Abstract
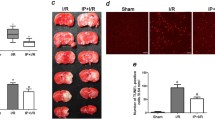
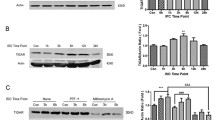
Inhalational anesthetic preconditioning can induce neuroprotective effects, and the notch signaling pathway plays an important role in neural progenitor cell differentiation and the inflammatory response after central nervous system injury. This study evaluated whether the neuroprotective effect of isoflurane preconditioning is mediated by the activation of the notch signaling pathway. Mice were divided into two groups consisting of those that did or did not receive preconditioning with isoflurane. The expression levels of notch-1, notch intracellular domain (NICD), and hairy and enhancer of split (HES-1) were measured in mice subjected to transient global cerebral ischemia–reperfusion injury. The notch signaling inhibitor DAPT and conditional notch-RBP-J knockout mice were used to investigate the mechanisms of isoflurane preconditioning-induced neuroprotection. Immunohistochemical staining, real-time polymerase chain reaction assays, and Western blotting were performed. Isoflurane preconditioning induced neuroprotection against global cerebral ischemia. Preconditioning up-regulated the expression of notch-1, HES-1, and NICD after ischemic–reperfusion. However, these molecules were down-regulated at 72 h after ischemic–reperfusion. The inhibition of notch signaling activity by DAPT significantly attenuated the isoflurane preconditioning-induced neuroprotection, and similar results were obtained using notch knockout mice. Our results demonstrate that the neuroprotective effects of isoflurane preconditioning are mediated by the pre-activation of the notch signaling pathway.






Similar content being viewed by others
References
Androutsellis-Theotokis, A., Leker, R. R., Soldner, F., Hoeppner, D. J., Ravin, R., Poser, S. W., et al. (2006). Notch signalling regulates stem cell numbers in vitro and in vivo. Nature, 442(7104), 823–826.
Artavanis-Tsakonas, S., Rand, M. D., & Lake, R. J. (1999). Notch signaling: Cell fate control and signal integration in development. [Research support, non-U.S. gov’t research support, U.S. gov’t, P.H.S. review]. Science, 284(5415), 770–776.
Arumugam, T. V., Chan, S. L., Jo, D. G., Yilmaz, G., Tang, S. C., Cheng, A., et al. (2006). Gamma secretase-mediated Notch signaling worsens brain damage and functional outcome in ischemic stroke. [Research support, N.I.H., intramural]. Nature Medicine, 12(6), 621–623.
Bein, B. (2011). Clinical application of the cardioprotective effects of volatile anaesthetics: PRO–get an extra benefit from a proven anaesthetic free of charge. [Research support, non-U.S. gov’t review]. European Journal of Anaesthesiology, 28(9), 620–622.
Bekker, A., Shah, R., Quartermain, D., Li, Y. S., & Blanck, T. (2006). Isoflurane preserves spatial working memory in adult mice after moderate hypoxia. Anesthesia and Analgesia, 102(4), 1134–1138.
Chen, J., Zacharek, A., Li, A., Cui, X., Roberts, C., Lu, M., et al. (2008). Atorvastatin promotes presenilin-1 expression and Notch1 activity and increases neural progenitor cell proliferation after stroke. Stroke, 39(1), 220–226.
Dovey, H. F., John, V., Anderson, J. P., Chen, L. Z., de Saint Andrieu, P., Fang, L. Y., et al. (2001). Functional gamma-secretase inhibitors reduce beta-amyloid peptide levels in brain. Journal of Neurochemistry, 76(1), 173–181.
Fujii, M., Hara, H., Meng, W., Vonsattel, J. P., Huang, Z., & Moskowitz, M. A. (1997). Strain-related differences in susceptibility to transient forebrain ischemia in SV-129 and C57black/6 mice. Stroke, 28(9), 1805–1810. (discussion 1811).
Gaiano, N., & Fishell, G. (2002). The role of notch in promoting glial and neural stem cell fates. Annual Review of Neuroscience, 25, 471–490.
Homi, H. M., Mixco, J. M., Sheng, H., Grocott, H. P., Pearlstein, R. D., & Warner, D. S. (2003). Severe hypotension is not essential for isoflurane neuroprotection against forebrain ischemia in mice. Anesthesiology, 99(5), 1145–1151.
Kitagawa, K., Matsumoto, M., Tsujimoto, Y., Ohtsuki, T., Kuwabara, K., Matsushita, K., et al. (1998a). Amelioration of hippocampal neuronal damage after global ischemia by neuronal overexpression of BCL-2 in transgenic mice. Stroke, 29(12), 2616–2621.
Kitagawa, K., Matsumoto, M., Yang, G., Mabuchi, T., Yagita, Y., Hori, M., et al. (1998b). Cerebral ischemia after bilateral carotid artery occlusion and intraluminal suture occlusion in mice: Evaluation of the patency of the posterior communicating artery. Journal of Cerebral Blood Flow and Metabolism, 18(5), 570–579.
Kitano, H., Kirsch, J. R., Hurn, P. D., & Murphy, S. J. (2007). Inhalational anesthetics as neuroprotectants or chemical preconditioning agents in ischemic brain. [Research support, N.I.H., extramural research support, non-U.S. gov’t review]. Journal of Cerebral Blood Flow and Metabolism, 27(6), 1108–1128.
Lanz, T. A., Himes, C. S., Pallante, G., Adams, L., Yamazaki, S., Amore, B., et al. (2003). The gamma-secretase inhibitor N-[N-(3, 5-difluorophenacetyl)-L-alanyl]-S-phenylglycine t-butyl ester reduces: A beta levels in vivo in plasma and cerebrospinal fluid in young (plaque-free) and aged (plaque-bearing) Tg2576 mice. Journal of Pharmacology and Experimental Therapeutics, 305(3), 864–871.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods, 25(4), 402–408.
Matchett, G. A., Allard, M. W., Martin, R. D., & Zhang, J. H. (2009). Neuroprotective effect of volatile anesthetic agents: Molecular mechanisms. Neurological Research, 31(2), 128–134.
Nagao, M., Sugimori, M., & Nakafuku, M. (2007). Cross talk between notch and growth factor/cytokine signaling pathways in neural stem cells. [Research support, non-U.S. gov’t]. Molecular and Cellular Biology, 27(11), 3982–3994.
Nakatomi, H., Kuriu, T., Okabe, S., Yamamoto, S., Hatano, O., Kawahara, N., et al. (2002). Regeneration of hippocampal pyramidal neurons after ischemic brain injury by recruitment of endogenous neural progenitors. Cell, 110(4), 429–441.
Ohtaki, H., Ylostalo, J. H., Foraker, J. E., Robinson, A. P., Reger, R. L., Shioda, S., et al. (2008). Stem/progenitor cells from bone marrow decrease neuronal death in global ischemia by modulation of inflammatory/immune responses. Proceedings of the National Academy of Science USA, 105(38), 14638–14643.
Oya, S., Yoshikawa, G., Takai, K., Tanaka, J. I., Higashiyama, S., Saito, N., et al. (2009). Attenuation of Notch signaling promotes the differentiation of neural progenitors into neurons in the hippocampal CA1 region after ischemic injury. [Research support, non-U.S. gov’t]. Neuroscience, 158(2), 683–692.
Presente, A., Andres, A., & Nye, J. S. (2001). Requirement of Notch in adulthood for neurological function and longevity. NeuroReport, 12(15), 3321–3325.
Redel, A., Stumpner, J., Tischer-Zeitz, T., Lange, M., Smul, T. M., Lotz, C., et al. (2009). Comparison of isoflurane-, sevoflurane-, and desflurane-induced pre- and postconditioning against myocardial infarction in mice in vivo. Experimental Biology and Medicine (Maywood), 234(10), 1186–1191.
Sang, H., Cao, L., Qiu, P., Xiong, L., Wang, R., & Yan, G. (2006). Isoflurane produces delayed preconditioning against spinal cord ischemic injury via release of free radicals in rabbits. Anesthesiology, 105(5), 953–960.
Schifilliti, D., Grasso, G., Conti, A., & Fodale, V. (2010). Anaesthetic-related neuroprotection: Intravenous or inhalational agents? [Research support, non-U.S. gov’t review]. CNS Drugs, 24(11), 893–907.
Sheng, H., Laskowitz, D. T., Mackensen, G. B., Kudo, M., Pearlstein, R. D., & Warner, D. S. (1999). Apolipoprotein E deficiency worsens outcome from global cerebral ischemia in the mouse. Stroke, 30(5), 1118–1124.
Wacker, B. K., Park, T. S., & Gidday, J. M. (2009). Hypoxic preconditioning-induced cerebral ischemic tolerance: Role of microvascular sphingosine kinase 2. Stroke, 40(10), 3342–3348.
Wang, L., Chopp, M., Zhang, R. L., Zhang, L., Letourneau, Y., Feng, Y. F., et al. (2009). The Notch pathway mediates expansion of a progenitor pool and neuronal differentiation in adult neural progenitor cells after stroke. Neuroscience, 158(4), 1356–1363.
Wang, L., Traystman, R. J., & Murphy, S. J. (2008). Inhalational anesthetics as preconditioning agents in ischemic brain. Current Opinion in Pharmacology, 8(1), 104–110.
Wei, Z., Chigurupati, S., Arumugam, T. V., Jo, D. G., Li, H., & Chan, S. L. (2011). Notch activation enhances the microglia-mediated inflammatory response associated with focal cerebral ischemia. [Research support, non-U.S. gov’t]. Stroke, 42(9), 2589–2594.
Xiong, L., Zheng, Y., Wu, M., Hou, L., Zhu, Z., Zhang, X., et al. (2003). Preconditioning with isoflurane produces dose-dependent neuroprotection via activation of adenosine triphosphate-regulated potassium channels after focal cerebral ischemia in rats. Anesthesia and Analgesia, 96(1), 233–237.
Yang, Y., Duan, W., Jin, Z., Bi, S., Yan, J., Jin, Y., et al. (2011). New role of Notch-mediated signaling pathway in myocardial ischemic preconditioning. [Research support, non-U.S. gov’t]. Medical Hypotheses, 76(3), 427–428.
Yang, Q., Yan, W., Li, X., Hou, L., Dong, H., Wang, Q., et al. (2012). Activation of canonical Notch signaling pathway is involved in the ischemic tolerance induced by sevoflurane preconditioning in mice. Anesthesiology. doi:10.1097/ALN.0b013e31826cb469.
Yilmaz, G., & Granger, D. N. (2010). Leukocyte recruitment and ischemic brain injury. [Research support, N.I.H., extramural research support, non-U.S. gov’t]. NeuroMolecular Medicine, 12(2), 193–204.
Yoon, K., & Gaiano, N. (2005). Notch signaling in the mammalian central nervous system: Insights from mouse mutants. Nature Neuroscience, 8(6), 709–715.
Yu, Z. C., Liu, W. C., Liu, D. H., & Fan, L. (2007). Effect of Notch ligand Delta-1 on the differentiation and maturation of erythroid progenitors in humans. Zhonghua Xue Ye Xue Za Zhi, 28(6), 401–403.
Zacharek, A., Chen, J., Cui, X., Yang, Y., & Chopp, M. (2009). Simvastatin increases notch signaling activity and promotes arteriogenesis after stroke. Stroke, 40(1), 254–260.
Zhang, H. P., Yuan, L. B., Zhao, R. N., Tong, L., Ma, R., Dong, H. L., et al. (2010). Isoflurane preconditioning induces neuroprotection by attenuating ubiquitin-conjugated protein aggregation in a mouse model of transient global cerebral ischemia. [Research support, non-U.S. gov’t]. Anesthesia and Analgesia, 111(2), 506–514.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Beijing, China; Grants 30772059/30972853 and 81371510 to Hailong Dong; Grant 81171051 to Yanyan Sun; Grant 81100901 to Binxiao Su) and the National Science Foundation for Distinguished Young Scholars (Beijing, China; Grant 30725039 to Lize Xiong).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Hao-peng Zhang, Yan-yan Sun, and Xiao-mei Chen have contributed equally to this work.
This work was conducted in the Department of Anesthesiology of Xijing Hospital, Fourth Military Medical University, Xi’an, Shaanxi, China.
Rights and permissions
About this article
Cite this article
Zhang, Hp., Sun, Yy., Chen, Xm. et al. The Neuroprotective Effects of Isoflurane Preconditioning in a Murine Transient Global Cerebral Ischemia–Reperfusion Model: The Role of the Notch Signaling Pathway. Neuromol Med 16, 191–204 (2014). https://doi.org/10.1007/s12017-013-8273-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-013-8273-7