Abstract
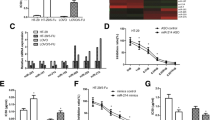
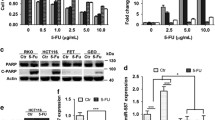
5-Fluorouracil (5-FU) is one of the most commonly used anticancer drugs in the treatment of colon cancer. However, acquired chemoresistance is becoming one of the major challenges for patients with advanced stages of colon cancer. Currently, the mechanisms underlying cancer cell resistance to 5-FU are not fully understood. MicroRNAs (miRNA) have been suggested to play important roles in tumorigenesis and drug resistance in colon cancer. In this study, we generated 5-FU-resistant colon cancer cell lines from which we found that miR-122 was downregulated in 5-FU-resistant cells compared with sensitive cells. Meanwhile, the glucose metabolism is significantly upregulated in 5-FU-resistant cells. We report that PKM2 is a direct target of miR-122 in colon cancer cell. Importantly, overexpression of miR-122 in 5-FU-resistant cells resensitizes 5-FU resistance through the inhibition of PKM2 both in vitro and in vivo. In summary, these findings reveal that the dysregulated glucose metabolism contributes to 5-FU resistance, and glycolysis inhibition by miR-122 might be a promising therapeutic strategy to overcome 5-FU resistance.




Similar content being viewed by others
References
Longley, D. B., Harkin, D. P., & Johnston, P. G. (2003). 5-Fluorouracil: Mechanisms of action and clinical strategies. Nature Reviews Cancer, 3, 330–338.
Curtin, N. J., Harris, A. L., & Aherne, G. W. (1991). Mechanism of cell death following thymidylate synthase inhibition: 2′-Deoxyuridine-5′-triphosphate accumulation, DNA damage, and growth inhibition following exposure to CB3717 and dipyridamole. Cancer Research, 51, 2346–2352.
Peters, G. J., van Triest, B., Backus, H. H., Kuiper, C. M., van der Wilt, C. L., & Pinedo, H. M. (2000). Molecular downstream events and induction of thymidylate synthase in mutant and wild-type p53 colon cancer cell lines after treatment with 5-fluorouracil and the thymidylate synthase inhibitor raltitrexed. European Journal of Cancer, 36, 916–924.
Zhang, N., Yin, Y., Xu, S. J., & Chen, W. S. (2008). 5-Fluorouracil: Mechanisms of resistance and reversal strategies. Molecules, 13, 1551–1569.
De Angelis, P. M., Svendsrud, D. H., Kravik, K. L., & Stokke, T. (2006). Cellular response to 5-fluorouracil (5-FU) in 5-FU-resistant colon cancer cell lines during treatment and recovery. Molecular Cancer, 5, 20.
Violette, S., Poulain, L., Dussaulx, E., Pepin, D., Faussat, A. M., Chambaz, J., et al. (2002). Resistance of colon cancer cells to long-term 5-fluorouracil exposure is correlated to the relative level of Bcl-2 and Bcl-X(L) in addition to Bax and p53 status. International Journal of Cancer, 98, 498–504.
Wu, S., & Le, H. (2013). Dual roles of PKM2 in cancer metabolism. Acta Biochimica et Biophysica Sinica (Shanghai), 45, 27–35.
Israelsen, W. J., Dayton, T. L., Davidson, S. M., Fiske, B. P., Hosios, A. M., Bellinger, G., et al. (2013). PKM2 isoform-specific deletion reveals a differential requirement for pyruvate kinase in tumor cells. Cell, 155, 397–409.
Christofk, H. R., Vander Heiden, M. G., Harris, M. H., Ramanathan, A., Gerszten, R. E., Wei, R., et al. (2008). The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumor growth. Nature, 452, 230–233.
Schetter, A. J., Okayama, H., & Harris, C. C. (2012). The role of microRNAs in colorectal cancer. Cancer Journal, 18, 244–252.
Zhou, Y., Tozzi, F., Chen, J., Fan, F., Xia, L., Wang, J., et al. (2012). Intracellular ATP levels are a pivotal determinant of chemoresistance in colon cancer cells. Cancer Research, 72, 304–314.
Warburg, O. (1956). On respiratory impairment in cancer cells. Science, 124, 269–270.
Kroemer, G., & Pouyssegur, J. (2008). Tumor cell metabolism: Cancer’s Achilles’ heel. Cancer Cell, 13, 472–482.
Vander Heiden, M. G., Cantley, L. C., & Thompson, C. B. (2009). Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science, 324, 1029–1033.
Dang, C. V. (2010). Rethinking the Warburg effect with Myc micromanaging glutamine metabolism. Cancer Research, 70, 859–862.
Zhao, Y., Butler, E. B., & Tan, M. (2013). Targeting cellular metabolism to improve cancer therapeutics. Cell Death and Disease, 4, e532.
Vander Heiden, M. G. (2011). Targeting cancer metabolism: A therapeutic window opens. Nature Reviews Drug Discovery, 10, 671–684.
Ameres, S. L., & Zamore, P. D. (2013). Diversifying microRNA sequence and function. Nature Reviews Molecular Cell Biology, 14, 475–488.
Yang, L., & Belaguli, N. (2009). Berger DH MicroRNA and colorectal cancer. World Journal of Surgery, 33, 446–638.
Tazawa, H., Tsuchiya, N., Izumiya, M., & Nakagama, H. (2007). Tumor-suppressive miR-34a induces senescence-like growth arrest through modulation of the E2F pathway in human colon cancer cells. Proceedings of the National Academy of Sciences of the United States of America, 104, 15472–15477.
Bandres, E., Bitarte, N., Arias, F., Agorreta, J., Fortes, P., Agirre, X., et al. (2009). microRNA-451 regulates macrophage migration inhibitory factor production and proliferation of gastrointestinal cancer cells. Clinical Cancer Research, 15, 2281–2290.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
He, J., Xie, G., Tong, J. et al. Overexpression of MicroRNA-122 Re-sensitizes 5-FU-Resistant Colon Cancer Cells to 5-FU Through the Inhibition of PKM2 In Vitro and In Vivo. Cell Biochem Biophys 70, 1343–1350 (2014). https://doi.org/10.1007/s12013-014-0062-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0062-x