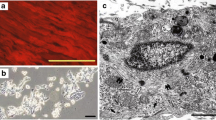
Abstract
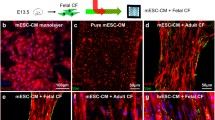
Parietal endoderm-secreted S100A4 promotes early cardiomyogenesis in embryoid bodies [1]. After an acute ischemic event, S100A4 protein appears in cardiac myocytes only in the border zone in rat and human hearts [2]. In wound research, a large outward current of 4 μA/cm2 was always measured at the wound edges of rat cornea and human skin [3]. We hypothesize that a special electrical circumstance at the border zone may contribute to the phenomenon. An electric stimulation system was designed to give the cells electric pulse current stimulation (EPCS), the feature of the signal is pulse polarity altered one after another, rectangular 2 ms, 2 Hz, 40 μA. This intensity of stimulation is proved to be safe to cardiac myocytes (both in structure and beating behavior compared with the cardiac myocytes which do not receive stimulation) and MSCs (in cell vitality, proliferation, cell cycle, and gap junction generation potential) through our previous work. Canine MSCs are capable of generating voltage-sensitive Ca2+ channel and Na+ channels and generating the Ca2+ handling system during differentiation. We found that CD44 was reduced in the MSCs monolayer treated with EPCS, compared with non-stimulated MSCs; and EPCS MSCs (3 h/day, 6 h/day, 5 days) showed an 14.04 ± 3.44 and 14.55 ± 3.97 % reduction in CD44, compared with the cotemporary MSCs; these reveal that CD44 reduction amplitude is not correlated with time for EPCS disposure and CD29 (integrin β1) expression is not affected by EPCS exposure. EPCS was given to the MSCs and cardiac myocytes coculture monolayer (ratio 3:1) for different time (1, 3, and 6 h/day) for 4 days to see the biological effects. Gap junction protein and troponin T show an increase after EPCS. We found that the gap junction protein Cx43 increased with treating time—in the EPCS group, it exhibited 1.5 and 1.7 fold in the 3 h/day group and 6 h/day group (P < 0.01), and troponin T exhibited to about 3.6 and 4.4 fold in the 3 h/day group (P < 0.01) and 6 h/day group (P < 0.05). Since coculture was used as stimuli, immunofluorescence was used to visualize the changes during EPCS for the purpose of elucidating the impact of EPCS on cardiac myocytes and MSCs. We found that after 5 days exposure, EPCS can enhance the expression of S100A4, which is 2.33 fold in cardiac myocytes (P < 0.01) and 1.99 fold in MSCs (P < 0.01) in gray value. A significant increasing expression of the myocyte enhancer factor (MEF) and GATA4 is detected in neonatal rat cardiac myocytes (P < 0.01) compared with cotemporary coculture monolayer in the control group. Also, EPCS can trigger the assembly of MEF2c in the nuclei. In addition, more cardiac myocytes were found to have two nuclei. But MSCs fail to active MEF2C transcriptional factor like that in cardiac myocytes after EPCS exposure. The elevation of MEF2 in both cytoplasm and nuclei of cardiac myocytes can always make a clear distinction of the cardiac myocytes and MSCs in coculture. Some factors show strong upregulation tendency with EPCS in both cardiac myocytes and MSCs—these include the troponin T (P < 0.01) and Cx43 (P < 0.05) in cardiac myocytes, and troponin T (P < 0.01) and Cx43 (P < 0.01) in MSCs. Collagen I expression is not affected with EPCS. In conclusion, mild EPCS can upregulate the secretion of S100A4 in both cardiac myocytes and MSCs, which is a factor supporting the cardiomyogenesis and angiogenesis; it further triggers the development of neonatal rat cardiac myocytes through upregulation of MEF2C and GATA4, the number of cardiac myocytes with two nuclei increases with EPCS, but this phenomenon does not appear in MSCs. Despite this, Cx43 and troponin T in both cardiac myocytes and MSCs are very sensitive to EPCS. EPCS can act as an effective and multi-targeted physical intervention method in cardiomyogenesis.














Similar content being viewed by others
Abbreviations
- CaMKII:
-
The Ca2+/calcium-dependent protein kinases II
- CaN:
-
Calcineurin
- Cx43:
-
Connexin 43
- EPCS:
-
Electrical pulse current stimulation
- FSP:
-
Fibroblast-specific protein
- HCN channels:
-
Hyperpolarization-activated, cyclic nucleotide-gated cation channels
- MEF:
-
Myocyte enhancer factor
- MHC:
-
Myosin heavy chain
- MSC:
-
Mesenchymal stem cell
- PVTs:
-
Polymorphic ventricular tachyarrhythmias
- TTX:
-
Tetrodotoxin
- VSCC:
-
Voltage sensitive calcium channels
References
Stary, M., Schneider, M., Sheikh, S. P., & Weitzer, G. (2006). Parietal endoderm secreted S100A4 promotes early cardiomyogenesis in embryoid bodies. Biochemical and Biophysical Research Communications, 343, 555–563.
Schneider, M., Kostin, S., Strøm, C. C., et al. (2007). S100A4 is upregulated in injured myocardium and promotes growth and survival of cardiac myocytes. Cardiovascular Research, 75, 40–50.
Zhao, M., Song, B., Pu, J., et al. (2006). Electrical signals control wound healing through phosphatidylinositol-3-OH kinase-γ and PTEN. Nature, 442, 457–460.
Thawer, H. A., & Houghton, P. E. (2001). Effects of electrical stimulation on the histological properties of wounds in diabetic mice. Wound Repair and Regeneration, 9, 107–115.
Tsai, M. T., Li, W. J., Tuan, R. S., et al. (2009). Modulation of osteogenesis in human mesenchymal stem cells by specific pulsed electromagnetic field stimulation. Journal of Orthopaedic Research, 27, 1169–1174.
Borgens, R. B., Vanable, J. W., & Jaffe, L. F. (1977). Bioelectricity and regeneration: Large currents leave the stumps of regenerating newt limbs. Proceedings of the National Academy of Sciences, 74, 4528–4532.
Berger, H. J., Prasad, S. K., Davidoff, A. J., et al. (1994). Continual electric field stimulation preserves contractile function of adult ventricular myocytes in primary culture. American Journal of Physiology, 266, 341–349.
Radisic, M., Park, H., Shing, H., et al. (2004). Functional assembly of engineered myocardium by electrical stimulation of cardiac myocytes cultured on scaffolds. Proceedings of the National Academy of Sciences, 101, 18129–18134.
Pittenger, M. F., & Martin, B. J. (2004). Mesenchymal stem cells and their potential as cardiac therapeutics. Circulation Research, 95, 9–20.
Orlic, D., Hill, J. M., & Arai, A. E. (2002). Stem cells for myocardial regeneration. Circulation Research, 91, 1092–1102.
Shake, J. G., Gruber, P. J., Baumgartne, W. A., et al. (2002). Mesenchymal stem cell implantation in a swine myocardial infarct model: Engraftment and functional effects. The Annals of Thoracic Surgery, 73, 1919–1926.
Boomsma, R. A., & Geenen, D. L. (2012). Mesenchymal stem cells secrete multiple cytokines that promote angiogenesis and have contrasting effects on chemotaxis and apoptosis. PLoS One, 7, e35685.
Plotnikov, A. N., Shlapakova, I., Szabolcs, M. J., et al. (2007). Xenografted adult human mesenchymal stem cells provide a platform for sustained biological pacemaker function in canine heart. Circulation, 116, 706–713.
Potapova, I., Plotnikov, A., Lu, Z., et al. (2004). Human mesenchymal stem cells as a gene delivery system to create cardiac pacemakers. Circulation Research, 94, 952–959.
Takano, H., Ohtsuka, M., Akazawa, H., et al. (2003). Pleiotropic effects of cytokines on acute myocardial infarction: G-CSF as a novel therapy for acute myocardial infarction. Current Pharmaceutical Design, 9, 1121–1127.
Orlic, D., Kajstura, J., Chimenti, S., et al. (2001). Mobilized bone marrow cells repair the infarcted heart, improving function and survival. Proceedings of the National academy of Sciences of the United States of America, 98, 10344–10349.
Cooper, M. S. (1995). Membrane potential perturbations induced in tissue cells by pulsed electric fields. Bioelectromagnetics, 16, 255–262.
Grayson, W. L., Martens, T. P., Eng, G. M., et al. (2009). Biomimetic approach to tissue engineering. Seminars in Cell & Developmental Biology, 20, 665–673.
Soleimani, M., & Nadri, S. (2009). A protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow. Nature Protocols, 4, 102–106.
Jun, C., Zhihui, Z., Lu, W., et al. (2012). Canine bone marrow mesenchymal stromal cells with lentiviral mHCN4 gene transfer create cardiac pacemakers. Cytotherapy, 14, 529–539.
Ieda, M., Fu, J. D., Delgado-Olguin, P., et al. (2010). Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell, 142, 375–386.
Ambartsumian, N., Klingelhöfer, J., Grigorian, M., et al. (2001). The metastasis-associated Mts1 (S100A4) protein could act as an angiogenic factor. Oncogene, 20, 4685.
Semov, A., Moreno, M. J., Onichtchenko, A., et al. (2005). Metastasis-associated protein S100A4 induces angiogenesis through interaction with annexin II and accelerated plasmin formation. Journal of Biological Chemistry, 280, 20833–20841.
Zeisberg, E. M., & Kalluri, R. (2010). Origins of cardiac fibroblasts. Circulation Research, 107, 1304–1312.
Hatzistergos, K. E., Quevedo, H., Oskouei, B. N., et al. (2010). Bone marrow mesenchymal stem cells stimulate cardiac stem cell proliferation and differentiation novelty and significance. Circulation Research, 107, 913–922.
Martherus, R. S. R. M., Vanherle, S. J. V., Timmer, E. D. J., et al. (2010). Electrical signals affect the cardiomyocyte transcriptome independently of contraction. Physiological Genomics, 42, 283–289.
Filatov, V., Katrukha, A., Bulargina, T., et al. (1999). Troponin: structure, properties, and mechanism of functioning. Biochemistry c/c of Biokhimia, 64, 969–985.
Ohtsuki, I. (1999). Calcium ion regulation of muscle contraction: The regulatory role of troponin T. Molecular and Cellular Biochemistry, 190, 33–38.
Chang, M. G., Tung, L., Sekar, R. B., et al. (2006). Proarrhythmic potential of mesenchymal stem cell transplantation revealed in an in vitro coculture model. Circulation, 113, 1832–1841.
Danik, S. B., Rosner, G., Lader, J., et al. (2008). Electrical remodeling contributes to complex tachyarrhythmias in connexin43-deficient mouse hearts. The Journal of the federatIon of American Societies for Experimental Biology, 22, 1204–1212.
Wang, D., Shen, W., Zhang, F., et al. (2010). Connexin43 promotes survival of mesenchymal stem cells in ischaemic heart. Cell Biology International, 34, 415–423.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 30700147, No. 30871060) and Key Program of National Science Foundation of Chongqing (CSTC2008BA5005). We would like to acknowledge professor Xian guang Ma (Department of Physics, Third Military Medical University, Chongqing, China) and Teacher Gui Jin (Department of Electronics and Electronical Engineering, Third Military Medical University, Chongqing, China) for kind advice and help in stimulation machine making.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wen, L., Zhang, C., Nong, Y. et al. Mild Electrical Pulse Current Stimulation Upregulates S100A4 and Promotes Cardiogenesis in MSC and Cardiac Myocytes Coculture Monolayer. Cell Biochem Biophys 65, 43–55 (2013). https://doi.org/10.1007/s12013-012-9402-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-012-9402-x