Abstract
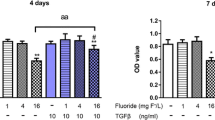
Little is known about the combined effect of fluoride (F) and arsenic (As) on bone metabolism. This study aims to explore the effect of co-exposure to F and As on the expressions of TNF receptor-associated factor 6 (TRAF-6), nuclear factor-kappa B (NF-κB), and the related factors in cell and animal experiments. With the rats exposed to different doses of F, As, and combined F-As, we found that F exposure doses were positively correlated with the protein expression of receptor activator of nuclear factor-kappa B ligand (RANKL), receptor activator of nuclear factor-kappa B (RANK), TRAF-6, NF-κB, and nuclear factor of activated T cells (NFAT-c1) (P < 0.001). As exposure doses were negatively correlated with RANK, TRAF-6, NF-κB, and NFAT-c1 (P < 0.001). The effect of F and As interaction on the protein expression of RANKL, TRAF-6, NF-κB, and NFAT-c1 was significant in bone tissue (P < 0.05). In the cellular experiment, F could promote the mRNA expression of RANK, TRAF-6, and NFAT-c1. A higher concentration of As could inhibit the mRNA expression of Tartrate-resistant acid phosphatase (TRAP), RANK, TRAF-6, and NFAT-c1. The effect of F and As interaction on the mRNA expression of TRAP, RANK, TRAF-6, and NFATc1 in osteoclasts was significant (P < 0.001). In conclusion, the expression of TRAF-6 and NF-κB pathway was affected by F and As co-exposure in osteogenic differentiation, and As could antagonize the promoting effect of F on the expression of TRAF-6, TRAP, RANKL, RANK, NF-κB, and NFAT-c1 in these exposure levels. These results could provide a scientific basis for understanding the interaction of F and As in bone formation.





Similar content being viewed by others
Data Availability
Data are available from the corresponding author upon reasonable request.
References
González-Horta C, Ballinas-Casarrubias L, Sánchez-Ramírez B, Ishida MC, Barrera-Hernández A, Gutiérrez-Torres D, Zacarias OL, Saunders RJ, Drobná Z, Mendez MA, García-Vargas G, Loomis D, Stýblo M, Del Razo LM (2015) A concurrent exposure to arsenic and fluoride from drinking water in Chihuahua, Mexico. Int J Environ Res Public Health 12(5):4587–4601
Podgorski J, Berg M (2020) Global threat of arsenic in groundwater. Science 368(6493):845–850. https://doi.org/10.1126/science.aba1510
Yadav AK, Kaushik CP, Haritash AK, Kansal A, Rani N (2006) Defluoridation of groundwater using brick powder as an adsorbent. J Hazard Mater 128(2–3):289–293. https://doi.org/10.1016/j.jhazmat.2005.08.006
Wang W, Xu J, Liu K, Liu X, Li C, Cui C, Zhang Y, Li H (2013) Suppression of Sclerostin and Dickkopf-1 levels in patients with fluorine bone injury. Environ Toxicol Pharmacol 35(3):402–407. https://doi.org/10.1016/j.etap.2013.01.005
Nakamoto T, Rawls HR (2018) Fluoride exposure in early life as the possible root cause of disease in later life. J Clin Pediatr Dent 42(5):325–330. https://doi.org/10.17796/1053-4625-42.5.1
Aybar Odstrcil AC, Carino SN, Ricci JC, Mandalunis PM (2010) Effect of arsenic in endochondral ossification of experimental animals. Exp Toxicol Pathol: Off J Ges fur Toxikologische Pathologie 62(3):243–249. https://doi.org/10.1016/j.etp.2009.04.001
Hu Y-C, Cheng H-L, Hsieh B-S, Huang L-W, Huang T-C, Chang K-L (2012) Arsenic trioxide affects bone remodeling by effects on osteoblast differentiation and function. Bone 50(6):1406–1415. https://doi.org/10.1016/j.bone.2012.03.012
Wu C-T, Lu T-Y, Chan D-C, Tsai K-S, Yang R-S, Liu S-H (2014) Effects of arsenic on osteoblast differentiation in vitro and on bone mineral density and microstructure in rats. Environ Health Perspect 122(6):559–565. https://doi.org/10.1289/ehp.1307832
Jia L, Jin T-Y Combined effect of fluoride and arsenate on gene expression of osteoclast differentiation factor and osteoprotegerin. Biomed Environ Sci 19(5):375–379. https://pubmed.ncbi.nlm.nih.gov/17190191. Accessed 20 Jul 2022
Hong F, Zheng C, Xu D-g, Qian Y-l (2013) Chronic combined effects of fluoride and arsenite on the Runx2 and downstream related factors of bone metabolism in rats. Zhonghua Yu Fang Yi Xue Za Zhi 47(9):794–798. https://doi.org/10.3760/cma.j.issn.0253-9624.2013.09.006
Zeng Q-b, Xu Y-y, Yu X, Yang J, Hong F, Zhang A-h (2014) Arsenic may be involved in fluoride-induced bone toxicity through PTH/ PKA/AP1 signaling pathway. Environ Toxicol Pharmacol 37(1):228–233. https://doi.org/10.1016/j.etap.2013.11.027
Park E, Lee CG, Lim E, Hwang S, Yun SH, Kim J, Jeong H, Yong Y, Yun S-H, Choi CW, Jin H-S, Jeong S-Y (2021) Osteoprotective effects of loganic acid on osteoblastic and osteoclastic cells and osteoporosis-induced mice. Int J Mole Sci 22(1):233. https://doi.org/10.3390/ijms22010233
Ma Z, Li S, Sun Y (2020) Effect of enhanced masticatory force on OPG, RANKL and MGF in alveolar bone of ovariectomized rats. J Appl Oral Sci 28:e20190409. https://doi.org/10.1590/1678-7757-2019-0409
Qiao L, Liu X, He Y, Zhang J, Huang H, Bian W, Chilufya MM, Zhao Y, Han J (2021) Progress of signaling pathways, stress pathways and epigenetics in the pathogenesis of skeletal fluorosis. Int J Mole Sci 22(21):11932. https://doi.org/10.3390/ijms222111932
Chung JY, Lu M, Yin Q, Lin S-C, Wu H (2007) Molecular basis for the unique specificity of TRAF6. Adv Exp Med Biol 597:122–130. https://doi.org/10.1007/978-0-387-70630-6_10
Lomaga MA, Yeh WC, Sarosi I, Duncan GS, Furlonger C, Ho A, Morony S, Capparelli C, Van G, Kaufman S, van der Heiden A, Itie A, Wakeham A, Khoo W, Sasaki T, Cao Z, Penninger JM, Paige CJ, Lacey DL, Dunstan CR, Boyle WJ, Goeddel DV, Mak TW (1999) TRAF6 deficiency results in osteopetrosis and defective interleukin-1, CD40, and LPS signaling. Genes Dev 13(8):1015–1024. https://doi.org/10.1101/gad.13.8.1015
Iotsova V, Caamaño J, Loy J, Yi Y, Lewin A, Bravo R (1997) Osteopetrosis in mice lacking NF-|[kappa]|B1 and NF-|[kappa]|B2. Nat Med 3(11):1285. https://doi.org/10.1038/nm1197-1285
Xu S, Li S, Liu X, Tan K, Zhang J, Li K, Bai X, Zhang Y (2021) Rictor is a novel regulator of TRAF6/TRAF3 in osteoclasts. J Bone Mineral Res: Off J Am Soc Bone Mineral Res 36(10):2053–2064. https://doi.org/10.1002/jbmr.4398
van Dam PA, Verhoeven Y, Trinh XB (2020) The non-bone-related role of RANK/RANKL signaling in cancer. Adv Exp Med Biol 1277:53–62. https://doi.org/10.1007/978-3-030-50224-9_3
Li R, Gong Z, Yu Y, Niu R, Bian S, Sun Z (2022) Alleviative effects of exercise on bone remodeling in fluorosis mice. Biol Trace Elem Res 200(3):1248–1261. https://doi.org/10.1007/s12011-021-02741-y
Liu X-L, Song J, Liu K-J, Wang W-P, Xu C, Zhang Y-Z, Liu Y (2015) Role of inhibition of osteogenesis function by Sema4D/Plexin-B1 signaling pathway in skeletal fluorosis in vitro. J Huazhong Univ Sci Technolog Med Sci 35(5):712–715. https://doi.org/10.1007/s11596-015-1495-1
Gao M, Sun L, Xu K, Zhang L, Zhang Y, He T, Sun R, Huang H, Zhu J, Zhang Y, Zhou G, Ba Y (2020) Association between low-to-moderate fluoride exposure and bone mineral density in Chinese adults: Non-negligible role of RUNX2 promoter methylation. Ecotoxicol Environ Saf 203:111031. https://doi.org/10.1016/j.ecoenv.2020.111031
Sun R, Zhou G, Liu L, Ren L, Xi Y, Zhu J, Huang H, Li Z, Li Y, Cheng X, Ba Y (2020) Fluoride exposure and CALCA methylation is associated with the bone mineral density of Chinese women. Chemosphere 253:126616. https://doi.org/10.1016/j.chemosphere.2020.126616
Yang H, Yao J, He X, Li B (2011) Experimental study of combined effects of fluoride and arsenic on bone mineral density in rats. Chin J Osteoporo 17(05):393–396. https://doi.org/10.3969/j.issn.1006-7108.2011.05.006 (in Chinese)
Yang H, Liu B, Luan L, Sun H, Zhang P, Li B (2012) Experimental study of the effect of arsenic poisoning on bone mineral density in rats. Chin J Osteoporos 18(02):146–148. https://doi.org/10.3969/j.issn.1006-7108.2012.02.012 (in Chinese)
Li H, Qin Z, Wang B, Hu J, Hong F (2018) Role of osteoprotegerin and receptor activator of nuclear factor-KB ligand in bone toxicity in rats co-exposed to fluoride and arsenite. Chin J Eindemiol 37(06):461–466. https://doi.org/10.3760/cma.j.issn.2095-4255.2018.06.007 (in Chinese)
Jiang N, Guo F, Xu W, Zhang Z, Jin H, Shi L, Zhang X, Gao J, Xu H (2020) Effect of fluoride on osteocyte-driven osteoclastic differentiation. Toxicology 436:152429. https://doi.org/10.1016/j.tox.2020.152429
Matsuo K, Galson DL, Zhao C, Peng L, Laplace C, Wang KZQ, Bachler MA, Amano H, Aburatani H, Ishikawa H, Wagner EF (2004) Nuclear factor of activated T-cells (NFAT) rescues osteoclastogenesis in precursors lacking c-Fos. J Biol Chem 279(25):26475–26480. https://doi.org/10.1074/jbc.M313973200
Kim MS, Yang Y-M, Son A, Tian YS, Lee S-I, Kang SW, Muallem S, Shin DM (2010) RANKL-mediated reactive oxygen species pathway that induces long lasting Ca2+ oscillations essential for osteoclastogenesis. J Biol Chem 285(10):6913–6921. https://doi.org/10.1074/jbc.M109.051557
Takayanagi H (2007) Osteoimmunology: shared mechanisms and crosstalk between the immune and bone systems. Nat Rev Immunol 7(4):292–304. https://doi.org/10.1038/nri2062
Xie Y, Yu Y, Wan L, Chen X (2012) Effect of fluoride on expression of CaN mRNA and protein bone tissue of rats. Chin J Pathol 11:761–764. https://doi.org/10.3760/cma.j.issn.0529-5807.2012.11.011 (in Chinese)
Yang X, Jin Z, Zhang J, Qin Z, Wang B, Zhan S, Feng H (2019) Effects of combined exposure to fluoride and arsenic on expressions of TRAF-6, NF-κB1, NFATc1, and TRAP mRNA in a co-culture system of osteoblasts and osteoclasts. J Environ Occup Med 36(01):26–32. https://doi.org/10.13213/j.cnki.jeom.2019.18512 (in Chinese)
Chou H, Grant MP, Bolt AM, Guilbert C, Plourde D, Mwale F, Mann KK (2021) Tungsten increases sex-specific osteoclast differentiation in murine bone. Toxicol Sci 179(1):135–146. https://doi.org/10.1093/toxsci/kfaa165
Zeng Q, Xu Y, Yu X, Yang J, Hong F, Zhang A (2019) Silencing GSK3β instead of DKK1 can inhibit osteogenic differentiation caused by co-exposure to fluoride and arsenic. Bone 123:196–203. https://doi.org/10.1016/j.bone.2019.03.016
Tian X, Xie J, Chen X, Dong N, Feng J, Gao Y, Tian F, Zhang W, Qiu Y, Niu R, Ren X, Yan X (2020) Deregulation of autophagy is involved in nephrotoxicity of arsenite and fluoride exposure during gestation to puberty in rat offspring. Arch Toxicol 94(3):749–760. https://doi.org/10.1007/s00204-019-02651-y
Yang X, Hong F, Xie W, Zhang J, Jin Z, Qin Z (2019) Effects of joint exposure to fluoride and arsenic on OPG/RANKL regulating osteoclast differentiation. J Environ Health 36(04):305–310. https://doi.org/10.16241/j.cnki.1001-5914.2019.04.006 (in Chinese)
Funding
This study was supported by the National Natural Science Foundation of China (No. 82173566).
Author information
Authors and Affiliations
Contributions
Chan Nie analyzed the data, prepared figures and tables, and wrote the paper. Bingjie Wang, Hao Li, and Xing Yang designed and performed the experiments, and prepared figures and tables. Feng Hong conceived the experiments and reviewed and edited the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics Approval
This study was performed according to the national and institutional guidelines and approved by the Animal Experimental Ethical Committee of Guizhou Medical University (approval number: 1413060).
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nie, C., Hu, J., Wang, B. et al. Effects of Co-exposure to Fluoride and Arsenic on TRAF-6 Signaling and NF-κB Pathway of Bone Metabolism. Biol Trace Elem Res 201, 4447–4455 (2023). https://doi.org/10.1007/s12011-022-03508-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03508-9