Abstract
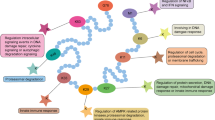
Ubiquitin (Ub) and ubiquitin-like (Ubl) proteins regulate a variety of important cellular processes by forming covalent conjugates with target proteins or lipids. Ubl conjugation is catalyzed by a cascade of proteins including activating enzymes (E1), conjugating enzymes (E2), and in many cases ligation enzymes (E3). The discovery of MLN4924 (Brownell et al., Mol Cell 37: 102–111, 1), an investigational small molecule that is a mechanism-based inhibitor of NEDD8-activating enzyme (NAE), reveals a promising strategy of targeting E1/Ubl pathway for therapeutic purposes. In order to better understand, the biochemical dynamics of Ubl conjugation in cells and tissues, we have developed a mass spectrometry-based method to quantify E1 and Ubls using isotope-labeled proteins as internal standards. Furthermore, we have used the described method to quantify levels of the covalent Nedd8-inhibitor adduct formed in MLN4924 treated cells and tissues. The Nedd8–MLN4924 adduct is a tight-binding inhibitor of NAE, and its cellular concentration represents an indirect pharmacodynamic readout of NAE/Nedd8 pathway inhibition.





Similar content being viewed by others
References
Brownell, J. E., Sintchak, M. D., Gavin, J. M., Liao, H., Bruzzese, F. J., Bump, N. J., et al. (2010). Substrate-assisted inhibition of ubiquitin-like protein-activating enzymes: The NEDD8 E1 inhibitor MLN4924 forms a NEDD8–AMP mimetic in situ. Molecular Cell, 37, 102–111.
Welchman, R. L., Gordon, C., & Mayer, R. J. (2005). Ubiquitin and ubiquitin-like proteins as multifunctional signals. Nature Reviews Molecular Cell Biology, 6, 599–609.
Schlesinger, D. H., Goldstein, G., & Niall, H. D. (1975). The complete amino acid sequence of ubiquitin, an adenylate cyclase stimulating polypeptide probably universal in living cells. Biochemistry, 14, 2214–2218.
Mayer, R. J., Landon, M., & Layfield, R. (1998). Ubiquitin superfolds: intrinsic and attachable regulators of cellular activities? Folding and Design, 3, R97–R99.
Hochstrasser, M. (2009). Origin and function of ubiquitin-like protein. Nature, 458, 422–429.
Bedford, L., Lowe, J., Dick, L. R., Mayer, R. J., & Brownell, J. E. (2011). Ubiquitin-like protein conjugation and the ubiquitin–proteasome system as drug targets. Nature Reviews Drug Discovery, 10, 29–46.
Kerscher, O., Felberbaum, R., & Hochstrasser, M. (2006). Modification of proteins by ubiquitin and ubiquitin-like proteins. Annual Review of Cell and Developmental Biology, 22, 159–180.
Komander, D., Clague, M. J., & Urbé, S. (2009). Breaking the chains: structure and function of the deubiquitinases. Nature Reviews Molecular Cell Biology, 10, 550–563.
Soucy, T. A., Smith, P. G., Milhollen, M. A., Berger, A. J., Gavin, J. M., Adhikari, S., et al. (2009). An inhibitor of NEDD8-activating enzyme as a new approach to treat cancer. Nature, 458, 732–736.
Chen, J. J., Tsu, C. A., Gavin, J. M., Milhollen, M. A., Bruzzese, F. J., Mallender, W. D., et al. (2011). Mechanistic studies of substrate-assisted inhibition of ubiquitin-activating enzyme by adenosine sulfamate analogues. Journal of Biological Chemistry, 286, 40867–40877.
Soucy, T. A., Dick, L. R., Smith, P. G., Milhollen, M. A., & Brownell, J. E. (2010). The NEDD8 conjugation pathway and its relevance in cancer biology and therapy. Genes Cancer, 1, 708–716.
Soucy, T. A., Smith, P. G., & Rolfe, M. (2009). Targeting NEDD8-activated cullin-RING ligases for the treatment of cancer. Clinical Cancer Research, 15, 3912–3916.
Dantuma, N. P., Lindsten, K., Glas, R., Jellne, M., & Masucci, M. G. (2000). Short-lived green fluorescent proteins for quantifying ubiquitin/proteasome-dependent proteolysis in living cells. Nature Biotechnology, 18, 538–543.
Kaiser, S. E., Riley, B. E., Shaler, T. A., Trevino, R. S., Becker, C. H., Schulman, H., et al. (2011). Protein standard absolute quantification (PSAQ) method for the measurement of cellular ubiquitin pools. Nature Methods, 8, 691–696.
Hanke, S., Besir, H., Oesterhelt, D., & Mann, M. (2008). Absolute SILAC for accurate quantitation of proteins in complex mixtures down to the attomole level. Journal of Proteome Research, 7, 1118–1130.
Stephen, W. H., Paul, F. G. S., & Claire, E. E. (2012). The use of selected reaction monitoring in quantitative proteomics. Bioanalysis, 4, 1763–1786.
Shevchenko, A., Tomas, H., Havlis, J., Olsen, J. V., & Mann, M. (2006). In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nature Protocols, 1, 2856–2860.
Cox, J., & Mann, M. (2008). MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nature Biotechnology, 26, 1367–1372.
Gerber, S. A., Rush, J., Stemman, O., Kirschner, M. W., & Gygi, S. P. (2003). Absolute quantification of proteins and phosphoproteins from cell lysates by tandem MS. Proceedings of the National Academy of Sciences of the United States of America, 100, 6940–6945.
Ren, J., Wang, Y., Gao, Y., Mehta, S. B., & Lee, C. G. (2011). Fat10 mediates the effect of TNF-α in inducing chromosomal instability. Journal of Cell Science, 124, 3665–3675.
Chiu, Y. H., Sun, Q., & Chen, Z. J. (2007). E1–L2 activates both ubiquitin and FAT10. Molecular Cell, 27, 1014–1023.
Acknowledgments
The authors would like to thank J. Bolen for support and guidance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, X., Brownell, J.E., Xu, Q. et al. Absolute Quantification of E1, Ubiquitin-Like Proteins and Nedd8–MLN4924 Adduct by Mass Spectrometry. Cell Biochem Biophys 67, 139–147 (2013). https://doi.org/10.1007/s12013-013-9625-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-013-9625-5