/m2, P < 0.05), higher 2 h post-load plasma glucose levels (6. 35 ± 1.74 vs. 5. 35 ± 1.17 mmol/l, P < 0.05), higher serum triglycerides (TG) (1.12 ± 0.64 vs. 0.63 ± 0.15 mmol/l, P < 0.01), and lower high-density lipoprotein cholesterol (HDL-c) (1.30 ± 0.39 vs. 1.67 ± 0.29 mmol/l, P < 0.01). Moreover, CAH patients had higher fasting insulin and homeostasis model assessment of insulin resistance (HOMA-IR) (1.81 ± 0.99 vs. 1.24 ± 0.50, P < 0.05), while ΔIns30/ΔGlu30 showed no statistically significant difference in two groups. In addition, a marked reduction of serum adiponectin levels were observed in CAH patients (7.0 ± 3.3 vs. 13.2 ± 4.8 μg/ml, P < 0.001), however, serum CRP levels were not different between patients and the controls. Further regression analysis showed that higher serum testosterone concentrations were associated with metabolic disorder indexes and reduction of serum adiponectin. Our study demonstrates that untreated CAH patients are prone to have metabolic disorders in association with elevated serum testosterone levels and reduced insulin insensitivity.


Similar content being viewed by others
References
P.C. White, M.I. New, Genetic basis of endocrine disease 2: congenital adrenal hyperplasia due to 21-hydroxylase deficiency. J. Clin. Endocrinol. Metab. 74(1), 6–11 (1992)
D.P. Merke, S.R. Bornstein, Congenital adrenal hyperplasia. Lancet 365(9477), 2125–2136 (2005)
E. Diamanti-Kandarakis, C. Christakou, H. Kandarakis, Polycystic ovarian syndrome: the commonest cause of hyperandrogenemia in women as a risk factor for metabolic syndrome. Minerva Endocrinol. 32(1), 35–47 (2007)
A. Corbould, Chronic testosterone treatment induces selective insulin resistance in subcutaneous adipocytes of women. J. Endocrinol. 192(3), 585–594 (2007)
P.W. Speiser et al., Insulin insensitivity in adrenal hyperplasia due to nonclassical steroid 21-hydroxylase deficiency. J. Clin. Endocrinol. Metab. 75(6), 1421–1424 (1992)
F. Saygili, A. Oge, C. Yilmaz, Hyperinsulinemia and insulin insensitivity in women with nonclassical congenital adrenal hyperplasia due to 21-hydroxylase deficiency: the relationship between serum leptin levels and chronic hyperinsulinemia. Horm. Res. 63(6), 270–274 (2005)
R.E. Cornean, P.C. Hindmarsh, C.G. Brook, Obesity in 21-hydroxylase deficient patients. Arch. Dis. Child. 78(3), 261–263 (1998)
L. Poretsky et al., The insulin-related ovarian regulatory system in health and disease. Endocr. Rev. 20(4), 535–582 (1999)
F.C. Wu, A. von Eckardstein, Androgens and coronary artery disease. Endocr. Rev. 24(2), 183–217 (2003)
S.H. Golden et al., Glucose and insulin components of the metabolic syndrome are associated with hyperandrogenism in postmenopausal women: the atherosclerosis risk in communities study. Am. J. Epidemiol. 160(6), 540–548 (2004)
D. Simon et al., Association between plasma total testosterone and cardiovascular risk factors in healthy adult men: The Telecom Study. J. Clin. Endocrinol. Metab. 82(2), 682–685 (1997)
M.N. Dieudonne et al., Androgen receptors in human preadipocytes and adipocytes: regional specificities and regulation by sex steroids. Am. J. Physiol. 274(6 Pt 1), C1645–C1652 (1998)
I. Janssen et al., Testosterone and visceral fat in midlife women: the Study of Women’s Health Across the Nation (SWAN) fat patterning study. Obesity 18(3), 604–610 (2010)
D.J. Evans, J.H. Barth, C.W. Burke, Body fat topography in women with androgen excess. Int. J. Obes. 12(2), 157–162 (1988)
J.C. Seidell et al., Androgenicity in relation to body fat distribution and metabolism in 38-year-old women—the European Fat Distribution Study. J. Clin. Epidemiol. 43(1), 21–34 (1990)
R. Pasquali, Obesity and androgens: facts and perspectives. Fertil. Steril. 85(5), 1319–1340 (2006)
J.S. Mayes, G.H. Watson, Direct effects of sex steroid hormones on adipose tissues and obesity. Obes. Rev. 5(4), 197–216 (2004)
J.A. Marcondes et al., Metabolic syndrome in women with polycystic ovary syndrome: prevalence, characteristics and predictors. Arq. Bras. Endocrinol. Metabol. 51(6), 972–979 (2007)
J.C. Marshall, Obesity in adolescent girls: is excess androgen the real bad actor? J. Clin. Endocrinol. Metab. 91(2), 393–395 (2006)
F.J. Paula et al., Androgen-related effects on peripheral glucose metabolism in women with congenital adrenal hyperplasia. Horm. Metab. Res. 26(11), 552–556 (1994)
E. Charmandari et al., Children with classic congenital adrenal hyperplasia have elevated serum leptin concentrations and insulin resistance: potential clinical implications. J. Clin. Endocrinol. Metab. 87(5), 2114–2120 (2002)
L. Green-Golan et al., Patients with classic congenital adrenal hyperplasia have decreased epinephrine reserve and defective glycemic control during prolonged moderate-intensity exercise. J. Clin. Endocrinol. Metab. 92(8), 3019–3024 (2007)
M. Berra et al., Testosterone decreases adiponectin levels in female to male transsexuals. Asian J. Androl. 8(6), 725–729 (2006)
S.T. Page et al., Testosterone administration suppresses adiponectin levels in men. J. Androl. 26(1), 85–92 (2005)
H. Nishizawa et al., Androgens decrease plasma adiponectin, an insulin-sensitizing adipocyte-derived protein. Diabetes 51(9), 2734–2741 (2002)
H.J. Zhang et al., Variations in the promoter of CYP21A2 gene identified in a Chinese patient with simple virilizing form of 21-hydroxylase deficiency. Clin. Endocrinol. 70(2), 201–207 (2009)
D. Ferriman, J.D. Gallwey, Clinical assessment of body hair growth in women. J. Clin. Endocrinol. Metab. 21, 1440–1447 (1961)
J.G. Fan et al., What are the risk factors and settings for non-alcoholic fatty liver disease in Asia-Pacific? J. Gastroenterol. Hepatol. 22(6), 794–800 (2007)
Acknowledgments
We are grateful to the consideration and cooperation of all the participants of the study. This study has been supported with the grants from Natural Science Foundation of China (No. 30725037 and 30973912) and Shanghai Committee of Science and Technology (No. 09DJ1400402 and 09XD1403400).
Author information
Authors and Affiliations
Corresponding author
Additional information
Hui-Jie Zhang and Jun Yang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, HJ., Yang, J., Zhang, MN. et al. Metabolic disorders in newly diagnosed young adult female patients with simple virilizing 21-hydroxylase deficiency. Endocr 38, 260–265 (2010). https://doi.org/10.1007/s12020-010-9382-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-010-9382-9