Abstract
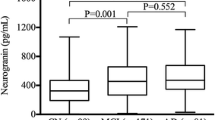
Stanniocalcin-1 (STC-1) is a nerve cell-enriched protein involved in intracellular calcium homeostasis regulation. Changes in calcium regulation are hypothesized to play a role in the pathophysiology of Alzheimer’s disease (AD). The expression of STC-1 increases in response to ischemic stroke, but whether it is altered in neurodegenerative disorder, particularly Alzheimer’s disease (AD), has not been investigated before. We measured STC-1 in cerebrospinal fluid (CSF) samples from a total of 163 individuals including AD, prodromal AD (pAD), mixed AD, stable mild cognitive impairment (sMCI), and diagnoses of other dementia than AD, as well as cognitively normal controls (CNC) enrolled at academic centers in France and Sweden. STC-1 concentration was reliably measureable in all CSF samples and was significantly increased in the initial exploratory cohort of neurochemically enriched AD patients versus AD biomarker-negative controls. In the second cohort, STC-1 was increased in AD versus pAD, and other dementia disorders, but the difference was not statistically significant. In the third cohort, there was no significant difference in STC-1 concentration between AD and CNC; however, STC-1 concentration was significantly decreased in patients with other dementia disorders compared with AD and CNC. Taken together, CSF STC-1 showed an increasing trend in AD, but the findings were not consistent across the three study cohorts. In contrast, CSF STC-1 concentrations were reduced in patients with dementia diagnoses other than AD, as compared with both AD patients and CNC. The findings from these studies suggest CSF STC-1 as a potential biomarker in differential diagnosis of dementias.


Similar content being viewed by others
References
Blennow, K., de Leon, M. J., & Zetterberg, H. (2006). Alzheimer’s disease. Lancet, 368(9533), 387–403.
Blennow, K., Hampel, H., Weiner, M., & Zetterberg, H. (2010). Cerebrospinal fluid and plasma biomarkers in Alzheimer disease. Nature Reviews Neurology, 6(3), 131–144.
Blennow, K., & Zetterberg, H. (2015). The past and the future of Alzheimer’s disease CSF biomarkers-a journey toward validated biochemical tests covering the whole spectrum of molecular events. Frontiers in Neuroscience, 9, 345.
Bostrom, F., Hansson, O., Gerhardsson, L., Lundh, T., Minthon, L., Stomrud, E., et al. (2009). CSF Mg and Ca as diagnostic markers for dementia with lewy bodies. Neurobiology of Aging, 30(8), 1265–1271.
Buerger, K., Ewers, M., Pirttila, T., Zinkowski, R., Alafuzoff, I., Teipel, S. J., et al. (2006). CSF phosphorylated tau protein correlates with neocortical neurofibrillary pathology in Alzheimer’s disease. Brain a Journal of Neurology, 129(Pt 11), 3035–3041.
Durukan Tolvanen, A., Westberg, J. A., Serlachius, M., Chang, A. C., Reddel, R. R., Andersson, L. C., et al. (2013). Stanniocalcin 1 is important for poststroke functionality, but dispensable for ischemic tolerance. Neuroscience, 229, 49–54.
Ferri, C. P., Prince, M., Brayne, C., Brodaty, H., Fratiglioni, L., Ganguli, M., et al. (2005). Global prevalence of dementia: A Delphi consensus study. Lancet, 366(9503), 2112–2117.
Hansson, O., Zetterberg, H., Buchhave, P., Londos, E., Blennow, K., & Minthon, L. (2006). Association between CSF biomarkers and incipient Alzheimer’s disease in patients with mild cognitive impairment: a follow-up study. Lancet Neurology, 5(3), 228–234.
Johansson, P., Mattsson, N., Hansson, O., Wallin, A., Johansson, J. O., Andreasson, U., et al. (2011). Cerebrospinal fluid biomarkers for Alzheimer’s disease: diagnostic performance in a homogeneous mono-center population. Journal of Alzheimer’s Disease JAD, 24(3), 537–546.
Khachaturian, Z. S. (1989). Calcium, membranes, aging, and Alzheimer’s disease. Introduction and overview. Annals of the New York Academy of Sciences, 568, 1–4.
Khachaturian, Z. S. (1994). Calcium hypothesis of Alzheimer’s disease and brain aging. Annals of the New York Academy of Sciences, 747, 1–11.
Lindsberg, P. J., Roine, R. O., Tatlisumak, T., Sairanen, T., & Kaste, M. (2000). The future of stroke treatment. Neurologic Clinics, 18(2), 495–510.
Masters, C. L., Simms, G., Weinman, N. A., Multhaup, G., McDonald, B. L., & Beyreuther, K. (1985). Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proceedings of the National Academy of Sciences of the United States of America, 82(12), 4245–4249.
Olsson, B., Lautner, R., Andreasson, U., Ohrfelt, A., Portelius, E., Bjerke, M., et al. (2016). CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: A systematic review and meta-analysis. Lancet Neurology, 15(7), 673–684.
Reitz, C., Tang, M. X., Schupf, N., Manly, J. J., Mayeux, R., & Luchsinger, J. A. (2010). Association of higher levels of high-density lipoprotein cholesterol in elderly individuals and lower risk of late-onset Alzheimer disease. Archives of Neurology, 67(12), 1491–1497.
Serlachius, M., Zhang, K. Z., & Andersson, L. C. (2004). Stanniocalcin in terminally differentiated mammalian cells. Peptides, 25(10), 1657–1662.
Sparks, D. L., Scheff, S. W., Hunsaker, J. C., 3rd, Liu, H., Landers, T., & Gross, D. R. (1994). Induction of Alzheimer-like beta-amyloid immunoreactivity in the brains of rabbits with dietary cholesterol. Experimental Neurology, 126(1), 88–94.
Subhash, M. N., Padmashree, T. S., Srinivas, K. N., Subbakrishna, D. K., & Shankar, S. K. (1991). Calcium and phosphorus levels in serum and CSF in dementia. Neurobiology of Aging, 12(4), 267–269.
Wagner, G. F., Hampong, M., Park, C. M., & Copp, D. H. (1986). Purification, characterization, and bioassay of teleocalcin, a glycoprotein from salmon corpuscles of Stannius. General and Comparative Endocrinology, 63(3), 481–491.
Yeung, B. H., Law, A. Y., & Wong, C. K. (2012). Evolution and roles of stanniocalcin. Molecular and Cellular Endocrinology, 349(2), 272–280.
Yoshiko, Y., & Aubin, J. E. (2004). Stanniocalcin 1 as a pleiotropic factor in mammals. Peptides, 25(10), 1663–1669.
Zhang, K., Lindsberg, P. J., Tatlisumak, T., Kaste, M., Olsen, H. S., & Andersson, L. C. (2000). Stanniocalcin: A molecular guard of neurons during cerebral ischemia. Proceedings of the National Academy of Sciences of the United States of America, 97(7), 3637–3642.
Zhang, K. Z., Westberg, J. A., Paetau, A., von Boguslawsky, K., Lindsberg, P., Erlander, M., et al. (1998). High expression of stanniocalcin in differentiated brain neurons. The American Journal of Pathology, 153(2), 439–445.
Acknowledgments
The study was supported by grants from the Swedish Research Council, the European Research Council, Frimurarestiftelsen, the Knut and Alice Wallenberg Foundation, Alzheimerfonden, Hjärnfonden, the Torsten Söderberg Foundation, and Swedish State Support for Clinical Research. HH is supported by the AXA Research Fund, the Fondation Université Pierre et Marie Curie and the “Fondation pour la Recherche sur Alzheimer,” Paris, France. The research leading to these results has received funding from the program “Investissements d’avenir” ANR-10-IAIHU-06 (Agence Nationale de la Recherche-10-IA Agence Institut Hospitalo-Universitaire-6).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
PS and JS report no conflict of interest. KB and HZ are co-founders of Brain Biomarker Solutions in Gothenburg AB, a GU Venture-based platform company at the University of Gothenburg. KB has served as a consultant or ad advisory Boards for Eli Lilly, IBL International, Roche Diagnostics, Fujirebio Europe, and Novartis.
Rights and permissions
About this article
Cite this article
Shahim, P., Blennow, K., Johansson, P. et al. Cerebrospinal Fluid Stanniocalcin-1 as a Biomarker for Alzheimer’s Disease and Other Neurodegenerative Disorders. Neuromol Med 19, 154–160 (2017). https://doi.org/10.1007/s12017-016-8439-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-016-8439-1