Abstract
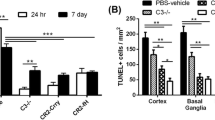
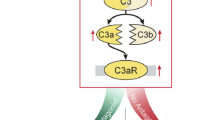
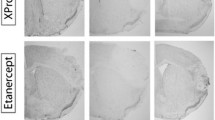
Acute ischemic stroke causes a high rate of deaths and permanent neurological deficits in survivors. Current interventional treatment, in the form of enzymatic thrombolysis, benefits only a small percentage of patients. Brain ischemia triggers mobilization of innate immunity, specifically the complement system and Toll-like receptors (TLRs), ultimately leading to an exaggerated inflammatory response. Here we demonstrate that intravenous immunoglobulin (IVIG), a scavenger of potentially harmful complement fragments, and C1-esterase inhibitor (C1-INH), an inhibitor of complement activation, exert a beneficial effect on the outcome of experimental brain ischemia (I) and reperfusion (R) injury induced by transient occlusion of middle cerebral artery in mice. Both IVIG and C1-INH significantly and in a dose–responsive manner reduced brain infarction size, neurological deficit and mortality when administered to male mice 30 min before ischemia or up to 6 h after the onset of reperfusion. When combined, suboptimal doses of IVIG and C1-INH potentiated each other’s neuroprotective therapeutic effects. Complement C3 and TLR2 signals were colocalized and significantly greater in brain cells adjacent to infracted brain lesions when compared to the corresponding regions of the contralateral hemisphere and to control (sham) mice. Treatment with IVIG and C1-INH effectively reduced deposition of C3b and downregulated excessive TLR2 and p-JNK1 expression at the site of I/R injury. Taken together, these results provide a rationale for potential use of IVIG and C1-INH, alone or in combination with ischemic stroke and other neurological conditions that involve inappropriately activated components of the innate immune system.






Similar content being viewed by others
References
Adams, H. P. (2003). Stroke: A vascular pathology with inadequate management. Journal of Hypertension. Supplement, 21, S3–S7.
Adeoye, O., Hornung, R., Khatri, P., & Kleindorfer, D. (2011). Recombinant tissue-type plasminogen activator use for ischemic stroke in the United States: A doubling of treatment rates over the course of 5 years. Stroke, 42, 1952–1955.
Akita, N., Nakase, H., Kaido, T., Kanemoto, Y., & Sakaki, T. (2003). Protective effect of C1 esterase inhibitor on reperfusion injury in the rat middle cerebral artery occlusion model. Neurosurgery, 52, 395–400.
Akita, N., Nakase, H., Kanemoto, Y., Kaido, T., Nishioka, T., & Sakaki, T. (2001). The effect of C1 esterase inhibitor on ischemia: Reperfusion injury in the rat brain. No To Shinkei, 53, 641–644.
Arumugam, T. V., Tang, S. C., Lathia, J. D., Cheng, A., Mughal, M. R., Chigurupati, S., et al. (2007). Intravenous immunoglobulin (IVIG) protects the brain against experimental stroke by preventing complement-mediated neuronal cell death. Proceedings of the National Academy of Sciences of the United States of America, 104, 14104–14109.
Brea, D., Blanco, M., Ramos-Cabrer, P., Moldes, O., Arias, S., Pérez-Mato, M., et al. (2011). Toll-like receptors 2 and 4 in ischemic stroke: Outcome and therapeutic values. Journal of Cerebral Blood Flow and Metabolism, 31, 1424–1431.
Chamorro, A., Meisel, A., Planas, A. M., Urra, X., van de Beek, D., & Veltkamp, R. (2012). The immunology of acute stroke. Nature Reviews Neurology, 8, 401–410.
Cojocaru, I. M., Cojocaru, M., Tănăsescu, R., Burcin, C., Atanasiu, A. N., Petrescu, A. M., et al. (2008). Changes in plasma levels of complement in patients with acute ischemic stroke. Romanian Journal of Internal Medicine, 46, 77–80.
D’Ambrosio, A. L., Pinsky, D. J., & Connolly, E. S. (2001). The role of the complement cascade in ischemia/reperfusion injury: Implications for neuroprotection. Molecular Medicine, 7, 367–382.
Davis, A. E., 3rd, Mejia, P., & Lu, F. (2008). Biological activities of C1 inhibitor. Molecular Immunology, 45, 4057–4063.
De Simoni, M. G., Rossi, E., Storini, C., Pizzimenti, S., Echart, C., Bergamaschini, L., et al. (2004). The powerful neuroprotective action of C1-inhibitor on brain ischemia-reperfusion injury does not require C1q. American Journal of Pathology, 64, 1857–1863.
De Simoni, M. G., Storini, C., Barba, M., Catapano, L., Arabia, A. M., Rossi, E., et al. (2003). Neuroprotection by complement (C1) inhibitor in mouse transient brain ischemia. Journal of Cerebral Blood Flow and Metabolism, 23, 232–239.
Ganz, D., Donin, N., & Fishelson, Z. (2009). Involvement of the c-jun N-terminal kinases JNK1 and JNK2 in complement-mediated cell death. Molecular Immunology, 47, 310–317.
Gesuete, R., Storini, C., Fantin, A., Stravalaci, M., Zanier, E. R., Orsini, F., et al. (2009). Recombinant C1 inhibitor in brain ischemic injury. Annals of Neurology, 66, 332–342.
Hajishengallis, G., & Lambris, J. D. (2010). Crosstalk pathways between Toll-like receptors and the complement system. Trends in Immunology, 31, 154–163.
Lloyd-Jones, D., Adams, R. J., Brown, T. M., Carnethon, M., Dai, S., De Simone, G., et al. (2009). Heart disease and stroke statistics—2009 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation, 119, e21–e181.
Longa, E. Z., Weinstein, P. R., Carlson, S., & Cummins, R. (1989). Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke, 20, 84–91.
Okun, E., Griffioen, K. J., & Mattson, M. P. (2011). Toll-like receptor signaling in neural plasticity and disease. Trends in Neurosciences, 34, 269–281.
Okuno, S., Saito, A., Hayashi, T., & Chan, P. H. (2004). The c-Jun N-terminal protein kinase signaling pathway mediates Bax activation and subsequent neuronal apoptosis through interaction with Bim after transient focal cerebral ischemia. Journal of Neuroscience, 24, 7879–7887.
Osthoff, M., Katan, M., Fluri, F., Schuetz, P., Bingisser, R., Kappos, L., et al. (2011). Mannose-binding lectin deficiency is associated with smaller infarction size and favorable outcome in ischemic stroke patients. PLoS ONE, 6, e21338.
Széplaki, G., Szegedi, R., Hirschberg, K., Gombos, T., Varga, L., Karádi, I., et al. (2009). Strong complement activation after acute ischemic stroke is associated with unfavorable outcomes. Atherosclerosis, 204, 315–320.
Tang, S. C., Arumugam, T. V., Xu, X., Cheng, A., Mughal, M. R., Jo, D. G., et al. (2007). Pivotal role for neuronal Toll-like receptors in ischemic brain injury and functional deficits. Proceedings of the National Academy of Sciences of the United States of America, 104, 13798–13803.
Acknowledgements
The authors wish to thank Dr. Mohamed R. Mughal for animal management and Drs. Ruiquian Wan, Eitan Okun and Dong Liu for their technical assistance. Supported, in part, by the Investigator Initiated Research grant from CSL Behring to Biovisions, Inc, and by the Intramural Research Program of the National Institute on Aging.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.

12017_2017_8474_MOESM1_ESM.jpg
IVIG, C1-INH and their combination exert neuroprotection 6 hours after the onset of ischemia. Mice were treated with IVIG at 1 g/kg (N=11), C1-INH at 300 U/kg (N=8) or the combination (N=10) 6 hours after the beginning of ischemia; the infarction area (A) and neurological deficit score (B) were determined at 72 h post-ischemia; *P < 0.001. Data are mean ± SEM (JPEG 31 kb)

12017_2017_8474_MOESM2_ESM.jpg
IVIG, C1-INH and their combination protect old and female mice from I/R-triggered brain injury. (A) 13–14 month-old male mice were subjected to I/R injury (N=8) and I/R injury plus IVIG at 1 g/kg (N=8), I/R + C1-INH at 300 U/kg (N=9) and I/R plus the combination of suboptimal doses of both (N=8) 6 hours after ischemia; *P < 0.001, compared with I/R only; #P < 0.01, combined compared with IVIG and C1-INH. (B) Neurological deficit score evaluated in the same groups of mice as in (A) 72 h post-ischemia; *P < 0.01, compared with I/R only; **P < 0.001, combined compared with I/R only; #P < 0.01, combined compared with IVIG and C1-INH. (C) Infarction area in non-treated female mice (N=9), infused with IVIG at 1 g/kg (N = 8), C1-INH at 300 U/Kg (N=9) and the combination (N=8) 6 hours post-ischemia; *P < 0.05, compared with I/R only. (D) Neurological deficit score in the same mice as in (C) at 72 h post-ischemia; *P < 0.01, compared with I/R only. Data are mean ± SEM (JPEG 51 kb)
Rights and permissions
About this article
Cite this article
Chen, X., Arumugam, T.V., Cheng, YL. et al. Combination Therapy with Low-Dose IVIG and a C1-esterase Inhibitor Ameliorates Brain Damage and Functional Deficits in Experimental Ischemic Stroke. Neuromol Med 20, 63–72 (2018). https://doi.org/10.1007/s12017-017-8474-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-017-8474-6