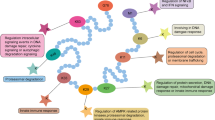
Abstract
The deubiquitylating enzyme USP7 (HAUSP) sits at a critical node regulating the activities of numerous proteins broadly characterized as tumor suppressors, DNA repair proteins, immune responders, viral proteins, and epigenetic modulators. Aberrant USP7 activity may promote oncogenesis and viral disease making it a compelling target for therapeutic intervention. Disclosed drug discovery programs have identified inhibitors of USP7 such as P005091 with cellular proof of concept and anti-proliferative activity in cancer models. Taken together, USP7 inhibitors hold promise as a new strategy for the treatment of disease.


Similar content being viewed by others
Abbreviations
- ATM:
-
Ataxia telangiectasia mutated
- ATR:
-
Ataxia telangiectasia and Rad3-related protein
- DAXX:
-
Death domain associated protein
- DUB:
-
Deubiquitylating enzyme
- EBNA1:
-
Epstein-Barr nuclear antigen 1
- EBV:
-
Epstein-Barr virus
- EGF:
-
Epidermal growth factor
- FOXO:
-
Forkhead box O
- GMPS:
-
Guanosine monophosphate synthase
- HDAC:
-
Histone deacetylase
- HDM2:
-
Human double minute 2
- HDMX:
-
Human double minute 4
- HSV:
-
Herpes simplex virus
- ICP0:
-
Infected cell protein 0
- PI3K:
-
Phosphatidylinositol-3-kinase
- PML:
-
Promyelocytic leukemia protein
- Pc:
-
Polycomb
- PRC1:
-
Polycomb repressive complex 1
- PTEN:
-
Phosphatase and tensin homologue
- TLR:
-
Toll-like receptor
- TRAF:
-
Tumor necrosis factor-receptor associated factor
- USP7:
-
Ubiquitin specific peptidase 7
References
Hershko, A., & Ciechanover, A. (1998). The ubiquitin system. Annual Review of Biochemistry, 67, 425–479.
Nicholson, B., Marblestone, J. G., Butt, T. R., & Mattern, M. R. (2007). Deubiquitinating enzymes as novel anticancer targets. Future Oncology, 3, 191–199.
Daviet, L., & Colland, F. (2008). Targeting ubiquitin specific proteases for drug discovery. Biochimie, 90, 270–283.
Reyes-Turcu, F. E., Ventii, K. H., & Wilkinson, K. D. (2009). Regulation and cellular roles of ubiquitin-specific deubiquitinating enzymes. Annual Review of Biochemistry, 78, 363–397.
Everett, R. D., Meredith, M., Orr, A., Cross, A., Kathoria, M., & Parkinson, J. (1997). A novel ubiquitin-specific protease is dynamically associated with the PML nuclear domain and binds to a herpesvirus regulatory protein. EMBO Journal, 16, 1519–1530.
Sowa, M. E., Bennett, E. J., Gygi, S. P., & Harper, J. W. (2009). Defining the human deubiquitinating enzyme interaction landscape. Cell, 138, 389–403.
Kessler, B. M., Fortunati, E., Melis, M., Pals, C. E., Clevers, H., & Maurice, M. M. (2007). Proteome changes induced by knock-down of the deubiquitylating enzyme HAUSP/USP7. Journal of Proteome Research, 6, 4163–4172.
Jones, S. N., Roe, A. E., Donehower, L. A., & Bradley, A. (1995). Rescue of embryonic lethality in Mdm2-deficient mice by absence of p53. Nature, 378, 206–208.
Montes de Oca Luna, R., Wagner, D. S., & Lozano, G. (1995). Rescue of early embryonic lethality in mdm2-deficient mice by deletion of p53. Nature, 378, 203–206.
Li, M., Chen, D., Shiloh, A., Luo, J., Nikolaev, A. Y., Qin, J., et al. (2002). Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization. Nature, 416, 648–653.
Li, M., Brooks, C. L., Kon, N., & Gu, W. (2004). A dynamic role of HAUSP in the p53-Mdm2 pathway. Molecular Cell, 13, 879–886.
Cummins, J. M., Rago, C., Kohli, M., Kinzler, K. W., Lengauer, C., & Vogelstein, B. (2004). Tumour suppression: Disruption of HAUSP gene stabilizes p53. Nature, 428, 1 pg following 486.
Hu, M., Gu, L., Li, M., Jeffrey, P. D., Gu, W., & Shi, Y. (2006). Structural basis of competitive recognition of p53 and MDM2 by HAUSP/USP7: implications for the regulation of the p53-MDM2 pathway. PLoS Biology, 4, e27.
Meulmeester, E., Maurice, M. M., Boutell, C., Teunisse, A. F., Ovaa, H., Abraham, T. E., et al. (2005). Loss of HAUSP-mediated deubiquitination contributes to DNA damage-induced destabilization of Hdmx and Hdm2. Molecular Cell, 18, 565–576.
Kon, N., Kobayashi, Y., Li, M., Brooks, C. L., Ludwig, T., & Gu, W. (2010). Inactivation of HAUSP in vivo modulates p53 function. Oncogene, 29, 1270–1279.
Tachibana, M., Shibakita, M., Ohno, S., Kinugasa, S., Yoshimura, H., Ueda, S., et al. (2002). Expression and prognostic significance of PTEN product protein in patients with esophageal squamous cell carcinoma. Cancer, 94, 1955–1960.
Perren, A., Komminoth, P., Saremaslani, P., Matter, C., Feurer, S., Lees, J. A., et al. (2000). Mutation and expression analyses reveal differential subcellular compartmentalization of PTEN in endocrine pancreatic tumors compared to normal islet cells. American Journal of Pathology, 157, 1097–1103.
Trotman, L. C., Wang, X., Alimonti, A., Chen, Z., Teruya-Feldstein, J., Yang, H., et al. (2007). Ubiquitination regulates PTEN nuclear import and tumor suppression. Cell, 128, 141–156.
Song, M. S., Salmena, L., Carracedo, A., Egia, A., Lo-Coco, F., Teruya-Feldstein, J., et al. (2008). The deubiquitinylation and localization of PTEN are regulated by a HAUSP-PML network. Nature, 455, 813–817.
Huang, H., & Tindall, D. J. (2007). Dynamic FoxO transcription factors. Journal of Cell Science, 120, 2479–2487.
van der Horst, A., de Vries-Smits, A. M., Brenkman, A. B., van Triest, M. H., van den Broek, N., Colland, F., et al. (2006). FOXO4 transcriptional activity is regulated by monoubiquitination and USP7/HAUSP. Nature Cell Biology, 8, 1064–1073.
Brenkman, A. B., de Keizer, P. L., van den Broek, N. J., van der Groep, P., van Diest, P. J., van der Horst, A., et al. (2008). The peptidyl-isomerase Pin1 regulates p27kip1 expression through inhibition of Forkhead box O tumor suppressors. Cancer Research, 68, 7597–7605.
Brenkman, A. B., de Keizer, P. L., van den Broek, N. J., Jochemsen, A. G., & Burgering, B. M. (2008). Mdm2 induces mono-ubiquitination of FOXO4. PLoS One, 3, e2819.
Yang, J. Y., Zong, C. S., Xia, W., Yamaguchi, H., Ding, Q., Xie, X., et al. (2008). ERK promotes tumorigenesis by inhibiting FOXO3a via MDM2-mediated degradation. Nature Cell Biology, 10, 138–148.
Gil, J., & Peters, G. (2006). Regulation of the INK4b-ARF-INK4a tumour suppressor locus: All for one or one for all. Nature Reviews Molecular Cell Biology, 7, 667–677.
Maertens, G. N., El Messaoudi-Aubert, S., Elderkin, S., Hiom, K., & Peters, G. (2010). Ubiquitin-specific proteases 7 and 11 modulate polycomb regulation of the INK4a tumour suppressor. EMBO Journal, 29, 2553–2565.
de Bie, P., Zaaroor-Regev, D., Ciechanover, A. (2010) Regulation of the polycomb protein RING1B ubiquitination by USP7. Biochemical and Biophysical Research Communications, 400(3), 389–395.
Henry, K. W., Wyce, A., Lo, W. S., Duggan, L. J., Emre, N. C., Kao, C. F., et al. (2003). Transcriptional activation via sequential histone H2B ubiquitylation and deubiquitylation, mediated by SAGA-associated Ubp8. Genes and Development, 17, 2648–2663.
Sun, Z. W., & Allis, C. D. (2002). Ubiquitination of histone H2B regulates H3 methylation and gene silencing in yeast. Nature, 418, 104–108.
Wang, H., Wang, L., Erdjument-Bromage, H., Vidal, M., Tempst, P., Jones, R. S., et al. (2004). Role of histone H2A ubiquitination in polycomb silencing. Nature, 431, 873–878.
Sarkari, F., Sanchez-Alcaraz, T., Wang, S., Holowaty, M. N., Sheng, Y., & Frappier, L. (2009). EBNA1-mediated recruitment of a histone H2B deubiquitylating complex to the Epstein-Barr virus latent origin of DNA replication. PLoS Pathogens, 5, e1000624.
van der Knaap, J. A., Kumar, B. R., Moshkin, Y. M., Langenberg, K., Krijgsveld, J., Heck, A. J., et al. (2005). GMP synthetase stimulates histone H2B deubiquitylation by the epigenetic silencer USP7. Molecular Cell, 17, 695–707.
van der Knaap, J. A., Kozhevnikova, E., Langenberg, K., Moshkin, Y. M., & Verrijzer, C. P. (2010). Biosynthetic enzyme GMP synthetase cooperates with ubiquitin-specific protease 7 in transcriptional regulation of ecdysteroid target genes. Molecular and Cellular Biology, 30, 736–744.
Hirst, M., Haliday, E., Nakamura, J., & Lou, L. (1994). Human GMP synthetase. Protein purification, cloning, and functional expression of cDNA. Journal of Biological Chemistry, 269, 23830–23837.
Boritzki, T. J., Jackson, R. C., Morris, H. P., & Weber, G. (1981). Guanosine-5′-phosphate synthetase and guanosine-5′-phosphate kinase in rat hepatomas and kidney tumors. Biochimica et Biophysica Acta, 658, 102–110.
Weber, G., Burt, M. E., Jackson, R. C., Prajda, N., Lui, M. S., & Takeda, E. (1983). Purine and pyrimidine enzymic programs and nucleotide pattern in sarcoma. Cancer Research, 43, 1019–1023.
Mossman, K. L., Saffran, H. A., & Smiley, J. R. (2000). Herpes simplex virus ICP0 mutants are hypersensitive to interferon. Journal of Virology, 74, 2052–2056.
Leib, D. A., Harrison, T. E., Laslo, K. M., Machalek, M. A., Moorman, N. J., & Virgin, H. W. (1999). Interferons regulate the phenotype of wild-type and mutant herpes simplex viruses in vivo. Journal of Experimental Medicine, 189, 663–672.
Hagglund, R., Van Sant, C., Lopez, P., & Roizman, B. (2002). Herpes simplex virus 1-infected cell protein 0 contains two E3 ubiquitin ligase sites specific for different E2 ubiquitin-conjugating enzymes. Proceedings of the National Academy of Sciences USA, 99, 631–636.
Hagglund, R., & Roizman, B. (2002). Characterization of the novel E3 ubiquitin ligase encoded in exon 3 of herpes simplex virus-1-infected cell protein 0. Proceedings of the National Academy of Sciences USA, 99, 7889–7894.
Halford, W.P., Puschel, R., Rakowski, B. (2010). Herpes simplex virus 2 ICP0 mutant viruses are avirulent and immunogenic: Implications for a genital herpes vaccine. PLoS One, 5(8), e12251.
Everett, R. D., Parsy, M. L., & Orr, A. (2009). Analysis of the functions of herpes simplex virus type 1 regulatory protein ICP0 that are critical for lytic infection and derepression of quiescent viral genomes. Journal of Virology, 83, 4963–4977.
Cliffe, A. R., & Knipe, D. M. (2008). Herpes simplex virus ICP0 promotes both histone removal and acetylation on viral DNA during lytic infection. Journal of Virology, 82, 12030–12038.
Halford, W. P., Weisend, C., Grace, J., Soboleski, M., Carr, D. J., Balliet, J. W., et al. (2006). ICP0 antagonizes stat 1-dependent repression of herpes simplex virus: implications for the regulation of viral latency. Virology Journal, 3, 44.
Hagglund, R., & Roizman, B. (2004). Role of ICP0 in the strategy of conquest of the host cell by herpes simplex virus 1. Journal of Virology, 78, 2169–2178.
Liu, M., Schmidt, E. E., & Halford, W. P. (2010). ICP0 dismantles microtubule networks in herpes simplex virus-infected cells. PLoS One, 5, e10975.
Boutell, C., & Everett, R. D. (2003). The herpes simplex virus type 1 (HSV-1) regulatory protein ICP0 interacts with and ubiquitinates p53. Journal of Biological Chemistry, 278, 36596–36602.
Gu, H., & Roizman, B. (2009). The two functions of herpes simplex virus 1 ICP0, inhibition of silencing by the CoREST/REST/HDAC complex and degradation of PML, are executed in tandem. Journal of Virology, 83, 181–187.
Lomonte, P., Thomas, J., Texier, P., Caron, C., Khochbin, S., & Epstein, A. L. (2004). Functional interaction between class II histone deacetylases and ICP0 of herpes simplex virus type 1. Journal of Virology, 78, 6744–6757.
Daubeuf, S., Singh, D., Tan, Y., Liu, H., Federoff, H. J., Bowers, W. J., et al. (2009). HSV ICP0 recruits USP7 to modulate TLR-mediated innate response. Blood, 113, 3264–3275.
Rickinson, A. B., & Kieff, E. (2001). Epstein-Barr virus. In D. M. Knipe & P. M. Howley (Eds.), Field virology, 4th ed. (pp. 2575–2627). Philadelphia: Lippincott Williams and Wilkins.
Krysan, P. J., Haase, S. B., & Calos, M. P. (1989). Isolation of human sequences that replicate autonomously in human cells. Molecular and Cellular Biology, 9, 1026–1033.
Reisman, D., & Sugden, B. (1986). Transactivation of an Epstein-Barr viral transcriptional enhancer by the Epstein-Barr viral nuclear antigen 1. Molecular and Cellular Biology, 6, 3838–3846.
Holowaty, M. N., Zeghouf, M., Wu, H., Tellam, J., Athanasopoulos, V., Greenblatt, J., et al. (2003). Protein profiling with Epstein-Barr nuclear antigen-1 reveals an interaction with the herpesvirus-associated ubiquitin-specific protease HAUSP/USP7. Journal of Biological Chemistry, 278, 29987–29994.
Holowaty, M. N., Sheng, Y., Nguyen, T., Arrowsmith, C., & Frappier, L. (2003). Protein interaction domains of the ubiquitin-specific protease, USP7/HAUSP. Journal of Biological Chemistry, 278, 47753–47761.
Saridakis, V., Sheng, Y., Sarkari, F., Holowaty, M. N., Shire, K., Nguyen, T., et al. (2005). Structure of the p53 binding domain of HAUSP/USP7 bound to Epstein-Barr nuclear antigen 1 implications for EBV-mediated immortalization. Molecular Cell, 18, 25–36.
Faustrup, H., Bekker-Jensen, S., Bartek, J., Lukas, J., & Mailand, N. (2009). USP7 counteracts SCFbetaTrCP- but not APCCdh1-mediated proteolysis of Claspin. Journal of Cell Biology, 184, 13–19.
Zhang, D., Zaugg, K., Mak, T. W., & Elledge, S. J. (2006). A role for the deubiquitinating enzyme USP28 in control of the DNA-damage response. Cell, 126, 529–542.
Wiltshire, T. D., Lovejoy, C. A., Wang, T., Xia, F., O’Connor, M. J., & Cortez, D. (2010). Sensitivity to poly(ADP-ribose) polymerase (PARP) inhibition identifies ubiquitin-specific peptidase 11 (USP11) as a regulator of DNA double-strand break repair. Journal of Biological Chemistry, 285, 14565–14571.
Song, L., & Rape, M. (2008). Reverse the curse–the role of deubiquitination in cell cycle control. Current Opinion in Cell Biology, 20, 156–163.
Zhou, B. B., & Elledge, S. J. (2000). The DNA damage response: putting checkpoints in perspective. Nature, 408, 433–439.
Bartek, J., & Lukas, J. (2007). DNA damage checkpoints: from initiation to recovery or adaptation. Current Opinion in Cell Biology, 19, 238–245.
Mailand, N., Falck, J., Lukas, C., Syljuasen, R. G., Welcker, M., Bartek, J., et al. (2000). Rapid destruction of human Cdc25A in response to DNA damage. Science, 288, 1425–1429.
Busino, L., Donzelli, M., Chiesa, M., Guardavaccaro, D., Ganoth, D., Dorrello, N. V., et al. (2003). Degradation of Cdc25A by beta-TrCP during S phase and in response to DNA damage. Nature, 426, 87–91.
Kumagai, A., & Dunphy, W. G. (2000). Claspin, a novel protein required for the activation of Chk1 during a DNA replication checkpoint response in Xenopus egg extracts. Molecular Cell, 6, 839–849.
Mailand, N., Bekker-Jensen, S., Bartek, J., & Lukas, J. (2006). Destruction of Claspin by SCFbetaTrCP restrains Chk1 activation and facilitates recovery from genotoxic stress. Molecular Cell, 23, 307–318.
Mamely, I., van Vugt, M. A., Smits, V. A., Semple, J. I., Lemmens, B., Perrakis, A., et al. (2006). Polo-like kinase-1 controls proteasome-dependent degradation of Claspin during checkpoint recovery. Current Biology, 16, 1950–1955.
Peschiaroli, A., Dorrello, N. V., Guardavaccaro, D., Venere, M., Halazonetis, T., Sherman, N. E., et al. (2006). SCFbetaTrCP-mediated degradation of Claspin regulates recovery from the DNA replication checkpoint response. Molecular Cell, 23, 319–329.
Bassermann, F., Frescas, D., Guardavaccaro, D., Busino, L., Peschiaroli, A., & Pagano, M. (2008). The Cdc14B-Cdh1-Plk1 axis controls the G2 DNA-damage-response checkpoint. Cell, 134, 256–267.
Vogel, C., Hager, C., & Bastians, H. (2007). Mechanisms of mitotic cell death induced by chemotherapy-mediated G2 checkpoint abrogation. Cancer Research, 67, 339–345.
Koniaras, K., Cuddihy, A. R., Christopoulos, H., Hogg, A., & O’Connell, M. J. (2001). Inhibition of Chk1-dependent G2 DNA damage checkpoint radiosensitizes p53 mutant human cells. Oncogene, 20, 7453–7463.
Zachos, G., Rainey, M. D., & Gillespie, D. A. (2003). Chk1-deficient tumour cells are viable but exhibit multiple checkpoint and survival defects. EMBO Journal, 22, 713–723.
Lapenna, S., & Giordano, A. (2009). Cell cycle kinases as therapeutic targets for cancer. Nature Reviews Drug Discovery, 8, 547–566.
Oh, Y. M., Yoo, S. J., & Seol, J. H. (2007). Deubiquitination of Chfr, a checkpoint protein, by USP7/HAUSP regulates its stability and activity. Biochemical and Biophysical Research Communications, 357, 615–619.
Scolnick, D. M., & Halazonetis, T. D. (2000). Chfr defines a mitotic stress checkpoint that delays entry into metaphase. Nature, 406, 430–435.
Oh, Y. M., Kwon, Y. E., Kim, J. M., Bae, S. J., Lee, B. K., Yoo, S. J., et al. (2009). Chfr is linked to tumour metastasis through the downregulation of HDAC1. Nature Cell Biology, 11, 295–302.
Hussain, S., Zhang, Y., & Galardy, P. J. (2009). DUBs and cancer: the role of deubiquitinating enzymes as oncogenes, non-oncogenes and tumor suppressors. Cell Cycle, 8, 1688–1697.
Colland, F., Formstecher, E., Jacq, X., Reverdy, C., Planquette, C., Conrath, S., et al. (2009). Small-molecule inhibitor of USP7/HAUSP ubiquitin protease stabilizes and activates p53 in cells. Molecular Cancer Therapeutics, 8, 2286–2295.
Guedat, P., Boissy, G., Borg-Capra, C., Colland, F., Daviet, L., Formstecher, E., Jacq, X., Rain, J.C., Delansorne, R., Vallese, S., Colombo, M. (2007). Novel cysteine protease inhibitors and their therapeutic applications. Paris: Hybrigenics SA. US20070032499.
Nicholson, B., Leach, C. A., Goldenberg, S. J., Francis, D. M., Kodrasov, M. P., Tian, X., et al. (2008). Characterization of ubiquitin and ubiquitin-like-protein isopeptidase activities. Protein Science, 17, 1035–1043.
Leach, C. A., Tian, X., Mattern, M. R., & Nicholson, B. (2009). Detection and characterization of SUMO protease activity using a sensitive enzyme-based reporter assay. Methods in Molecular Biology, 497, 269–281.
Cao, P., Weinstock, J., Kingsbury, W.D., Leach, C.A., Kizhakkethil George, S.K., Nicholson, B. (2009). Antineoplastic compounds, compositions and methods. Malvern, PA: Progenra Inc. PCT/US2010/029358.
Acknowledgments
We wish to thank our collaborators and our colleagues at Progenra for their many insightful comments on the roles of USP7 in disease. This study was supported in part by NIH grants CA115205 and DK071391.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nicholson, B., Suresh Kumar, K.G. The Multifaceted Roles of USP7: New Therapeutic Opportunities. Cell Biochem Biophys 60, 61–68 (2011). https://doi.org/10.1007/s12013-011-9185-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-011-9185-5