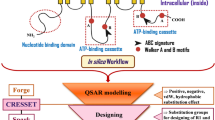
Abstract
Breast cancer resistance protein (ABCG2) is a human ATP-binding cassette (ABC) that plays a paramount role in multidrug resistance (MDR) in cancer therapy. The discovery of ABCG2 inhibitors could assist in designing unprecedented therapeutic strategies for cancer treatment. There is as yet no approved drug targeting ABCG2, although a large number of drug candidates have been clinically investigated. In this work, binding affinities of 181 drug candidates in clinical-trial or investigational stages as ABCG2 inhibitors were inspected using in silico techniques. Based on available experimental data, the performance of AutoDock4.2.6 software was first validated to predict the inhibitor-ABCG2 binding mode and affinity. Combined molecular docking calculations and molecular dynamics (MD) simulations, followed by molecular mechanics-generalized Born surface area (MM-GBSA) binding energy calculations, were then performed to filter out the studied drug candidates. From the estimated docking scores and MM-GBSA binding energies, six auspicious drug candidates—namely, pibrentasvir, venetoclax, ledipasvir, avatrombopag, cobicistat, and revefenacin—exhibited auspicious binding energies with value < −70.0 kcal/mol. Interestingly, pibrentasvir, venetoclax, and ledipasvir were observed to show even higher binding affinities with the ABCG2 transporter with binding energies of < −80.0 kcal/mol over long MD simulations of 100 ns. The stabilities of these three promising candidates in complex with ABCG2 transporter were demonstrated by their energetics and structural analyses throughout the 100 ns MD simulations. The current study throws new light on pibrentasvir, venetoclax, and ledipasvir as curative options for multidrug resistant cancers by inhibiting ABCG2 transporter.







Similar content being viewed by others
References
Gottesman, M. M. (2002). Mechanisms of cancer drug resistance. Annual Review of Medicine, 53, 615–27.
Szakacs, G., Paterson, J. K., Ludwig, J. A., Booth-Genthe, C., & Gottesman, M. M. (2006). Targeting multidrug resistance in cancer. Nature Reviews Drug Discovery, 5, 219–34.
Wu, C. P., Hsieh, C. H., & Wu, Y. S. (2011). The emergence of drug transporter-mediated multidrug resistance to cancer chemotherapy. Molecular Pharmaceutics, 8, 1996–2011.
Robey, R. W., Pluchino, K. M., Hall, M. D., Fojo, A. T., Bates, S. E., & Gottesman, M. M. (2018). Revisiting the role of ABC transporters in multidrug-resistant cancer. Nature Reviews: Cancer, 18, 452–64.
Dassa, E., & Bouige, P. (2001). The ABC of ABCS: a phylogenetic and functional classification of ABC systems in living organisms. Research in Microbiology, 152, 211–29.
Vasiliou, V., Vasiliou, K., & Nebert, D. W. (2009). Human ATP-binding cassette (ABC) transporter family. Human Genomics, 3, 281–90.
Matsuo, H., Takada, T., Ichida, K., Nakamura, T., Nakayama, A., Ikebuchi, Y., Ito, K., Kusanagi, Y., Chiba, T., Tadokoro, S., Takada, Y., Oikawa, Y., Inoue, H., Suzuki, K., Okada, R., Nishiyama, J., Domoto, H., Watanabe, S., Fujita, M., Morimoto, Y., Naito, M., Nishio, K., Hishida, A., Wakai, K., Asai, Y., Niwa, K., Kamakura, K., Nonoyama, S., Sakurai, Y., Hosoya, T., Kanai, Y., Suzuki, H., Hamajima, N., & Shinomiya, N. (2009). Common defects of ABCG2, a high-capacity urate exporter, cause gout: a function-based genetic analysis in a Japanese population. Science Translational Medicine, 1, 5ra11.
Szakacs, G., Varadi, A., Ozvegy-Laczka, C., & Sarkadi, B. (2008). The role of ABC transporters in drug absorption, distribution, metabolism, excretion and toxicity (ADME-Tox). Drug Discovery Today, 13, 379–93.
Doyle, L. A., Yang, W., Abruzzo, L. V., Krogmann, T., Gao, Y., Rishi, A. K., & Ross, D. D. (1998). A multidrug resistance transporter from human MCF-7 breast cancer cells. Proceedings of the National Academy of Sciences of the United States of America, 95, 15665–70.
Litman, T., Brangi, M., Hudson, E., Fetsch, P., Abati, A., Ross, D. D., Miyake, K., Resau, J. H., & Bates, S. E. (2000). The multidrug-resistant phenotype associated with overexpression of the new ABC half-transporter, MXR (ABCG2). Journal of Cell Science, 113, 2011–21.
Perego, P., De Cesare, M., De Isabella, P., Carenini, N., Beggiolin, G., Pezzoni, G., Palumbo, M., Tartaglia, L., Pratesi, G., Pisano, C., Carminati, P., Scheffer, G. L., & Zunino, F. (2001). A novel 7-modified camptothecin analog overcomes breast cancer resistance protein-associated resistance in a mitoxantrone-selected colon carcinoma cell line. Cancer Research, 61, 6034–7.
van Hattum, A. H., Hoogsteen, I. J., Schluper, H. M., Maliepaard, M., Scheffer, G. L., Scheper, R. J., Kohlhagen, G., Pommier, Y., Pinedo, H. M., & Boven, E. (2002). Induction of breast cancer resistance protein by the camptothecin derivative DX-8951f is associated with minor reduction of antitumour activity. British Journal of Cancer, 87, 665–72.
Kage, K., Tsukahara, S., Sugiyama, T., Asada, S., Ishikawa, E., Tsuruo, T., & Sugimoto, Y. (2002). Dominant-negative inhibition of breast cancer resistance protein as drug efflux pump through the inhibition of S-S dependent homodimerization. International Journal of Cancer, 97, 626–30.
Ferreira, R. J., Bonito, C. A., Cordeiro, M., Ferreira, M. U. & Dos Santos, D. (2017). Structure-function relationships in ABCG2: insights from molecular dynamics simulations and molecular docking studies. Scientific Reports, 7, 15534–50.
Litman, T., Jensen, U., Hansen, A., Covitz, K. M., Zhan, Z. R., Fetsch, P., Abati, A., Hansen, P. R., Horn, T., Skovsgaard, T. & Bates, S. E. (2002). Use of peptide antibodies to probe for the mitoxantrone resistance-associated protein MXR/BCRP/ABCP/ABCG2. Biochimica et Biophysica Acta-Biomembranes, 1565, 6–16.
Orlando, B. J. & & Liao, M. (2020). ABCG2 transports anticancer drugs via a closed-to-open switch. Nature Communications, 11, 2264–74.
Iqbal, N. & Iqbal, N. (2014). Imatinib: a breakthrough of targeted therapy in cancer. Chemotherapy Research and Practice, 2014, 357027–36.
Homolya, L., Orban, T. I., Csanady, L. & Sarkadi, B. (2011). Mitoxantrone is expelled by the ABCG2 multidrug transporter directly from the plasma membrane. Biochimica et Biophysica Acta-Biomembranes, 1808, 154–63.
Burger, H., van Tol, H., Boersma, A. W., Brok, M., Wiemer, E. A., Stoter, G., & Nooter, K. (2004). Imatinib mesylate (STI571) is a substrate for the breast cancer resistance protein (BCRP)/ABCG2 drug pump. Blood, 104, 2940–2.
Eadie, L. N., Hughes, T. P., & White, D. L. (2014). Interaction of the efflux transporters ABCB1 and ABCG2 with imatinib, nilotinib, and dasatinib. Clinical Pharmacology and Therapeutics, 95, 294–306.
Houghton, P. J., Germain, G. S., Harwood, F. C., Schuetz, J. D., Stewart, C. F., Buchdunger, E., & Traxler, P. (2004). Imatinib mesylate is a potent inhibitor of the ABCG2 (BCRP) transporter and reverses resistance to topotecan and SN-38 in vitro. Cancer Research, 64, 2333–7.
Kannan, P., Telu, S., Shukla, S., Ambudkar, S. V., Pike, V. W., Halldin, C., Gottesman, M. M., Innis, R. B., & Hall, M. D. (2011). The “specific” P-glycoprotein inhibitor tariquidar is also a substrate and an inhibitor for breast cancer resistance protein (BCRP/ABCG2). ACS Chemical. Neuroscience, 2, 82–9.
Henrich, C. J., Robey, R. W., Bokesch, H. R., Bates, S. E., Shukla, S., Ambudkar, S. V., Dean, M., & McMahon, J. B. (2007). New inhibitors of ABCG2 identified by high-throughput screening. Molecular Cancer Therapeutics, 6, 3271–8.
Quezada, H., Martinez-Vazquez, M., Lopez-Jacome, E., Gonzalez-Pedrajo, B., Andrade, A., Fernandez-Presas, A. M., Tovar-Garcia, A., & Garcia-Contreras, R. (2020). Repurposed anti-cancer drugs: the future for anti-infective therapy? Expert Review of Anti-Infective Therapy, 18, 609–12.
Jackson, S. M., Manolaridis, I., Kowal, J., Zechner, M., Taylor, N. M. I., Bause, M., Bauer, S., Bartholomaeus, R., Bernhardt, G., Koenig, B., Buschauer, A., Stahlberg, H., Altmann, K. H., & Locher, K. P. (2018). Structural basis of small-molecule inhibition of human multidrug transporter ABCG2. Nature Structural & Molecular Biology, 25, 333–40.
Marti-Renom, M. A., Stuart, A. C., Fiser, A., Sanchez, R., Melo, F., & Sali, A. (2000). Comparative protein structure modeling of genes and genomes. Annual Review of Biophysics and Biomolecular Structure, 29, 291–325.
Gordon, J. C., Myers, J. B., Folta, T., Shoja, V., Heath, L. S., & Onufriev, A. (2005). H++: a server for estimating pKas and adding missing hydrogens to macromolecules. Nucleic Acids Research, 33, W368–71.
Wishart, D. S., Knox, C., Guo, A. C., Shrivastava, S., Hassanali, M., Stothard, P., Chang, Z., & Woolsey, J. (2006). DrugBank: a comprehensive resource for in silico drug discovery and exploration. Nucleic Acids Research, 34, D668–72.
Wishart, D. S., Feunang, Y. D., Guo, A. C., Lo, E. J., Marcu, A., Grant, J. R., Sajed, T., Johnson, D., Li, C., Sayeeda, Z., Assempour, N., Iynkkaran, I., Liu, Y., Maciejewski, A., Gale, N., Wilson, A., Chin, L., Cummings, R., Le, D., Pon, A., Knox, C., & Wilson, M. (2018). DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Research, 46, D1074–D82.
Hawkins, P. C., Skillman, A. G., Warren, G. L., Ellingson, B. A., & Stahl, M. T. (2010). Conformer generation with OMEGA: algorithm and validation using high quality structures from the Protein Databank and Cambridge Structural Database. Journal of Chemical Information and Modeling, 50, 572–84.
OMEGA 2.5.1.4. Santa Fe, NM, USA. OpenEye Scientific Software, 2013.
SZYBKI 1.9.0.3. Santa Fe, NM, USA. OpenEye Scientific Software, 2016.
Gasteiger, J., & Marsili, M. (1980). Iterative partial equalization of orbital electronegativity—a rapid access to atomic charges. Tetrahedron, 36, 3219–28.
Morris, G. M., Huey, R., Lindstrom, W., Sanner, M. F., Belew, R. K., Goodsell, D. S., & Olson, A. J. (2009). AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. Journal of Computational Chemistry, 30, 2785–91.
Forli, S., Huey, R., Pique, M. E., Sanner, M. F., Goodsell, D. S., & Olson, A. J. (2016). Computational protein-ligand docking and virtual drug screening with the AutoDock suite. Nature Protocols, 11, 905–19.
Case, D. A., Betz, R. M., Cerutti, D. S., Cheatham, T. E., Darden, T. A., Duke, R. E., Giese, T. J., Gohlke, H., Goetz, A. W., Homeyer, N., Izadi, S., Janowski, P., Kaus, J., Kovalenko, A., Lee, T. S., LeGrand, S., Li, P., Lin, C., Luchko, T., Luo, R., Madej, B., Mermelstein, D., Merz, K. M., Monard, G., Nguyen, H., Nguyen, H. T., Omelyan, I., Onufriev, A., Roe, D. R., Roitberg, A., Sagui, C., Simmerling, C. L., Botello-Smith, W. M., Swails, J., Walker, R. C., Wang, J., Wolf, R. M., Wu, X., Xiao, L., & Kollman, P. A. (2016). AMBER16. San Francisco, USA: University of California.
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A., & Case, D. A. (2004). Development and testing of a general amber force field. Journal of Computational Chemistry, 25, 1157–74.
Maier, J. A., Martinez, C., Kasavajhala, K., Wickstrom, L., Hauser, K. E., & Simmerling, C. (2015). ff14SB: improving the accuracy of protein side chain and backbone parameters from ff99SB. Journal of Chemical Theory and Computation, 11, 3696–713.
Jakalian, A., Jack, D. B., & Bayly, C. I. (2002). Fast, efficient generation of high-quality atomic charges. AM1-BCC model: II. Parameterization and validation. Journal of Computational Chemistry, 23, 1623–41.
Roux, B., & Simonson, T. (1999). Implicit solvent models. Biophysical Chemistry, 78, 1–20.
Bayly, C. I., Cieplak, P., Cornell, W. D., & Kollman, P. A. (1993). A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges - the RESP model. Journal of Physical Chemistry, 97, 10269–80.
Frisch, M. J., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., Scalmani, G., Barone, V., Mennucci, B., Petersson, G. A., Nakatsuji, H., Caricato, M., Li, X., Hratchian, H. P., Izmaylov, A. F., Bloino, J., Zheng, G., Sonnenberg, J. L., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Vreven, T., Montgomery, J. A., Peralta, J. E., Ogliaro, F., Bearpark, M., Heyd, J. J., Brothers, E., Kudin, K. N., Staroverov, V. N., Kobayashi, R., Normand, J., Raghavachari, K., Rendell, A., Burant, J. C., Iyengar, S. S., Tomasi, J., Cossi, M., Rega, N., Millam, J. M., Klene, M., Knox, J. E., Cross, J. B., Bakken, V., Adamo, C., Jaramillo, J., Gomperts, R., Stratmann, R. E., Yazyev, O., Austin, A. J., Cammi, R., Pomelli, C., Ochterski, J. W., Martin, R. L., Morokuma, K., Zakrzewski, V. G., Voth, G. A., Salvador, P., Dannenberg, J. J., Dapprich, S., Daniels, A. D., Farkas, Ö., Foresman, J. B., Ortiz, J. V., Cioslowski, J., & Fox, D. J. (2009). Gaussian 09. Wallingford CT, USA.: Gaussian Inc.
Darden, T., York, D., & Pedersen, L. (1993). Particle mesh Ewald: AnN·log(N) method for Ewald sums in large systems. Journal of Chemical Physics, 98, 10089–92.
Berendsen, H. J. C., Postma, J. P. M., Vangunsteren, W. F., Dinola, A., & Haak, J. R. (1984). Molecular-dynamics with coupling to an external bath. Journal of Chemical Physics, 81, 3684–90.
Dassault Systèmes BIOVIA. (2019). BIOVIA Discovery Studio Visualize 2019, version 2019. San Diego, CA, USA: Dassault Systèmes BIOVIA.
Massova, I., & Kollman, P. A. (2000). Combined molecular mechanical and continuum solvent approach (MM-PBSA/GBSA) to predict ligand binding. Perspectives in Drug Discovery and Design, 18, 113–35.
Onufriev, A., Bashford, D., & Case, D. A. (2004). Exploring protein native states and large-scale conformational changes with a modified generalized born model. Proteins, 55, 383–94.
Liu, Z., Xiao, X., Wei, X., Li, J., Yang, J., Tan, H., Zhu, J., Zhang, Q., Wu, J., & Liu, L. (2020). Composition and divergence of coronavirus spike proteins and host ACE2 receptors predict potential intermediate hosts of SARS-CoV-2. Journal of Medical Virology, 92, 595–601.
Toyoda, Y., Takada, T. & Suzuki, H. (2019). Inhibitors of human ABCG2: from technical background to recent updates with clinical implications. Frontiers in Pharmacology, 10, 208–16.
De Vivo, M., Masetti, M., Bottegoni, G., & Cavalli, A. (2016). Role of molecular dynamics and related methods in drug discovery. Journal of Medicinal Chemistry, 59, 4035–61.
Kerrigan, J. E. (2013) Molecular dynamics simulations in drug design. In S. Kortagere (ed.), In Silico Models for Drug Discovery. (pp. 95–113). Totowa, NJ: Humana Press.
Acknowledgements
M.F.M. extends his appreciation to the Deanship of Scientific Research at King Khalid University for funding this work under grant no. (R.G.P.1/143/42). The computational work was completed with resources supported by the Science and Technology Development Fund, STDF, Egypt, Grants No. 5480 & 7972.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Ethical Approval
This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ibrahim, M.A.A., Badr, E.A.A., Abdelrahman, A.H.M. et al. Prospective Drug Candidates as Human Multidrug Transporter ABCG2 Inhibitors: an In Silico Drug Discovery Study. Cell Biochem Biophys 79, 189–200 (2021). https://doi.org/10.1007/s12013-021-00985-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-021-00985-y