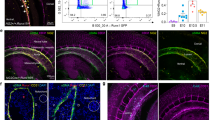
Abstract
The aorta-gonad-mesonephros (AGM) region contains intra-aortic clusters (IACs) thought to have acquired hematopoietic stem cell (HSC) potential in vertebrate embryos. To assess extrinsic regulation of IACs in the AGM region, we employed mouse embryos harboring a Sall1-GFP reporter gene, which allows identification of mesonephros cells based on GFP expression. Analysis of AGM region tissue sections confirmed mesonephros GFP expression. Mesonephric cells sorted at E10.5 expressed mRNA encoding Csf1, a hematopoietic cytokine, and corresponding protein, based on real-time PCR and immunocytochemistry, respectively. Further analysis indicated that some IACs express the CSF1 receptor, CSF1R. Expression of Cebpa and Irf8 mRNAs was higher in CSF1R-positive IACs, whereas that of Cebpε and Gfi1 mRNAs was lower relative to CSF1R-negative IACs, suggesting that CSF1/CSF1R signaling functions in IAC myeloid differentiation by modulating expression of these transcription factors. Colony formation assays using CSF1R-positive IACs revealed increased numbers of myeloid colonies in the presence of CSF1. Analysis using an intra-cellular signaling array indicated the greatest fold increase of Cleaved Caspase-3 in AGM cells in the presence of CSF1. Immunohistochemistry revealed that Cleaved Caspase-3 is primarily expressed in IACs in the AGM region, and incubation of IACs with CSF1 up-regulated Cleaved Caspase-3. Overall, our findings suggest that CSF1 secreted from mesonephros accelerates IAC myeloid differentiation in the AGM region, possibly via Caspase-3 cleavage.








Similar content being viewed by others
References
Cumano, A., Dieterlen-Lievre, F., & Godin, I. (1996). Lymphoid potential, probed before circulation in mouse, is restricted to caudal intraembryonic splanchnopleura. Cell, 86(6), 907–916.
Ferkowicz, M. J., & Yoder, M. C. (2005). Blood island formation: longstanding observations and modern interpretations. Experimental Hematology, 33(9), 1041–1047.
Li, W., Johnson, S. A., Shelley, W. C., et al. (2003). Primary endothelial cells isolated from the yolk sac and Para-aortic splanchnopleura support the expansion of adult marrow stem cells in vitro. Blood, 102(13), 4345–4353.
McGrath, K. E., & Palis, J. (2005). Hematopoiesis in the yolk sac: more than meets the eye. Experimental Hematology, 33(9), 1021–1028.
Palis, J., Robertson, S., Kennedy, M., Wall, C., & Keller, G. (1999). Development of erythroid and myeloid progenitors in the yolk sac and embryo proper of the mouse. Development, 126(22), 5073–5084.
Sugiyama, D., & Tsuji, K. (2006). Definitive hematopoiesis from endothelial cells in the mouse embryo; a simple guide. Trends in Cardiovascular Medicine, 16(2), 45–49.
Medvinsky, A., & Dzierzak, E. (1996). Definitive hematopoiesis is autonomously initiated by the AGM region. Cell, 86(6), 897–906.
Kumano, K., Chiba, S., Kunisato, A., et al. (2003). Notch1 but not Notch2 is essential for generating hematopoietic stem cells from endothelial cells. Immunity, 18(5), 699–711.
Yoder, M. C., Hiatt, K., Dutt, P., Mukherjee, P., Bodine, D. M., & Orlic, D. (1997). Characterization of definitive lymphohematopoietic stem cells in the day 9 murine yolk sac. Immunity, 7(3), 335–344.
Taoudi, S., Gonneau, C., Moore, K., et al. (2008). Extensive hematopoietic stem cell generation in the AGM region via maturation of VE-cadherin + CD45+ pre-definitive HSCs. Cell Stem Cell, 3(1), 99–108.
Rybtsov, S., Sobiesiak, M., Taoudi, S., et al. (2011). Hierarchical organization and early hematopoietic specification of the developing HSC lineage in the AGM region. The Journal of Experimental Medicine, 208(6), 1305–1315.
Sugiyama, D., Inoue-Yokoo, T., Fraser, S. T., Kulkeaw, K., Mizuochi, C., & Horio, Y. (2011). Embryonic regulation of the mouse hematopoietic niche. The Scientific World Journal, 11, 1770–1780.
Mizuochi, C., Fraser, S. T., Biasch, K., et al. (2012). Intra-aortic clusters undergo endothelial to hematopoietic phenotypic transition during early embryogenesis. PloS One, 7(4), e35763.
North, T., Gu, T. L., Stacy, T., et al. (1999). Cbfa2 is required for the formation of intra-aortic hematopoietic clusters. Development, 126(11), 2563–2575.
Cai, Z., de Bruijn, M., Ma, X., et al. (2000). Haploinsufficiency of AML1 affects the temporal and spatial generation of hematopoietic stem cells in the mouse embryo. Immunity, 13(4), 423–431.
Ling, K. W., Ottersbach, K., Van Hamburg, J. P., et al. (2004). GATA-2 plays two functionally distinct roles during the ontogeny of hematopoietic stem cells. The Journal of Experimental Medicine, 200(7), 871–882.
Lugus, J. J., Chung, Y. S., Mills, J. C., et al. (2007). GATA2 functions at multiple steps in hemangioblast development and differentiation. Development, 134(2), 393–405.
Bigas, A., Guiu, J., & Gama-Norton, L. (2013). Notch and Wnt signaling in the emergence of hematopoietic stem cells. Blood Cells, Molecules, and Diseases, 51(4), 264–270.
Desgrange, A., & Cereghini, S. (2015). Nephron patterning: lessons from Xenopus, zebrafish, and mouse studies. Cell, 4(3), 483–499.
Takasato, M., Osafune, K., Matsumoto, Y., et al. (2004). Identification of kidney mesenchymal genes by a combination of microarray analysis and Sall1-GFP knockin mice. Mechanisms of Development, 121(6), 547–557.
Nishinakamura, R., & Takasato, M. (2005). Essential roles of Sall1 in kidney development. Kidney International, 68(5), 1948–1950.
Sasaki, T., Mizuochi, C., Horio, Y., et al. (2010). Regulation of hematopoietic cell clusters in the placental niche through SCF/kit signaling in embryonic mouse. Development, 137(23), 3941–3952.
McKinney-Freeman, S. L., Naveiras, O., Yates, F., et al. (2009). Surface antigen phenotypes of hematopoietic stem cells from embryos and murine embryonic stem cells. Blood, 114(2), 268–278.
Rosenbauer, F., & Tenen, D. G. (2007). Transcription factors in myeloid development: balancing differentiation with transformation. Nature Reviews Immunology, 7(2), 105–117.
Chittenden, T., Harrington, E. A., O'Connor, R., et al. (1995). Induction of apoptosis by the Bcl-2 homologue Bak. Nature, 374(6524), 733–736.
Oltval, Z. N., Milliman, C. L., & Korsmeyer, S. J. (1993). Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programed cell death. Cell, 74(4), 609–619.
Bae, J., Leo, C. P., Hsu, S. Y., & Hsueh, A. J. (2000). MCL-1S, a splicing variant of the antiapoptotic BCL-2 family member MCL-1, encodes a proapoptotic protein possessing only the BH3 domain. Journal of Biological Chemistry, 275(33), 25255–25261.
Yang, J., Liu, X., Bhalla, K., et al. (1997). Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science, 275(5303), 1129–1132.
Robin, C., Ottersbach, K., Durand, C., et al. (2006). An unexpected role for IL-3 in the embryonic development of hematopoietic stem cells. Developmental Cell, 11(2), 171–180.
Petit-Cocault, L., Volle-Challier, C., Fleury, M., Péault, B., & Souyri, M. (2007). Dual role of Mpl receptor during the establishment of definitive hematopoiesis. Development, 134(16), 3031–3040.
Huang, X., Sakamoto, H., & Ogawa, M. (2009). Thrombopoietin controls proliferation of embryonic multipotent hematopoietic progenitors. Genes to Cells, 14(7), 851–860.
Crisan, M., Kartalaei, P. S., Vink, C., et al. (2015). BMP signalling differentially regulates distinct haematopoietic stem cell types. Nature Communications. doi:10.1038/ncomms9040.
Robert-Moreno, À., Espinosa, L., de la Pompa, J. L., & Bigas, A. (2005). RBPjκ-dependent notch function regulates Gata2 and is essential for the formation of intra-embryonic hematopoietic cells. Development, 132(5), 1117–1126.
Robert-Moreno, À., Guiu, J., Ruiz-Herguido, C., et al. (2008). Impaired embryonic haematopoiesis yet normal arterial development in the absence of the notch ligand Jagged1. The EMBO Journal, 27(13), 1886–1895.
Durand, C., Robin, C., Bollerot, K., Baron, M. H., Ottersbach, K., & Dzierzak, E. (2007). Embryonic stromal clones reveal developmental regulators of definitive hematopoietic stem cells. Proceedings of the National Academy of Sciences, 104(52), 20838–20843.
Peeters, M., Ottersbach, K., Bollerot, K., et al. (2009). Ventral embryonic tissues and hedgehog proteins induce early AGM hematopoietic stem cell development. Development, 136(15), 2613–2621.
Godin, I., Garcia-Porrero, J. A., Dieterlen-Lièvre, F., & Cumano, A. (1999). Stem cell emergence and hemopoietic activity are incompatible in mouse intraembryonic sites. The Journal of Experimental Medicine, 190(1), 43–52.
Guilbert, L. J., & Stanley, E. R. (1986). The interaction of 125I-colony-stimulating factor-1 with bone marrow-derived macrophages. Journal of Biological Chemistry, 261(9), 4024–4032.
Yeung, Y. G., Jubinsky, P. T., Sengupta, A., Yeung, D., & Stanley, E. R. (1987). Purification of the colony-stimulating factor 1 receptor and demonstration of its tyrosine kinase activity. Proceedings of the National Academy of Sciences, 84(5), 1268–1271.
Lin, H., Lee, E., Hestir, K., et al. (2008). Discovery of a cytokine and its receptor by functional screening of the extracellular proteome. Science, 320(5877), 807–811.
Mukouyama, Y. S., Hara, T., Xu, M. J., et al. (1998). In vitro expansion of murine multipotential hematopoietic progenitors from the embryonic aorta–gonad–mesonephros region. Immunity, 8(1), 105–114.
Dakic, A., Metcalf, D., Di Rago, L., Mifsud, S., Wu, L., & Nutt, S. L. (2005). PU. 1 regulates the commitment of adult hematopoietic progenitors and restricts granulopoiesis. The Journal of Experimental Medicine, 201(9), 1487–1502.
Scott, E. W., Fisher, R. C., Olson, M. C., Kehrli, E. W., Simon, M. C., & Singh, H. (1997). PU. 1 functions in a cell-autonomous manner to control the differentiation of multipotential lymphoid–myeloid progenitors. Immunity, 6(4), 437–447.
Iwasaki, H., Somoza, C., Shigematsu, H., et al. (2005). Distinctive and indispensable roles of PU. 1 in maintenance of hematopoietic stem cells and their differentiation. Blood, 106(5), 1590–1600.
Zhang, D. E., Zhang, P., Wang, N. D., Hetherington, C. J., Darlington, G. J., & Tenen, D. G. (1997). Absence of granulocyte colony-stimulating factor signaling and neutrophil development in CCAAT enhancer binding protein α-deficient mice. Proceedings of the National Academy of Sciences, 94(2), 569–574.
Zhang, P., Iwasaki-Arai, J., Iwasaki, H., et al. (2004). Enhancement of hematopoietic stem cell repopulating capacity and self-renewal in the absence of the transcription factor C/EBPα. Immunity, 21(6), 853–863.
Yamanaka, R., Barlow, C., Lekstrom-Himes, J., et al. (1997). Impaired granulopoiesis, myelodysplasia, and early lethality in CCAAT/enhancer binding protein α-deficient mice. Proceedings of the National Academy of Sciences, 94(24), 13187–13192.
Hock, H., Hamblen, M. J., Rooke, H. M., et al. (2003). Intrinsic requirement for zinc finger transcription factor Gfi-1 in neutrophil differentiation. Immunity, 18(1), 109–120.
Tamura, T., Nagamura-Inoue, T., Shmeltzer, Z., Kuwata, T., & Ozato, K. (2000). ICSBP directs bipotential myeloid progenitor cells to differentiate into mature macrophages. Immunity, 13(2), 155–165.
Scheller, M., Foerster, J., Heyworth, C. M., et al. (1999). Altered development and cytokine responses of myeloid progenitors in the absence of transcription factor, interferon consensus sequence binding protein. Blood, 94(11), 3764–3771.
Richardson, E. T., Shukla, S., Nagy, N., Boom, W. H., Beck, R. C., Zhou, L., Landreth, G. E., Harding, C. V., (2015). ERK signaling is essential for macrophage development. PloS One, 10(10), e0140064.
Kelley, T. W., Graham, M. M., Doseff, A. I., Pomerantz, R. W., Lau, S. M., Ostrowski, M. C., Franke, T. F., Marsh, C. B., (1999). Macrophage colony-stimulating factor promotes cell survival through Akt/protein kinase B. Journal of Biological Chemistry, 274(37), 26393–26398.
Cohen, G. M. A. P. (1997). Caspases: the executioners of apoptosis. The Biochemical Journal, 326, 1–16.
Fujita, J., Crane, A. M., Souza, M. K., et al. (2008). Caspase activity mediates the differentiation of embryonic stem cells. Cell Stem Cell, 2(6), 595–601.
Janzen, V., Fleming, H. E., Riedt, T., et al. (2008). Hematopoietic stem cell responsiveness to exogenous signals is limited by caspase-3. Cell Stem Cell, 2(6), 584–594.
Acknowledgments
This work was supported by a Grant-in-Aid for Exploratory Research by the Project for Realization of Regenerative Medicine, of the Ministry of Education, Culture, Sports, Science and Technology; and by a Bilateral grant to promote scientific exchange between Germany and Japan from the Japan Society for the Promotion of Science. We thank Dr. Elise Lamar for critical reading of our manuscript; Ms. Chiyo Yanagi-Mizuochi for technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no potential conflicts of interest.
Electronic Supplementary Material
ESM 1
(DOCX 1379 kb)
Rights and permissions
About this article
Cite this article
Sasaki, T., Tanaka, Y., Kulkeaw, K. et al. Embryonic Intra-Aortic Clusters Undergo Myeloid Differentiation Mediated by Mesonephros-Derived CSF1 in Mouse. Stem Cell Rev and Rep 12, 530–542 (2016). https://doi.org/10.1007/s12015-016-9668-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-016-9668-2