Abstract
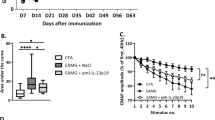
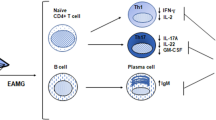
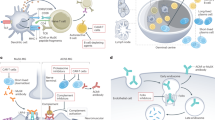
Abnormal toll-like receptor (TLR) activation and uncontrolled resolution of inflammation are suspected to play a key role in the development of autoimmune diseases. Acquired myasthenia gravis (MG) is an invalidating neuromuscular disease leading to muscle weaknesses. MG is mainly mediated by anti-acetylcholine receptor (AChR) autoantibodies, and thymic hyperplasia characterized by ectopic germinal centers is a common feature in MG. An abnormal expression of certain TLRs is observed in the thymus of MG patients associated with the overexpression of interferon (IFN)-β, the orchestrator of thymic changes in MG. Experimental models have been developed for numerous autoimmune diseases. These models are induced by animal immunization with a purified antigen solubilized in complete Freund’s adjuvant (CFA) containing heat-inactivated mycobacterium tuberculosis (MTB). Sensitization against the antigen is mainly due to the activation of TLR signaling pathways by the pathogen motifs displayed by MTB, and attempts have been made to substitute the use of CFA by TLR agonists. AChR emulsified in CFA is used to induce the classical experimental autoimmune MG model (EAMG). However, the TLR4 activator lipopolysaccharide (LPS) has proved to be efficient to replace MTB and induce a sensitization against purified AChR. Poly(I:C), the well-known TLR3 agonist, is also able by itself to induce MG symptoms in mice associated with early thymic changes as observed in human MG. In this review, we discuss the abnormal expression of TLRs in MG patients and we describe the use of TLR agonists to induce EAMG in comparison with other autoimmune experimental models.


Similar content being viewed by others
Abbreviations
- AChR:
-
Acetylcholine receptor
- CFA:
-
Complete Freund’s adjuvant
- EAMG:
-
Experimental autoimmune myasthenia gravis
- EBV:
-
Epstein-Barr virus
- EOMG:
-
Early-onset MG
- GC:
-
Germinal center
- IFA:
-
Incomplete Freund’s adjuvant
- IFN:
-
Interferon
- IL:
-
Interleukin
- IRF:
-
IFN-regulatory factor
- KO:
-
Knock-out
- MG:
-
Myasthenia gravis
- MyD88:
-
Myeloid differentiation primary response gene 88
- PBMCs:
-
Peripheral blood mononuclear cells
- Poly(I:C):
-
Polyinosinic-polycytidylic acid
- TEC:
-
Thymic epithelial cell
- TLR:
-
Toll-like receptor
- TRIF:
-
TIR domain-containing adapter-inducing interferon-β
References
Berrih-Aknin S, Le Panse R (2014) Myasthenia gravis: a comprehensive review of immune dysregulation and etiological mechanisms. J Autoimmun 52:90–100
Hoch W, McConville J, Helms S, Newsom-Davis J, Melms A et al (2001) Auto-antibodies to the receptor tyrosine kinase MuSK in patients with myasthenia gravis without acetylcholine receptor antibodies. Nat Med 7:365–368
Higuchi O, Hamuro J, Motomura M, Yamanashi Y (2011) Autoantibodies to low-density lipoprotein receptor-related protein 4 in myasthenia gravis. Ann Neurol 69:418–422
Zhang B, Shen C, Bealmear B, Ragheb S, Xiong WC et al (2014) Autoantibodies to agrin in myasthenia gravis patients. PLoS One 9, e91816
Nicolle MW (2002) Myasthenia gravis. Neurologist 8:2–21
Berrih-Aknin S, Morel E, Raimond F, Safar D, Gaud C et al (1987) The role of the thymus in myasthenia gravis: immunohistological and immunological studies in 115 cases. Ann N Y Acad Sci 505:50–70
Levinson AI, Wheatley LM (1996) The thymus and the pathogenesis of myasthenia gravis. Clin Immunol Immunopathol 78:1–5
Evoli A, Tonali PA, Padua L, Monaco ML, Scuderi F et al (2003) Clinical correlates with anti-MuSK antibodies in generalized seronegative myasthenia gravis. Brain 126:2304–2311
Ponseti JM, Caritg N, Gamez J, Lopez-Cano M, Vilallonga R et al (2009) A comparison of long-term post-thymectomy outcome of anti-AChR-positive, anti-AChR-negative and anti-MuSK-positive patients with non-thymomatous myasthenia gravis. Expert Opin Biol Ther 9:1–8
Anderson G, Takahama Y (2012) Thymic epithelial cells: working class heroes for T cell development and repertoire selection. Trends Immunol 33:256–263
Suniara RK, Jenkinson EJ, Owen JJ (2000) An essential role for thymic mesenchyme in early T cell development. J Exp Med 191:1051–1056
Derbinski J, Gabler J, Brors B, Tierling S, Jonnakuty S et al (2005) Promiscuous gene expression in thymic epithelial cells is regulated at multiple levels. J Exp Med 202:33–45
Berrih-Aknin S, Ruhlmann N, Bismuth J, Cizeron-Clairac G, Zelman E et al (2009) CCL21 overexpressed on lymphatic vessels drives thymic hyperplasia in myasthenia. Ann Neurol 66:521–531
Le Panse R, Bismuth J, Cizeron-Clairac G, Weiss JM, Cufi P et al (2010) Thymic remodeling associated with hyperplasia in myasthenia gravis. Autoimmunity 43:1–12
Weiss JM, Cufi P, Bismuth J, Eymard B, Fadel E et al (2013) SDF-1/CXCL12 recruits B cells and antigen-presenting cells to the thymus of autoimmune myasthenia gravis patients. Immunobiology 218:373–381
Weiss JM, Cufi P, Le Panse R, Berrih-Aknin S (2013) The thymus in autoimmune myasthenia gravis: paradigm for a tertiary lymphoid organ. Rev Neurol (Paris) 169:640–649
Ruddle NH (2014) Lymphatic vessels and tertiary lymphoid organs. J Clin Invest 124:953–959
Wakkach A, Guyon T, Bruand C, Tzartos S, Cohen-Kaminsky S et al (1996) Expression of acetylcholine receptor genes in human thymic epithelial cells: implications for myasthenia gravis. J Immunol 157:3752–3760
Safar D, Berrih-Aknin S, Morel E (1987) In vitro anti-acetylcholine receptor antibody synthesis by myasthenia gravis patient lymphocytes: correlations with thymic histology and thymic epithelial-cell interactions. J Clin Immunol 7:225–234
Leprince C, Cohen-Kaminsky S, Berrih-Aknin S, Vernet-Der Garabedian B, Treton D et al (1990) Thymic B cells from myasthenia gravis patients are activated B cells phenotypic and functional analysis. J Immunol 145:2115–2122
Melms A, Schalke BC, Kirchner T, Muller-Hermelink HK, Albert E et al (1988) Thymus in myasthenia gravis. Isolation of T-lymphocyte lines specific for the nicotinic acetylcholine receptor from thymuses of myasthenic patients. J Clin Invest 81:902–908
Kuks JB, Oosterhuis HJ, Limburg PC, The TH (1991) Anti-acetylcholine receptor antibodies decrease after thymectomy in patients with myasthenia gravis clinical correlations. J Autoimmun 4:197–211
Papatestas AE, Alpert LI, Osserman KE, Osserman RS, Kark AE (1971) Studies in myasthenia gravis: effects of thymectomy. Results on 185 patients with nonthymomatous and thymomatous myasthenia gravis, 1941–1969. Am J Med 50:465–474
Feferman T, Maiti PK, Berrih-Aknin S, Bismuth J, Bidault J et al (2005) Overexpression of IFN-induced protein 10 and its receptor CXCR3 in myasthenia gravis. J Immunol 174:5324–5331
Cordiglieri C, Marolda R, Franzi S, Cappelletti C, Giardina C et al (2014) Innate immunity in myasthenia gravis thymus: pathogenic effects of Toll-like receptor 4 signaling on autoimmunity. J Autoimmun 52:74–89
Legler DF, Loetscher M, Roos RS, Clark-Lewis I, Baggiolini M et al (1998) B cell-attracting chemokine 1, a human CXC chemokine expressed in lymphoid tissues, selectively attracts B lymphocytes via BLR1/CXCR5. J Exp Med 187:655–660
Barone F, Bombardieri M, Manzo A, Blades MC, Morgan PR et al (2005) Association of CXCL13 and CCL21 expression with the progressive organization of lymphoid-like structures in Sjogren’s syndrome. Arthritis Rheum 52:1773–1784
Méraouna A, Cizeron-Clairac G, Le Panse R, Bismuth J, Truffault F et al (2006) The chemokine CXCL13 is a key molecule in autoimmune myasthenia gravis. Blood 108:432–440
Le Panse R, Cizeron-Clairac G, Bismuth J, Berrih-Aknin S (2006) Microarrays reveal distinct gene signatures in the thymus of seropositive and seronegative myasthenia gravis patients and the role of CC chemokine ligand 21 in thymic hyperplasia. J Immunol 177:7868–7879
Shiao YM, Lee CC, Hsu YH, Huang SF, Lin CY et al (2010) Ectopic and high CXCL13 chemokine expression in myasthenia gravis with thymic lymphoid hyperplasia. J Neuroimmunol 221:101–106
Zhang M, Guo J, Li H, Zhou Y, Tian F et al (2013) Expression of immune molecules CD25 and CXCL13 correlated with clinical severity of myasthenia gravis. J Mol Neurosci 50:317–323
Stubgen JP (2009) Interferon alpha and neuromuscular disorders. J Neuroimmunol 207:3–17
Meager A, Wadhwa M, Dilger P, Bird C, Thorpe R et al (2003) Anti-cytokine autoantibodies in autoimmunity: preponderance of neutralizing autoantibodies against interferon-alpha, interferon-omega and interleukin-12 in patients with thymoma and/or myasthenia gravis. Clin Exp Immunol 132:128–136
Meloni A, Furcas M, Cetani F, Marcocci C, Falorni A et al (2008) Autoantibodies against type I interferons as an additional diagnostic criterion for autoimmune polyendocrine syndrome type I. J Clin Endocrinol Metab 93:4389–4397
Poea-Guyon S, Christadoss P, Le Panse R, Guyon T, De Baets M et al (2005) Effects of cytokines on acetylcholine receptor expression: implications for myasthenia gravis. J Immunol 174:5941–5949
Le Panse R, Cizeron-Clairac G, Cuvelier M, Truffault F, Bismuth J et al (2008) Regulatory and pathogenic mechanisms in human autoimmune myasthenia gravis. Ann N Y Acad Sci 1132:135–142
Golding A, Rosen A, Petri M, Akhter E, Andrade F (2010) Interferon-alpha regulates the dynamic balance between human activated regulatory and effector T cells: implications for antiviral and autoimmune responses. Immunology 131:107–117
Cufi P, Dragin N, Weiss JM, Martinez-Martinez P, De Baets MH et al (2013) Implication of double-stranded RNA signaling in the etiology of autoimmune myasthenia gravis. Ann Neurol 73:281–293
Cufi P, Dragin N, Ruhlmann N, Weiss JM, Fadel E et al (2014) Central role of interferon-beta in thymic events leading to myasthenia gravis. J Autoimmun 52:44–52
Weiss JM, Robinet M, Aricha R, Cufi P, Villeret B et al (2016) Novel CXCL13 transgenic mouse: inflammation drives pathogenic effect of CXCL13 in experimental myasthenia gravis., Oncotarget
Cufi P, Soussan P, Truffault F, Fetouchi R, Robinet M et al (2014) Thymoma-associated myasthenia gravis: on the search for a pathogen signature. J Autoimmun 52:29–35
Okada H, Kuhn C, Feillet H, Bach JF (2010) The “hygiene hypothesis” for autoimmune and allergic diseases: an update. Clin Exp Immunol 160:1–9
Munz C, Lunemann JD, Getts MT, Miller SD (2009) Antiviral immune responses: triggers of or triggered by autoimmunity? Nat Rev Immunol 9:246–258
Savino W (2006) The thymus is a common target organ in infectious diseases. PLoS Pathog 2, e62
Cavalcante P, Barberis M, Cannone M, Baggi F, Antozzi C et al (2010) Detection of poliovirus-infected macrophages in thymus of patients with myasthenia gravis. Neurology 74:1118–1126
Cavalcante P, Serafini B, Rosicarelli B, Maggi L, Barberis M et al (2010) Epstein-Barr virus persistence and reactivation in myasthenia gravis thymus. Ann Neurol 67:726–738
Niller HH, Wolf H, Ay E, Minarovits J (2011) Epigenetic dysregulation of Epstein-Barr virus latency and development of autoimmune disease. Adv Exp Med Biol 711:82–102
Ning S (2011) Innate immune modulation in EBV infection. Herpesviridae 2:1
Kawai T, Akira S (2010) The role of pattern-recognition receptors in innate immunity: update on toll-like receptors. Nat Immunol 11:373–384
Kawasaki T, Kawai T (2014) Toll-like receptor signaling pathways. Front Immunol 5:461
Mohammad Hosseini A, Majidi J, Baradaran B, Yousefi M (2015) Toll-like receptors in the pathogenesis of autoimmune diseases. Adv Pharm Bull 5:605–614
Chen JQ, Szodoray P, Zeher M (2016) Toll-like receptor pathways in autoimmune diseases. Clin Rev Allergy Immunol 50:1–17
Choi YJ, Im E, Chung HK, Pothoulakis C, Rhee SH (2010) TRIF mediates toll-like receptor 5-induced signaling in intestinal epithelial cells. J Biol Chem 285:37570–37578
Volpi C, Fallarino F, Pallotta MT, Bianchi R, Vacca C et al (2013) High doses of CpG oligodeoxynucleotides stimulate a tolerogenic TLR9-TRIF pathway. Nat Commun 4:1852
Nilsen NJ, Vladimer GI, Stenvik J, Orning MP, Zeid-Kilani MV et al (2015) A role for the adaptor proteins TRAM and TRIF in toll-like receptor 2 signaling. J Biol Chem 290:3209–3222
Perkins DJ, Vogel SN (2015) Space and time: new considerations about the relationship between toll-like receptors (TLRs) and type I interferons (IFNs). Cytokine 74:171–174
Pohar J, Pirher N, Bencina M, Mancek-Keber M, Jerala R (2013) The role of UNC93B1 protein in surface localization of TLR3 receptor and in cell priming to nucleic acid agonists. J Biol Chem 288:442–454
Kanno A, Tanimura N, Ishizaki M, Ohko K, Motoi Y et al (2015) Targeting cell surface TLR7 for therapeutic intervention in autoimmune diseases. Nat Commun 6:6119
Itoh H, Tatematsu M, Watanabe A, Iwano K, Funami K et al (2011) UNC93B1 physically associates with human TLR8 and regulates TLR8-mediated signaling. PLoS One 6, e28500
Guerrier T, Pochard P, Lahiri A, Youinou P, Pers JO et al (2014) TLR9 expressed on plasma membrane acts as a negative regulator of human B cell response. J Autoimmun 51:23–29
Hurst J, von Landenberg P (2008) Toll-like receptors and autoimmunity. Autoimmun Rev 7:204–208
Duthie MS, Windish HP, Fox CB, Reed SG (2011) Use of defined TLR ligands as adjuvants within human vaccines. Immunol Rev 239:178–196
Jin B, Sun T, Yu XH, Yang YX, Yeo AE (2012) The effects of TLR activation on T-cell development and differentiation. Clin Dev Immunol 2012:836485
Crampton SP, Voynova E, Bolland S (2010) Innate pathways to B-cell activation and tolerance. Ann N Y Acad Sci 1183:58–68
Green NM, Moody KS, Debatis M, Marshak-Rothstein A (2012) Activation of autoreactive B cells by endogenous TLR7 and TLR3 RNA ligands., J Biol Chem
Meyer-Bahlburg A, Rawlings DJ (2008) B cell autonomous TLR signaling and autoimmunity. Autoimmun Rev 7:313–316
Green NM, Marshak-Rothstein A (2011) Toll-like receptor driven B cell activation in the induction of systemic autoimmunity. Semin Immunol 23:106–112
Wang YZ, Yan M, Tian FF, Zhang JM, Liu Q et al (2013) Possible involvement of toll-like receptors in the pathogenesis of myasthenia gravis. Inflammation 36:121–130
Lu J, Yan M, Wang Y, Zhang J, Yang H et al (2013) Altered expression of miR-146a in myasthenia gravis. Neurosci Lett 555:85–90
Zarember KA, Godowski PJ (2002) Tissue expression of human toll-like receptors and differential regulation of toll-like receptor mRNAs in leukocytes in response to microbes, their products, and cytokines. J Immunol 168:554–561
Bernasconi P, Barberis M, Baggi F, Passerini L, Cannone M et al (2005) Increased toll-like receptor 4 expression in thymus of myasthenic patients with thymitis and thymic involution. Am J Pathol 167:129–139
Cavalcante P, Galbardi B, Franzi S, Marcuzzo S, Barzago C et al (2016) Increased expression of toll-like receptors 7 and 9 in myasthenia gravis thymus characterized by active Epstein-Barr virus infection. Immunobiology 221:516–527
Quan TE, Roman RM, Rudenga BJ, Holers VM, Craft JE (2010) Epstein-Barr virus promotes interferon-alpha production by plasmacytoid dendritic cells. Arthritis Rheum 62:1693–1701
Iwakiri D, Zhou L, Samanta M, Matsumoto M, Ebihara T et al (2009) Epstein-Barr virus (EBV)-encoded small RNA is released from EBV-infected cells and activates signaling from toll-like receptor 3. J Exp Med 206:2091–2099
Gaudreault E, Fiola S, Olivier M, Gosselin J (2007) Epstein-Barr virus induces MCP-1 secretion by human monocytes via TLR2. J Virol 81:8016–8024
Valente RM, Ehlers E, Xu D, Ahmad H, Steadman A et al (2012) Toll-like receptor 7 stimulates the expression of Epstein-Barr virus latent membrane protein 1. PLoS One 7, e43317
Fuchs S, Aricha R, Reuveni D, Souroujon MC (2014) Experimental autoimmune myasthenia gravis (EAMG): from immunochemical characterization to therapeutic approaches. J Autoimmun 54:51–59
Tuzun E, Berrih-Aknin S, Brenner T, Kusner LL, Le Panse R et al (2015) Guidelines for standard preclinical experiments in the mouse model of myasthenia gravis induced by acetylcholine receptor immunization., Exp Neurol
Wu B, Goluszko E, Huda R, Tuzun E, Christadoss P (2013) Experimental autoimmune myasthenia gravis in the mouse. Curr Protoc Immunol Chapter 15: Unit 15 18.
Losen M, Martinez-Martinez P, Molenaar PC, Lazaridis K, Tzartos S et al (2015) Standardization of the experimental autoimmune myasthenia gravis (EAMG) model by immunization of rats with Torpedo californica acetylcholine receptors—recommendations for methods and experimental designs. Exp Neurol 270:18–28
Billiau A, Matthys P (2001) Modes of action of Freund’s adjuvants in experimental models of autoimmune diseases. J Leukoc Biol 70:849–860
Milani M, Ostlie N, Wu H, Wang W, Conti-Fine BM (2006) CD4+ T and B cells cooperate in the immunoregulation of experimental autoimmune myasthenia gravis. J Neuroimmunol 179:152–162
Shibaki A, Katz SI (2002) Induction of skewed Th1/Th2 T-cell differentiation via subcutaneous immunization with Freund’s adjuvant. Exp Dermatol 11:126–134
Balasa B, Deng C, Lee J, Bradley LM, Dalton DK et al (1997) Interferon gamma (IFN-gamma) is necessary for the genesis of acetylcholine receptor-induced clinical experimental autoimmune myasthenia gravis in mice. J Exp Med 186:385–391
Zhang GX, Xiao BG, Bai XF, van der Meide PH, Orn A et al (1999) Mice with IFN-gamma receptor deficiency are less susceptible to experimental autoimmune myasthenia gravis. J Immunol 162:3775–3781
Schaffert H, Pelz A, Saxena A, Losen M, Meisel A et al (2015) IL-17-producing CD4(+) T cells contribute to the loss of B-cell tolerance in experimental autoimmune myasthenia gravis. Eur J Immunol 45:1339–1347
Gavin AL, Hoebe K, Duong B, Ota T, Martin C et al (2006) Adjuvant-enhanced antibody responses in the absence of toll-like receptor signaling. Science 314:1936–1938
Su SB, Silver PB, Grajewski RS, Agarwal RK, Tang J et al (2005) Essential role of the MyD88 pathway, but nonessential roles of TLRs 2, 4, and 9, in the adjuvant effect promoting Th1-mediated autoimmunity. J Immunol 175:6303–6310
Marty RR, Dirnhofer S, Mauermann N, Schweikert S, Akira S et al (2006) MyD88 signaling controls autoimmune myocarditis induction. Circulation 113:258–265
Sadanaga A, Nakashima H, Akahoshi M, Masutani K, Miyake K et al (2007) Protection against autoimmune nephritis in MyD88-deficient MRL/lpr mice. Arthritis Rheum 56:1618–1628
Fang J, Fang D, Silver PB, Wen F, Li B et al (2010) The role of TLR2, TRL3, TRL4, and TRL9 signaling in the pathogenesis of autoimmune disease in a retinal autoimmunity model. Invest Ophthalmol Vis Sci 51:3092–3099
Scadding GK, Calder L, Vincent A, Prior C, Wray D et al (1986) Anti-acetylcholine receptor antibodies induced in mice by syngeneic receptor without adjuvants. Immunology 58:151–155
Jermy A, Beeson D, Vincent A (1993) Pathogenic autoimmunity to affinity-purified mouse acetylcholine receptor induced without adjuvant in BALB/c mice. Eur J Immunol 23:973–976
Kool M, Soullie T, van Nimwegen M, Willart MA, Muskens F et al (2008) Alum adjuvant boosts adaptive immunity by inducing uric acid and activating inflammatory dendritic cells. J Exp Med 205:869–882
Oshima M, Maruta T, Ohtani M, Deitiker PR, Mosier D et al (2006) Vaccination with a MHC class II peptide in alum and inactive pertussis strongly ameliorates clinical MG in C57BL/6 mice. J Neuroimmunol 171:8–16
Bennett B, Check IJ, Olsen MR, Hunter RL (1992) A comparison of commercially available adjuvants for use in research. J Immunol Methods 153:31–40
Shenoy M, Christadoss P (1993) Induction of experimental autoimmune myasthenia gravis with acetylcholine receptors using a nonionic block copolymer as adjuvant. Immunol Investig 22:267–282
Lu YC, Yeh WC, Ohashi PS (2008) LPS/TLR4 signal transduction pathway. Cytokine 42:145–151
Demon D, Vande Walle L, Lamkanfi M (2014) Sensing the enemy within: how macrophages detect intracellular Gram-negative bacteria. Trends Biochem Sci 39:574–576
Liu Y, Yin H, Zhao M, Lu Q (2014) TLR2 and TLR4 in autoimmune diseases: a comprehensive review. Clin Rev Allergy Immunol 47:136–147
Allman W, Qi H, Saini SS, Li J, Tuzun E et al (2012) CD4 costimulation is not required in a novel LPS-enhanced model of myasthenia gravis. J Neuroimmunol 249:1–7
Rose NR (2008) The adjuvant effect in infection and autoimmunity. Clin Rev Allergy Immunol 34:279–282
Damotte D, Goulvestre C, Charreire J, Carnaud C (2003) LPS and Freund’s adjuvant initiate different inflammatory circuits in experimental autoimmune thyroiditis. Eur Cytokine Netw 14:52–59
Deane JA, Bolland S (2006) Nucleic acid-sensing TLRs as modifiers of autoimmunity. J Immunol 177:6573–6578
Berman PW, Patrick J (1980) Experimental myasthenia gravis. A murine system. J Exp Med 151:204–223
Fujii Y, Monden Y, Hashimoto J, Nakahara K, Kawashima Y (1985) Acetylcholine receptor antibody-producing cells in thymus and lymph nodes in myasthenia gravis. Clin Immunol Immunopathol 34:141–146
Zare F, Bokarewa M, Nenonen N, Bergstrom T, Alexopoulou L et al (2004) Arthritogenic properties of double-stranded (viral) RNA. J Immunol 172:5656–5663
Okada C, Akbar SM, Horiike N, Onji M (2005) Early development of primary biliary cirrhosis in female C57BL/6 mice because of poly I:C administration. Liver Int 25:595–603
Asada M, Nishio A, Akamatsu T, Tanaka J, Saga K et al (2010) Analysis of humoral immune response in experimental autoimmune pancreatitis in mice. Pancreas 39:224–231
Patole PS, Grone HJ, Segerer S, Ciubar R, Belemezova E et al (2005) Viral double-stranded RNA aggravates lupus nephritis through toll-like receptor 3 on glomerular mesangial cells and antigen-presenting cells. J Am Soc Nephrol 16:1326–1338
Jorgensen TN, Thurman J, Izui S, Falta MT, Metzger TE et al (2006) Genetic susceptibility to polyI:C-induced IFNalpha/beta-dependent accelerated disease in lupus-prone mice. Genes Immun 7:555–567
Nandula SR, Scindia YM, Dey P, Bagavant H, Deshmukh US (2011) Activation of innate immunity accelerates sialoadenitis in a mouse model for Sjogren’s syndrome-like disease. Oral Dis 17:801–807
Moriyama H, Wen L, Abiru N, Liu E, Yu L et al (2002) Induction and acceleration of insulitis/diabetes in mice with a viral mimic (polyinosinic-polycytidylic acid) and an insulin self-peptide. Proc Natl Acad Sci U S A 99:5539–5544
Ren X, Zhou H, Li B, Su SB (2011) Toll-like receptor 3 ligand polyinosinic:polycytidylic acid enhances autoimmune disease in a retinal autoimmunity model. Int Immunopharmacol 11:769–773
Ambrosini YM, Yang GX, Zhang W, Tsuda M, Shu S et al (2011) The multi-hit hypothesis of primary biliary cirrhosis: polyinosinic-polycytidylic acid (poly I:C) and murine autoimmune cholangitis. Clin Exp Immunol 166:110–120
Touil T, Fitzgerald D, Zhang GX, Rostami A, Gran B (2006) Cutting Edge: TLR3 stimulation suppresses experimental autoimmune encephalomyelitis by inducing endogenous IFN-beta. J Immunol 177:7505–7509
Khorooshi R, Morch MT, Holm TH, Berg CT, Dieu RT et al (2015) Induction of endogenous type I interferon within the central nervous system plays a protective role in experimental autoimmune encephalomyelitis. Acta Neuropathol 130:107–118
Jankovic SM (2010) Injectable interferon beta-1b for the treatment of relapsing forms of multiple sclerosis. J Inflamm Res 3:25–31
Lennon VA, Lindstrom JM, Seybold ME (1975) Experimental autoimmune myasthenia: a model of myasthenia gravis in rats and guinea pigs. J Exp Med 141:1365–1375
Fuchs S, Nevo D, Tarrab-Hazdai R, Yaar I (1976) Strain differences in the autoimmune response of mice to acetylcholine receptors. Nature 263:329–330
Meinl E, Klinkert WE, Wekerle H (1991) The thymus in myasthenia gravis. Changes typical for the human disease are absent in experimental autoimmune myasthenia gravis of the Lewis rat. Am J Pathol 139:995–1008
Lennon VA, Lindstrom JM, Seybold ME (1976) Experimental autoimmune myasthenia gravis: cellular and humoral immune responses. Ann N Y Acad Sci 274:283–299
Fallarino F, Volpi C, Zelante T, Vacca C, Calvitti M et al (2009) IDO mediates TLR9-driven protection from experimental autoimmune diabetes. J Immunol 183:6303–6312
Gilboa-Geffen A, Wolf Y, Hanin G, Melamed-Book N, Pick M et al (2011) Activation of the alternative NFkappaB pathway improves disease symptoms in a model of Sjogren’s syndrome. PLoS One 6, e28727
Longhini AL, Santos MP, Pradella F, Moraes AS, Dionete AC et al (2014) In vivo administration of TLR9 agonist reduces the severity of experimental autoimmune encephalomyelitis. The role of plasmacytoid dendritic cells and B lymphocytes. CNS Neurosci Ther 20:787–790
Farhat K, Riekenberg S, Heine H, Debarry J, Lang R et al (2008) Heterodimerization of TLR2 with TLR1 or TLR6 expands the ligand spectrum but does not lead to differential signaling. J Leukoc Biol 83:692–701
van Bergenhenegouwen J, Plantinga TS, Joosten LA, Netea MG, Folkerts G et al (2013) TLR2 & Co: a critical analysis of the complex interactions between TLR2 and coreceptors. J Leukoc Biol 94:885–902
Nishiya T, Kajita E, Miwa S, Defranco AL (2005) TLR3 and TLR7 are targeted to the same intracellular compartments by distinct regulatory elements. J Biol Chem 280:37107–37117
Tatematsu M, Seya T, Matsumoto M (2014) Beyond dsRNA: toll-like receptor 3 signalling in RNA-induced immune responses. Biochem J 458:195–201
Liaunardy-Jopeace A, Gay NJ (2014) Molecular and cellular regulation of toll-like receptor-4 activity induced by lipopolysaccharide ligands. Front Immunol 5:473
Miao EA, Andersen-Nissen E, Warren SE, Aderem A (2007) TLR5 and Ipaf: dual sensors of bacterial flagellin in the innate immune system. Semin Immunopathol 29:275–288
Guiducci C, Gong M, Cepika AM, Xu Z, Tripodo C et al (2013) RNA recognition by human TLR8 can lead to autoimmune inflammation. J Exp Med 210:2903–2919
Huang X, Yang Y (2010) Targeting the TLR9-MyD88 pathway in the regulation of adaptive immune responses. Expert Opin Ther Targets 14:787–796
Hasan U, Chaffois C, Gaillard C, Saulnier V, Merck E et al (2005) Human TLR10 is a functional receptor, expressed by B cells and plasmacytoid dendritic cells, which activates gene transcription through MyD88. J Immunol 174:2942–2950
Lee SM, Kok KH, Jaume M, Cheung TK, Yip TF et al (2014) Toll-like receptor 10 is involved in induction of innate immune responses to influenza virus infection. Proc Natl Acad Sci U S A 111:3793–3798
Hatai H, Lepelley A, Zeng W, Hayden MS, Ghosh S (2016) Toll-like receptor 11 (TLR11) interacts with flagellin and profilin through disparate mechanisms. PLoS One 11, e0148987
Cho YG, Cho ML, Min SY, Kim HY (2007) Type II collagen autoimmunity in a mouse model of human rheumatoid arthritis. Autoimmun Rev 7:65–70
Libbey JE, Fujinami RS (2011) Experimental autoimmune encephalomyelitis as a testing paradigm for adjuvants and vaccines. Vaccine 29:3356–3362
Allenbach Y, Solly S, Gregoire S, Dubourg O, Salomon B et al (2009) Role of regulatory T cells in a new mouse model of experimental autoimmune myositis. Am J Pathol 174:989–998
Lohse AW, Manns M, Dienes HP, Buschenfelde KH M z, Cohen IR (1990) Experimental autoimmune hepatitis: disease induction, time course and T-cell reactivity. Hepatology 11:24–30
Jones DE, Palmer JM, Kirby JA, De Cruz DJ, McCaughan GW et al (2000) Experimental autoimmune cholangitis: a mouse model of immune-mediated cholangiopathy. Liver 20:351–356
Little MA, Smyth L, Salama AD, Mukherjee S, Smith J et al (2009) Experimental autoimmune vasculitis: an animal model of anti-neutrophil cytoplasmic autoantibody-associated systemic vasculitis. Am J Pathol 174:1212–1220
Tincani A, Gilburd B, Abu-Shakra M, Blank M, Allegri F et al (2002) Immunization of naive BALB/c mice with human beta2-glycoprotein I breaks tolerance to the murine molecule. Arthritis Rheum 46:1399–1404
Acknowledgments
We thank Profs Remi Nottin and Elie Fadel for the thymic samples and Vincent de Montpreville for the histological analyses. We thank the “Agence Nationale de la Recherche” (ANR-06-MRAR-001-01) from the European Community (MYASTAID/LSHM-CT-2006-037833 and FIGHT-MG/HEALTH-2009-242-210) and from the “Association Française contre les Myopathies” (AFM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors have declared that no conflict of interest exists.
Rights and permissions
About this article
Cite this article
Robinet, M., Maillard, S., Cron, M.A. et al. Review on Toll-Like Receptor Activation in Myasthenia Gravis: Application to the Development of New Experimental Models. Clinic Rev Allerg Immunol 52, 133–147 (2017). https://doi.org/10.1007/s12016-016-8549-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-016-8549-4