Abstract
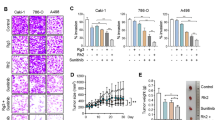
The aim of the study was to evaluate the effect of the hispidulin, a naturally occurring flavonoid, in combination with a new multi-targeted oral medication, sunitinib on renal cell carcinoma (RCC) cell proliferation in vitro and on tumor growth in vivo. After treatment with hispidulin or sunitinib, either alone or in combination, MTT assay was used to examine cell viability and flow cytometry analysis was employed to examine cell cycle distribution and apoptosis of the RCC cell lines 786-0 and Caki-1. Western blotting was employed to examine the expression of proteins related to pStat3 signaling pathway. Furthermore, a xenograft mouse model was applied to study the antitumor efficacy of sunitinib or hispidulin alone or in combination, with immunohistochemistry to detect expression of proteins related to xenograft growth and angiogenesis. Hispidulin dose-dependently inhibited proliferation and induced apoptosis in both of the tested RCC cell lines when used alone; when combined with sunitinib, relatively low concentration of hispidulin enhanced the antitumor activity of the latter. The antitumor activity of hispidulin and its enhancement of the antitumor activity of sunitinib correlated with the suppression of pStat3 signaling and the consequent downregulation of Bcl-2 and survivin. Moreover, combination of hispidulin and sunitinib inhibited the growth and angiogenesis of xenografts generated from Caki-1 significantly. Immunohistochemistry indicated decreased expression of proteins promoting xenograft growth and angiogenesis after combination treatment of hispidulin and sunitinib. Our results showed that hispidulin, by inhibiting pStat3 signaling, exhibited antitumor activity and the joint use of hispidulin and sunitinib could provide greater antitumor efficacy compared to either drug alone. Therefore, combination treatment with hispidulin and sunitinib might offer a novel therapeutic option for patients with RCC.





Similar content being viewed by others
References
Antonelli, A., et al. (2007). The follow-up management of non-metastatic renal cell carcinoma: definition of a surveillance protocol. British Journal of Urology, 99(2), 296–300.
Itsumi, M., & Tatsugami, K. (2010). Immunotherapy for renal cell carcinoma. Clinical and Developmental Immunology, 2010, 284581.
Reddy, K. (2006). Phase III study of sunitinib malate (SU11248) versus interferon-alpha as first-line treatment in patients with metastatic renal cell carcinoma. Clinical Genitourinary Cancer, 5(1), 23–25.
Faivre, S., et al. (2007). Molecular basis for sunitinib efficacy and future clinical development. Nature reviews. Drug discovery, 6(9), 734–745.
Faivre, S., et al. (2006). Safety, pharmacokinetic, and antitumor activity of SU11248, a novel oral multitarget tyrosine kinase inhibitor, in patients with cancer. Journal of Clinical Oncology, 24(1), 25–35.
Motzer, R. J., et al. (2006). Activity of SU11248, a multitargeted inhibitor of vascular endothelial growth factor receptor and platelet-derived growth factor receptor, in patients with metastatic renal cell carcinoma. Journal of Clinical Oncology, 24(1), 16–24.
Motzer, R. J., et al. (2009). Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. Journal of Clinical Oncology, 27(22), 3584–3590.
Way, T. D., et al. (2010). Inhibition of epidermal growth factor receptor signaling by Saussurea involucrata, a rare traditional Chinese medicinal herb, in human hormone-resistant prostate cancer PC-3 cells. Journal of Agriculture and Food Chemistry, 58(6), 3356–3365.
Yin, Y., et al. (2008). Anti-inflammatory and immunosuppressive effect of flavones isolated from Artemisia vestita. Journal of Ethnopharmacology, 120(1), 1–6.
Kavvadias, D., et al. (2004). The flavone hispidulin, a benzodiazepine receptor ligand with positive allosteric properties, traverses the blood-brain barrier and exhibits anticonvulsive effects. British Journal of Pharmacology, 142(5), 811–820.
Tan, R. X., et al. (1999). Mono- and sesquiterpenes and antifungal constituents from Artemisia species. Planta Medica, 65(1), 64–67.
Nagao, T., et al. (2002). Antiproliferative constituents in plants 10. Flavones from the leaves of Lantana montevidensis Briq. and consideration of structure-activity relationship. Biological and Pharmaceutical Bulletin, 25(7), 875–879.
Chen, Y. T., et al. (1990). Flavonoids as superoxide scavengers and antioxidants. Free Radical Biology and Medicine, 9(1), 19–21.
Bourdillat, B., et al. (1988). Mechanism of action of hispidulin, a natural flavone, on human platelets. Progress in Clinical and Biological Research, 280, 211–214.
Niu, X., et al. (2014). The Effects of Hispidulin on Bupivacaine-Induced Neurotoxicity: Role of AMPK Signaling Pathway. Cell Biochemistry and Biophysics,. doi:10.1007/s12013-014-9888-5.
Zhou, R., Wang, Z., & Ma, C. (2013). Hispidulin Exerts Anti-osteoporotic Activity in Ovariectomized Mice via Activating AMPK Signaling Pathway. Cell Biochemistry and Biophysics, 69(2), 311–317.
Nepal, M., et al. (2013). Hispidulin attenuates bone resorption and osteoclastogenesis via the RANKL-induced NF-kappaB and NFATc1 pathways. European Journal of Pharmacology, 715(1–3), 96–104.
Yang, J. M., et al. (2010). Hispidulin sensitizes human ovarian cancer cells to TRAIL-induced apoptosis by AMPK activation leading to Mcl-1 block in translation. Journal of Agriculture and Food Chemistry, 58(18), 10020–10026.
Lin, Y. C., et al. (2010). Hispidulin potently inhibits human glioblastoma multiforme cells through activation of AMP-activated protein kinase (AMPK). Journal of Agriculture and Food Chemistry, 58(17), 9511–9517.
He, L., et al. (2011). Hispidulin, a small flavonoid molecule, suppresses the angiogenesis and growth of human pancreatic cancer by targeting vascular endothelial growth factor receptor 2-mediated PI3 K/Akt/mTOR signaling pathway. Cancer Science, 102(1), 219–225.
Yu, C. Y., et al. (2013). Potential Therapeutic Role of Hispidulin in Gastric Cancer through Induction of Apoptosis via NAG-1 Signaling. Evid Based Complement Alternat Med, 2013, 518301.
Gao, H., Wang, H., & Peng, J. (2014). Hispidulin induces apoptosis through mitochondrial dysfunction and inhibition of P13 k/Akt signalling pathway in HepG2 cancer cells. Cell Biochemistry and Biophysics, 69(1), 27–34.
Jiang, X. L., et al. (2012). Targeting renal cell carcinoma with gambogic acid in combination with sunitinib in vitro and in vivo. Asian Pacific Journal of Cancer Prevention, 13(12), 6463–6468.
Miyake, M., et al. (2012). 5-fluorouracil enhances the antitumor effect of sorafenib and sunitinib in a xenograft model of human renal cell carcinoma. Oncology Letters, 3(6), 1195–1202.
Yu, H., Pardoll, D., & Jove, R. (2009). STATs in cancer inflammation and immunity: a leading role for STAT3. Nature Reviews Cancer, 9(11), 798–809.
Yu, H., & Jove, R. (2004). The STATs of cancer–new molecular targets come of age. Nature Reviews Cancer, 4(2), 97–105.
Yu, H., Kortylewski, M., & Pardoll, D. (2007). Crosstalk between cancer and immune cells: role of STAT3 in the tumour microenvironment. Nature Reviews Immunology, 7(1), 41–51.
Catlett-Falcone, R., et al. (1999). Constitutive activation of Stat3 signaling confers resistance to apoptosis in human U266 myeloma cells. Immunity, 10(1), 105–115.
Bollrath, J., et al. (2009). gp130-mediated Stat3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell, 15(2), 91–102.
Grivennikov, S., et al. (2009). IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell, 15(2), 103–113.
Rebouissou, S., et al. (2009). Frequent in-frame somatic deletions activate gp130 in inflammatory hepatocellular tumours. Nature, 457(7226), 200–204.
Shang, D., et al. (2011). Interferon-alpha induces G1 cell-cycle arrest in renal cell carcinoma cells via activation of Jak-Stat signaling. Cancer Investigation, 29(5), 347–352.
El-Hashemite, N., & Kwiatkowski, D. J. (2005). Interferon-gamma-Jak-Stat signaling in pulmonary lymphangioleiomyomatosis and renal angiomyolipoma: a potential therapeutic target. American Journal of Respiratory Cell and Molecular Biology, 33(3), 227–230.
Horiguchi, A., et al. (2010). STAT3 inhibitor WP1066 as a novel therapeutic agent for renal cell carcinoma. British Journal of Cancer, 102(11), 1592–1599.
Xin, H., et al. (2009). Sunitinib inhibition of Stat3 induces renal cell carcinoma tumor cell apoptosis and reduces immunosuppressive cells. Cancer Research, 69(6), 2506–2513.
Li, S., et al. (2013). Icaritin inhibits JAK/STAT3 signaling and growth of renal cell carcinoma. PLoS ONE, 8(12), e81657.
Banerjee, S., et al. (2009). Antitumor activity of gemcitabine and oxaliplatin is augmented by thymoquinone in pancreatic cancer. Cancer Research, 69(13), 5575–5583.
Masuda, M., et al. (2002). Constitutive activation of signal transducers and activators of transcription 3 correlates with cyclin D1 overexpression and may provide a novel prognostic marker in head and neck squamous cell carcinoma. Cancer Research, 62(12), 3351–3355.
Leslie, K., et al. (2006). Cyclin D1 is transcriptionally regulated by and required for transformation by activated signal transducer and activator of transcription 3. Cancer Research, 66(5), 2544–2552.
Sherr, C. J. (1994). G1 phase progression: cycling on cue. Cell, 79(4), 551–555.
Kitagawa, M., et al. (1996). The consensus motif for phosphorylation by cyclin D1-Cdk4 is different from that for phosphorylation by cyclin A/E-Cdk2. EMBO Journal, 15(24), 7060–7069.
Acknowledgments
This work was supported by National Natural Science Foundation (No. 31470570) and funds from Qingdao University (No. 600201304).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Hui Gao and Qixiao Jiang are contributed equally to this work.
Rights and permissions
About this article
Cite this article
Gao, H., Jiang, Q., Han, Y. et al. Hispidulin Potentiates the Antitumor Effect of Sunitinib Against Human Renal Cell Carcinoma in Laboratory Models. Cell Biochem Biophys 71, 757–764 (2015). https://doi.org/10.1007/s12013-014-0260-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0260-6